Abstract
Pancreatic neuroendocrine tumors are an uncommon group of insidious neoplasms that are often diagnosed in the advanced stages. Diagnosing pancreatic neuroendocrine tumors involves proper biopsy technique and immunohistochemistry, while treatments include targeted therapies and systemic chemotherapy. When diagnosed early, patients with localized disease generally have a positive prognosis. We share a case of a 56-year-old man presenting to the emergency department with acute abdominal pain found to have poorly differentiated pancreatic neuroendocrine tumor with metastasis on repeat endoscopic ultrasound-guided fine needle biopsy. Despite multiple treatment changes, sustained tumor presence led to numerous complications and ultimately patient death. This case highlights the nuances and challenges of diagnosing and treating a rare pancreatic neuroendocrine tumor, such as the importance of biopsy technique.
Introduction
Neuroendocrine tumors (NETs) are a heterogenous set of neoplasms that arise typically from the gastrointestinal tract, lungs, and pancreas. 1 Together, they represent 0.46% of gastrointestinal and bronchopulmonary malignancies with rising incidence potentially due to increased detection. 2 Pancreatic NETs (panNETs) are a subset of NETs originating from islet cells with an incidence of 1.0 case/100,000/year. 3 PanNETs are classified based on the extent of cell differentiation, location, metastasis, production of hormones (known as functional panNETs), and immunohistochemical markers. 3 Treat-ment of panNETs is challenging due to the aggressive nature of the disease and late presentation with extensive metastasis. This typically renders care teams to utilize systemic chemotherapy such as carboplatin and etoposide as a first-line regimen. 4
We report a unique case of appropriately utilizing repeat endoscopic ultrasound-guided fine needle biopsy (EUS-FNB) for accurate and timely diagnosis of an indeterminate lesion later confirmed to be a poorly differentiated, nonfunctional panNET.
Case presentation
A 56-year-old Hispanic male with a history of type 2 diabetes mellitus and hypertension presented to the emergency department with progressive, intermittent right and left upper quadrant pain for 2 days associated with gastroesophageal reflux disease (GERD). The patient reported increased eructation over the past 3 weeks, but denied any nausea, vomiting, diarrhea, or hematochezia. He also endorsed an unintentional 20 lb weight loss over the past 2 months. The patient denied smoking but confirmed drinking alcohol two to four times per month. His family history was negative for gastrointestinal malignancies and pathologies.
Lab workup revealed elevated ALT (1553 U/L), AST (1738 U/L), ALP (311 U/L), and direct bilirubin (0.5 mg/dL) levels. Total bilirubin (1.0 mg/dL), albumin (4.4 g/dL), lipase (52 U/L), hemoglobin (13.8 g/dL), mean corpuscular volume (91.7 fL), and WBC count (8.20 × 103/μL) were all within range. Subsequent hepatitis A, B, and C serologies were also negative. Right upper quadrant ultrasound displayed a 10 mm thickening of the gallbladder wall with likely pericholecystic fluid, and the common bile duct (CBD) was dilated to 9 mm. Furthermore, two hypoechoic masses were noted upon pancreatic examination. Evaluation with magnetic resonance cholangiopancreatography (MRCP) revealed head and uncinate pancreatic mass lesions (4.7 × 3.5 cm) causing compression of the distal section of the CBD and dilation of the proximal region (Figure 1). Additional findings included an enlarged peripancreatic lymph node of 2.1 × 1.5 cm in size. Due to these findings, an EUS and endoscopic retrograde cholangiopancreatography (ERCP) were performed.

MRCP portraying pancreatic head/uncinate lesion in (a) T1 transverse and (b) T2 coronal imaging as marked by red arrows.
On initial EUS-FNB, an irregular hypoechoic mass measuring 4.0 cm by 3.4 cm was visualized with sonographic evidence of invasion into the hepatic artery and inferior vena cava. A 2.2 × 1.8 cm peripancreatic lymph node was also visualized. Three total biopsy samples were taken of the hypoechoic mass using 22- and 25-gauge needles, respectively. One pass with the 22-gauge needle was made for the lymph node. On ERCP, a biliary stricture was found in the middle third of the main bile duct. A sphincterotomy was performed and brushing of cells was obtained for cytology. A metal stent was placed into the CBD with subsequent improvement of liver function tests.
Pathology results from the first EUS-FNB were positive for atypical cells in the pancreatic head, peripancreatic lymph node, and CBD, however, malignancy status was inconclusive and a repeat EUS-FNB was recommended. Unfortunately, due to lab turnaround time and scheduling conflicts, repeat EUS-FNB occurred 3 weeks later. During the second EUS-FNB, five total passes of the mass were made using a 22-gauge needle. Additionally, two passes of the lymph node were made with the 22-gauge needle (Figure 2). Both procedures used a Franseen-tip needle (Acquire; Boston Scientific, Natick, MA, USA). The pathology report from the repeat EUS-FNB exhibited positive morphology for a high grade panNET (small cell carcinoma) with extensive crushing artifact (Figure 3). Additionally, the tumor immunohistochemistry was positive for synaptophysin and displayed 90% Ki-67 index (Figure 3) consistent with an aggressive stage 3 classification. No mitosis or necrosis was appreciated in the sample. In both biopsies, FNB samples were prepped using cell block for histological evaluation. 5 A full immunohistochemical profile of the tumor is found in Table 1.

Repeat EUS-FNB portraying (a) poorly defined hypoechoic pancreatic head mass and (b) well-defined peripancreatic lymph node with needle.

Histopathology of repeat EUS-FNB sample with (a) H&E stain at 20× magnification, (b) H&E stain at 40× magnification, (c) synaptophysin IHC stain at 20× magnification, and (d) Ki-67 IHC stain at 20× magnification.
Immunohistochemical results from EUS-FNB samples.

EUS-FNB: endoscopic ultrasound-guided fine needle biopsy.
The patient was initially planned to receive chemotherapy with cisplatin and etoposide, alongside radiation therapy beginning in the second cycle. However, pretreatment staging with PET-CT scan presented additional hypermetabolic para-aortic, retrocaval, retro-crural, paraesophageal, mediastinal, and supraclavicular lymphadenopathy requiring changes in his treatment (Figure 4). Radiation therapy was excluded and a more aggressive, systemic six cycle chemotherapy regimen consisting of carboplatin, etoposide, and atezolizumab was started. This was followed by two cycles of atezolizumab for maintenance. During this regimen, the patient responded well with improvement in symptoms, labs, and lymphadenopathy. The patient’s course was later complicated during the third cycle by a Klebsiella oxytoca infection of the prior biliary stent requiring a 3-day hospital stay with stent replacement and antibiotics.
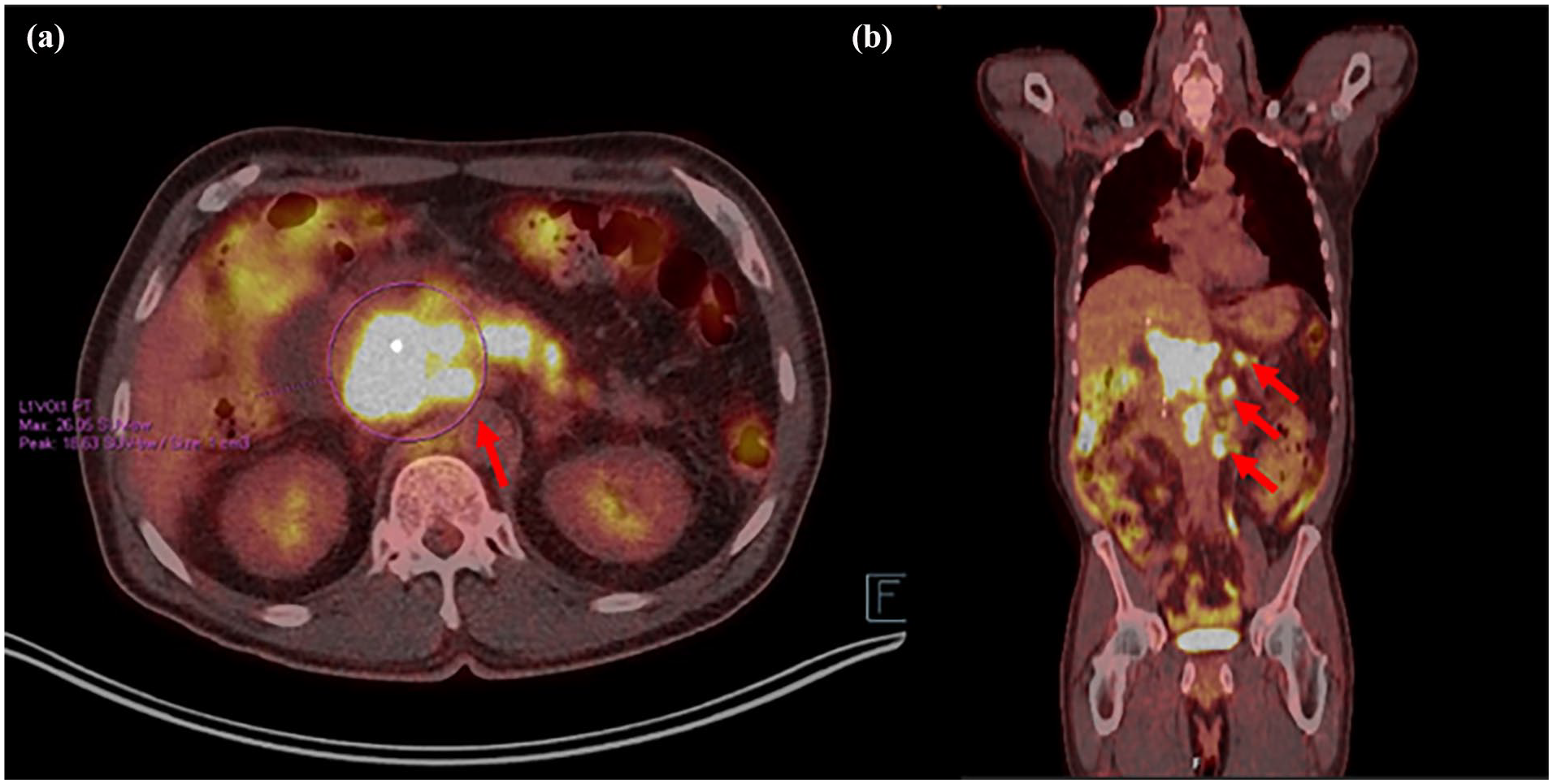
PET-CT scan results prior to chemotherapy initiation showing (a) hypermetabolic pancreatic head/uncinate mass and (b) multiple hypermetabolic lymph nodes as marked by red arrows.
Toward the end of the regimen the patient re-presented with abdominal pain requiring hospitalization and an increase in the primary tumor was noted on repeat CT imaging (4.6 cm). During this hospitalization, oncology agreed to start a chemotherapy regimen consisting of four cycles of lurbinectedin with palliative radiation. Subsequently, the patient was then re-hospitalized after the first cycle for neutropenic fever and ascites. After treatment he was discharged and cleared for cancer treatment. Later, the patient was hospitalized after his fourth cycle due to spontaneous bacterial peritonitis. He underwent a repeat ERCP for stenting of the right intrahepatic duct and CBD. Despite this, liver function tests (LFTs) and bilirubin did not return to baseline, thus preventing further chemotherapy. The patient was ultimately discharged to home hospice and passed away 1 year after the initial diagnosis.
Discussion
This case underscores the diagnostic and treatment challenges of an advanced, poorly differentiated, nonfunctional panNET. PanNETs have a median age of onset of 59 and represent only 1%–2% of pancreatic neoplasms, though the incidence is rising. Risk factors for panNETs include autosomal dominant genetic conditions such as multiple endocrine neoplasia type 1, von Hippel–Lindau syndrome, neurofibromatosis type 1, and tuberous sclerosis complex. 6 Previous literature from the SEER-18 NCI database demonstrates a median overall survival of 68 months and 5-year-survival rate of 28.13% for metastatic panNETs. 7 Non-functio-nal panNETs, specifically, are difficult to diagnose due to indistinct symptoms such as abdominal pain, weight loss, nausea, and anorexia. 8 Therefore, this case report supports the recent trend in broader application of imaging for malignancy detection especially for panNETs.
Though panNETs are typically found incidentally on a CT scan, the helical triple-phase contrast-enhanced CT serves as the best option for more aggressive cases. MRI with contrast, PET-CT, and more recently EUS all contain value for further imaging. 1 In this patient, the incidental finding of lesions on the pancreas led to MRCP and later two EUS-FNB, highlighting the possibility of sampling error. While EUS-guided tissue acquisition procedures are heralded for their high sensitivity, they do face limitations as marked by their negative predictive values, ranging from 51% to 91% for pancreatic lesions when compared to gold standard surgical resections. 9
Two factors that may determine diagnostic yield are needle gauge and number of samples taken. In this patient, the original EUS-FNB was performed with three passes total with both a 22- and 25-G needle. The results were atypical, but inconclusive, requiring repeat sampling. On repeat EUS-FNB, a 22-G needle was used with five passes of the mass which resulted in the diagnosis. Although the 25-G needle provides a safer option compared to a 22-G, it may limit diagnostic yield for histology studies, therefore, we recommend the use of larger needle sizes. 10 A study reviewing 463 patients who underwent EUS-FNB of solid pancreatic lesions concluded that larger needle caliper size affected diagnostic adequacy, while number of passes did not. 11 Furthermore, two separate studies also concluded that a 20-G was superior in retrieving samples as compared to a 25-G and should be preferentially used underscoring the importance of needle size.12,13 Typically, EUS-FNB allows for higher tissue acquisition and guidelines suggest two to three passes may be adequate, but due to the softer and more rubbery tissue of neuroendocrine cells, we suggest that more passes may be necessary. As a result, in addition to a larger gauge needle, we also recommend greater than four passes especially if using a smaller gauge, to increase sample yield.14,15
If repeat EUS sampling is necessary, there are no clear guidelines on how quickly this should be performed. Some factors to consider are patient health status, location, staffing, and resources availability. Based on this case we determine within 3 weeks is optimal, but sooner timing would also be advised given the aggressive nature of this neoplasm. A high degree of clinical suspicion is necessary and clinical expertise such as tactile changes felt during EUS biopsy may aid an endoscopist to pursue repeating testing in an expedited manner.
The treatment plan for an aggressive panNET consists of at least partial surgical resection in addition to pharmacologic therapy. First-line drugs for advanced panNETs include temozolomide, capecitabine, cisplatin, etoposide, 5-FU, and streptozocin. 16 Locally confined panNETs not eligible for surgery may be appropriate candidates for ethanol injection of radio-ablation via EUS.17,18 In our case, earlier diagnosis may have enabled the use of the gold standard surgical resection in tandem with adjuvant targeted therapies that have been shown to increase survival. 9
During his treatment, the patient suffered numerous infections despite efforts to incorporate colony stimulating factors into the chemotherapy regimen. Many of these complications involved the CBD, intrahepatic duct, and portal vein suggesting potentially increased tumor burden in the liver along with immunocompromised status. This aligns with previous studies citing the liver as a common site of metastasis for panNETs along with the lungs, bone, and peritoneum. 8
Conclusions
PanNETs are a rare subset of neoplasms which are often found at a late stage with diverse clinical presentations. Early imaging of suspicious lesions in addition to timely EUS-FNB with larger gauge size and increased passes for sampling can facilitate prompt diagnosis and initiation of targeted therapy to prolong survival time and quality of life.
Footnotes
Ethical considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent for publication
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article after death.
Author contributions
Conceptualization, S.D., A.S., A.G.; writing – original draft, S.D., A.S., A.G.; writing – review and editing, S.D., Z.A., A.S., A.G., P.S., K.D. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
