Abstract
A male patient underwent gastrojejunal bypass surgery in 2017. In 2020, he was referred to our hospital for suspected obstructive jaundice. Subsequently, he was diagnosed with cholangiocarcinoma, and endoscopic retrograde cholangiopancreatography was attempted via balloon-assisted enteroscopy. However, the endoscope did not reach the duodenal papilla owing to the abdomen–small intestine adhesion. Therefore, endoscopic ultrasound-guided hepaticogastrostomy was performed using a dedicated plastic stent. After stent placement, obstructive jaundice and cholangitis promptly improved. However, we replaced the plastic stent with a fully covered self-expandable metal stent because stent occlusions occurred frequently. Two months after fully covered self-expandable metal stent placement, the patient developed cholangitis again. Notably, during the endoscopic procedure, the stent was found to be completely migrated. Nevertheless, the fistula was still open, and the patient was successfully retreated with the maintained fistula of endoscopic ultrasound-guided hepaticogastrostomy.
Introduction
Endoscopic ultrasound-guided hepaticogastrostomy (EUS-HGS) is generally performed in cases where endoscopic retrograde cholangiopancreatography (ERCP) is unsuccessful or difficult. In particular, covered self-expandable metallic stents (SEMSs) are recommended owing to their long patency and ability to prevent bile leakage into the abdominal cavity. 1 Moreover, EUS-HGS using a fully covered SEMS (FC-SEMS) with a smaller diameter is reported to be useful for easy reintervention.2,3 However, FC-SEMSs do not have a bare distal end, rendering them at a potential risk of complete spontaneous migration into the gastrointestinal tract. Here we report a rare case of complete stent migration into the gastric side after EUS-HGS with FC-SEMS, which was successfully retreated using the maintained fistula.
Case
A male patient presented to our hospital with a complaint of jaundice. He had previously undergone gastrojejunal bypass for duodenal obstruction caused by a duodenal ulcer at the age of 73 years in 2017. His family history was unremarkable. On physical examination, he had yellowish conjunctiva, spontaneous pain in the cardiac region, and tenderness in the right costal region, with no signs of peritoneal irritation.
In March 2020, he complained of jaundice to his previous physician. The abdominal computed tomography (CT) scan revealed intrahepatic bile duct dilation. Hence, he was diagnosed with acute cholangitis and obstructive jaundice and referred to our hospital. During his first visit to our hospital, his blood test results revealed elevated hepatobiliary enzyme, direct bilirubin, and C-reactive protein levels. His abdominal contrast-enhanced CT scan revealed an infiltrative spread of poorly demarcated soft tissue shadows around the duodenum, pancreatic head, common bile duct, and cystic duct. Notably, his intrahepatic bile ducts were clearly dilated. Therefore, he was diagnosed with cholangiocarcinoma (Figure 1(a)).

(a) Abdominal computed tomography scan showing an infiltrative spread of poorly demarcated soft tissue shadows in the hilar region and around the common bile duct. The intrahepatic bile ducts are clearly dilated. (b) Bile duct stenosis can be seen from the hilar region to the middle part of the common bile duct, with no passage of the contrast medium to the duodenum. (c) The dedicated plastic stent is placed.
ERCP was attempted using a double-balloon endoscope (EC-450BI5; Fujifilm, Osaka, Japan). However, the endoscope could not reach the duodenal papilla because of strong adhesions near the Treitz ligament after insertion into the afferent limb. He had no symptoms of cancer other than cholangitis. The advantages and disadvantages of EUS-HGS and percutaneous transhepatic biliary drainage (PTBD) were explained to the patient, and he preferred internal biliary drainage via EUS-HGS. The intrahepatic bile ducts were visualized from the stomach using a linear array echoendoscope (GF-UCT240; Olympus Optical, Tokyo, Japan). As the segment 3 bile duct branch could not be visualized, we punctured the intrahepatic segment 2 bile duct branch with a 19-gauge fine aspiration needle (SonoTip Pro Control 19G; Medi-Globe GmbH, Achenmuhle, Germany). The cholangiogram revealed stenosis from the hilar region to the middle part of the common bile duct, and the contrast medium could not pass to the duodenum (Figure 1(b)). Subsequently, a 0.025-inch guidewire was inserted (VisiGlide2; Olympus Medical Systems, Tokyo, Japan) from the segment 2 bile duct branch across the right intrahepatic bile duct and a dedicated plastic stent was placed (Type IT, 7 Fr, 14 cm; Gadelius Medical K. K., Tokyo, Japan; Figure 1(c)). 4 Notably, the pigtail-shaped end of the stent on the gastric side has a strong anchoring effect; hence, theoretically, unlike SEMS, the proximal side of the plastic stent cannot migrate into the abdominal cavity. 5 Moreover, this stent can be easily removed and replaced. In the present case, before performing additional procedures, we planned to complete the fistula between the gastrointestinal tract and liver by placing the stent. However, the patient developed recurrent cholangitis shortly after EUS-HGS; thus, we placed an FC-SEMS (HANAROSTENT 6 mm × 12 cm, Boston Scientific Japan, Tokyo, Japan; Figure 2) 3 months after the initial EUS-HGS. Two months after the FC-SEMS placement, cholangitis recurred. Thus, 4 days after diagnosing cholangitis, the endoscopic procedure was performed again. The side-viewing endoscope (TJF-260V; Olympus Optical, Tokyo, Japan) inserted into the stomach revealed that the stent completely migrated to the gastric side (Figure 3(a)). However, we confirmed an EUS-HGS fistula opening on the lesser curvature of the gastric wall. Finally, the guidewire was successfully inserted into the bile duct through the fistula (Figure 3(b)), and the procedure was completed by placing an antegrade transpapillary stent (Flexima™ Plus, 8.5 Fr, 10 cm; Boston Scientific Japan, Tokyo, Japan) and a 7-Fr plastic stent into the fistula (Figure 3(c) and (d)). After the procedure, no adverse events occurred, and cholangitis improved; thus, the patient was discharged. Subsequently, no cholangitis occurred until the patient died 2 months later due to the primary disease.
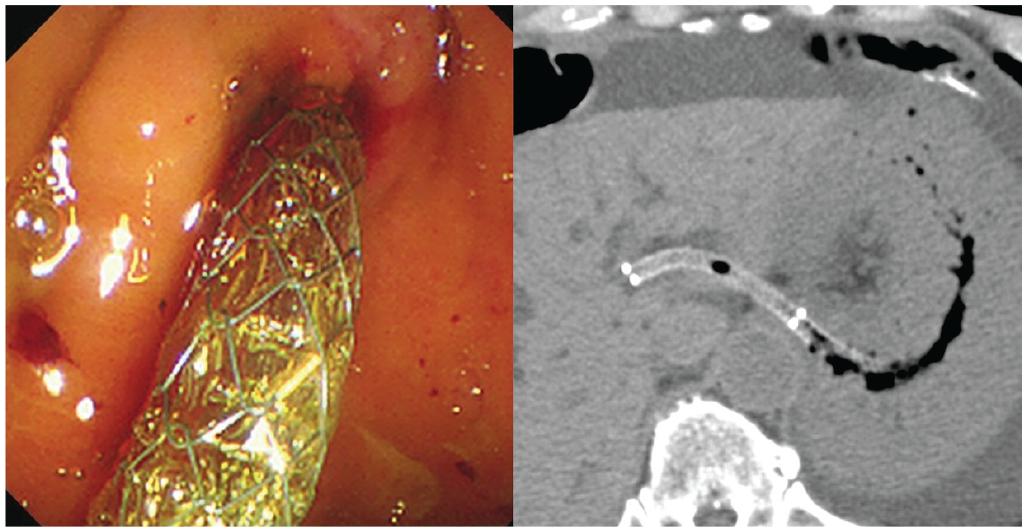
A fully covered self-expandable metallic stent is placed.

Endoscopic procedure performed for reintervention. (a) The stent is completely migrated. However, an EUS-HGS fistula can be seen on the lesser curvature of the stomach wall (
Discussion
Recently, ERCP using balloon-assisted enteroscopy (BAE) has been reported to be useful for pancreaticobiliary disorders in patients with surgically altered anatomy. 6 However, even when it is possible to reach the target site, the tools used for enteroscopy have limitations; hence, the desired procedure may not be completed. Notably, Giovanini et al. 7 were the first to report the EUS-guided choledochoduodenostomy (EUS-CDS) in 2001. Two years later, EUS-HGS was reported, 8 and recently, it has been considered a useful drainage method for patients in whom ERCP is difficult because of surgically altered anatomy, especially at high-volume centers. In particular, during EUS-HGS, once the fistula is completed between the gastrointestinal tract and liver, the procedure can be performed using a conventional side-viewing endoscope. In contrast to ERCP with BAE, almost all instruments used in ERCP can be inserted through the fistula site while performing EUS-HGS. Although EUS-HGS has been reported to have serious adverse events, such as peritonitis due to bile leakage, stent migration into the abdominal cavity, and bleeding,9–11 it provides benefits in terms of the quality of life of patients as it allows internal biliary drainage, unlike PTBD. Notably, EUS-HGS is best indicated for patients with malignant biliary stricture in whom ERCP is unsuccessful because of duodenal obstruction or surgically altered anatomy.
The stents used for EUS-HGS can be broadly classified into metal stents (SEMSs) and plastic stents. Although dedicated plastic stents are reported to be useful,4,5 SEMSs are recommended because they have longer stent patency and can prevent bile leakage into the abdominal cavity. 1 However, EUS-HGS using SEMS is associated with serious adverse events, including the migration of the proximal side of the stent into the abdominal cavity. Thus, the proximal side of SEMSs must be placed with a long protrusion into the gastric lumen to prevent stent migration into the abdominal cavity. 12 Partially covered SEMSs (PC-SEMS) with a bare end on the distal side are usually used in EUS-HGS to avoid obstruction of the side branch of the bile duct at the distal end of the stent. Given that the distal end of PC-SEMSs is embedded in the intrahepatic bile duct mucosa, stent removal can be difficult. Thus, reintervention after EUS-HGS with PC-SEMSs is often challenging.13,14
FC-SEMS with a smaller diameter has also been reported to be useful owing to the simplicity of its placement during EUS-HGS and ease of reintervention.2,3 However, it may obstruct the side branch of the intrahepatic bile duct, resulting in a liver abscess; hence, a relatively small diameter stent (6 mm) has been used in previous studies. 2 A fully covered braided type stent is easy to remove and replace and may be an ideal device to compensate for the disadvantages of conventional PC-SEMSs. However, this type of stent may completely migrate into the gastrointestinal tract because its bare distal end may not be anchored. Previous studies have reported the complete migration of FC-SEMSs, but they claimed that the frequency of migration was not high, and that salvage was easy. Additionally, in the study by Ogura et al., 15 no migration occurred after the implantation of an 8-mm-diameter FC-SEMS through sufficient hepatic parenchyma. However, the small diameter FC-SEMS had almost no anchoring effect, and even the best placement could not completely prevent migration. 2 We believe that complete stent migration is a serious adverse event in EUS-HGS with FC-SEMSs. In the present case, the endoscopic procedure was performed early after the onset of cholangitis; thus, the fistula was still open, allowing the stent to be successfully replaced. If the reintervention was delayed, the fistula could have completely closed.
Conclusion
Compared with PC-SEMS, FC-SEMS can be easily removed and replaced. However, the patient must be closely monitored, and if cholangitis occurs, reintervention must be provided early, considering the possibility of a complete stent migration. Delay in detecting complete migration may make the reintervention impossible.
Footnotes
Author contributions
F.T. and K.K. contributed to conceptualization; F.T., K.K., Y.F., S.A., N.N., T.O., Y.M., and M.T. contributed to formal analysis and investigation; F.T. contributed to writing – original draft; K.K. and A.M. contributed to writing – review & editing; H.Y. contributed to supervision.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Research ethics and patient consent
All procedures were conducted in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and the Helsinki Declaration of 1975, as revised in 2008. This case report does not involve any experimental research on animals or human patients; thus, ethical approval was not necessary. Written informed consent was obtained from a legally authorized representative for publishing anonymized patient information in this article.
