Abstract
A 61-year-old female with systemic lupus erythematosus presented with refractory discoid lupus. Despite treatment with numerous immunosuppressive therapies, including prednisone, hydroxychloroquine and mycophenolate mofetil, her cutaneous lupus remained active and significantly impacted her quality of life. Introduction of anifrolumab led to remarkable improvement, highlighting its potential for refractory discoid lupus erythematosus.
Keywords
Introduction
Discoid lupus erythematosus (DLE) is the most common cutaneous manifestation of chronic cutaneous systemic lupus erythematosus (SLE). 1 Chronic DLE can be refractory to traditional measures including corticosteroids, topical calcineurin inhibitors and systemic therapies such as antimalarials, azathioprine, thalidomide and mycophenolate mofetil (MMF). 2 Biologic therapies can be trialled for recalcitrant DLE. Anifrolumab, a monoclonal antibody targeting type I interferon receptor subunit 1, has shown efficacy in managing refractory SLE-associated DLE in clinical trials. 3 We present a case that demonstrates the clinical utility of anifrolumab in treating refractory cutaneous lupus.
Case report
A 61-year-old female was initially diagnosed with SLE, confirmed by a positive anti nuclear antibody (ANA), anti-Smith antibodies, low C4 levels and lupus anticoagulant. Her symptoms manifested as discoid lupus, arthralgias, sicca symptoms and biopsy-confirmed membranous lupus nephropathy. Her previous treatments included various prednisone tapering regimens, hydroxychloroquine (discontinued due to hives), azathioprine (discontinued due to vomiting and diarrhoea) and cyclosporine. With a rise in creatinine and nephrotic range proteinuria on routine blood work, a repeat renal biopsy was done and showed membranous lupus nephropathy without chronicity or proliferative changes. She was subsequently started on MMF 500 mg BID for management of her DLE and lupus nephritis, her renal parameters remained stable on MMF.
Following this, she presented with a chief complaint of recurrent flare-ups of DLE despite being on MMF. She continued to have persistent symptoms of painful scalp erosions associated with scarring alopecia, tender oral ulcers causing sloughing, myalgias with joint stiffness, ulceration and bruising of skin in response to pressure, and a photosensitive rash.
On clinical examination, there was evidence of ulcerative plaques on the scalp associated with scarring alopecia and decreased hair density (Figure 1(a) and (b)). There were also well-demarcated light brown to violaceous atrophic plaques present on the forehead (Figure 2(a)), conchal bowls (Figure 2(b)) and post-auricular regions. There were multiple superficial ulcerated lesions over the upper lip (Figure 2(d)), hands and legs. Erosive lesions were also present over the distal interphalangeal joints and proximal phalanges (Figure 3(a)). Darker coloured lesions found on the dorsal aspect of the hands, breast and lumbosacral region were in keeping with post-inflammatory hyperpigmentation.
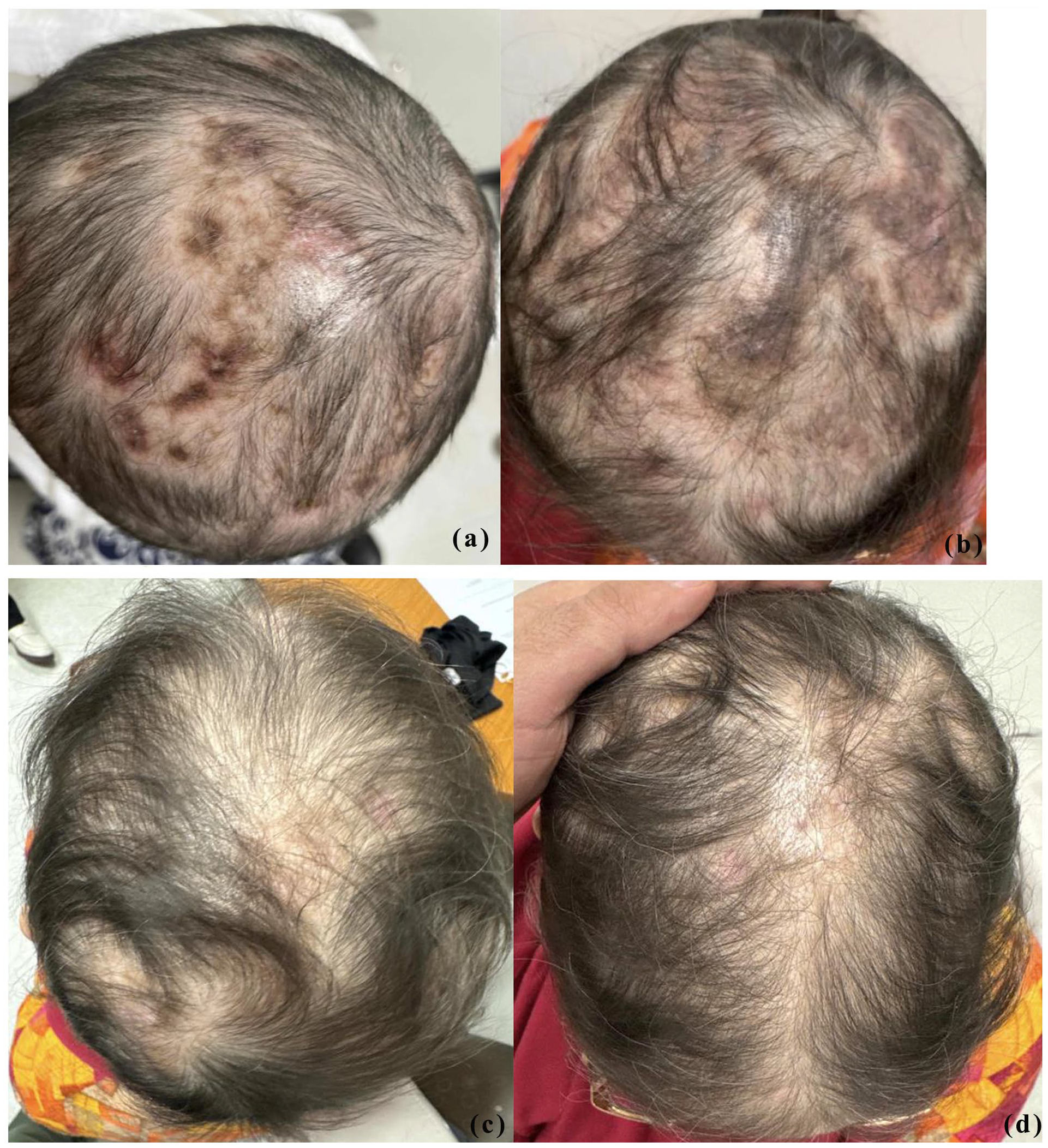
Comparison of the lesions on the scalp before and after treatment with four injections of anifrolumab. (a, b) Appearance of the scalp prior to treatment. (c, d) Appearance of the scalp after treatment.

Comparison of the discoid lupus involving the face before and after treatment with four injections of anifrolumab. (a) Forehead prior to treatment. (b) Right conchal bowl prior to treatment. (c) Right conchal bowl after treatment. (d) Perioral region prior to treatment. (e) Perioral region after treatment.

Comparison of the lesions on phalanges before and after treatment with four injections of anifrolumab. (a) Phalanges prior to treatment. (b) Phalanges after treatment.
Given the ongoing disease burden of DLE refractory to first-line agents and systemic therapy, she was initiated on anifrolumab (Saphnelo®, AstraZeneca AB, 02522845 (Canada) AQ) in conjunction with a decreased dose of MMF 500 mg QD. She had regular follow-up in clinic and routine blood work, including complete blood count, liver function tests, creatinine and an autoimmune panel every 3 months. In addition, she was counselled on sun safety and regular sunscreen use at every visit.
After receiving four infusions of anifrolumab, the patient endorsed significant improvement in overall symptoms and quality of life. There was a significant reduction in her scalp inflammation (Figure 1(c) and (d)), conchal bowls and upper lip (Figure 2(c) and (e)), along with complete resolution of the cutaneous lesions involving the hand (Figure 3(b)). In addition, the patient reported a reduction in pain and improvement in daily functioning.
Discussion
Our case highlights the use of anifrolumab as a potential treatment for DLE that is refractory to conventional therapies. Current management for DLE includes photoprotective measures and the use of corticosteroids, topical calcineurin inhibitors, antimalarials and other immunosuppressive agents. In common cases where DLE is refractory to these treatments, current biologic therapies available target type I interferon, which are key mediators for the pathogenesis of SLE including DLE. 4 Anifrolumab, a monoclonal antibody against IFN alpha receptor subunit-1, FDA-approved for SLE in 2021, was found to be efficacious in the management of cutaneous lupus refractory to mainstay treatments.4,5
In our case, previous treatments were discontinued either due to side effects or minimal treatment response. She was subsequently started on MMF 500 mg BID for management of her DLE and lupus nephritis. Despite being on MMF, she continued to experience ongoing scalp, mucosal membrane and widespread involvement, prompting initiation of anifrolumab. This was the preferred biologic treatment given that patients with moderate to severe SLE who were administered anifrolumab reported a lower SLE Disease Activity Index 2000. 6 Additionally, anifrolumab is known to be more effective when cutaneous manifestations predominate and is associated with a rapid onset of action compared to belimumab, another biologic option. 7
While on anifrolumab and a decreased dose of MMF 500 mg QD, our patient experienced a marked improvement of systemic symptoms and cutaneous lesions after just four infusions of anifrolumab. The findings of our case are supported by similar reports demonstrating the benefits of anifrolumab on the resolution of cutaneous manifestations of SLE.5,7–9 The safety profile of anifrolumab has been deemed favourable, although it was observed to worsen renal disease in one patient, suggesting its limited use in lupus nephritis. 4 However, in our case, the patient was monitored closely and demonstrated an unremarkable change in baseline blood work, including creatinine levels. Our case demonstrates the potential use of anifrolumab for the clearance of cutaneous lupus without exacerbating underlying renal concerns in the setting of lupus nephritis.
In addition, it was found that anifrolumab reduced the amount of hair loss and increased hair re-growth in patients experiencing alopecia due to DLE. 10 This aligns with our case, where our patient exhibited regression of ulcerations on the scalp and subsequent hair re-growth in areas previously affected. This further supports the use of anifrolumab as an effective treatment option for the improvement of cutaneous lesions involving the scalp in refractory DLE.
Conclusion
In summary, we present a 61-year-old female diagnosed with SLE associated with refractory DLE and other systemic symptoms, who had marked benefit using anifrolumab (Saphnelo®). This case highlights the potential of anifrolumab as a promising therapeutic option for refractory DLE. Despite its limited evidence in lupus nephritis, the notable improvement in DLE lesions underscores its utility for cutaneous manifestations. Close monitoring remains essential to evaluate long-term outcomes and manage potential adverse effects. Further studies are warranted to establish its role in broader manifestations of SLE.
Footnotes
Consent for publication
Informed consent was obtained for publishing the included clinical images.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
