Abstract
Scleritis results from an inflammatory response in the sclera and can be associated with a variety of infectious and non-infectious ocular and systemic diseases. It can cause vision loss, uveitis, high intraocular pressure and other complications, resulting in poor prognosis. There are no reports of non-infectious scleritis in children. Herein, we describe a case of sequential tocilizumab treatment on tofacitinib in a child with non-infectious scleritis. Extensive examinations ruled out the cause of infection, and the previous conventional glucocorticoid treatment was effective, but the disease recurred during drug reduction. Tocilizumab and tofacitinib successfully cured the patient, glucocorticoids were stopped, and all drugs were stopped 2 years later. This report provides a new diagnosis and treatment method for clinicians.
Introduction
Scleritis, 1 caused by an inflammatory response of the sclera, is associated with various infectious and non-infectious ocular and systemic diseases. Non-steroidal anti-inflammatory drugs, corticosteroids, and immunosuppressive drugs are commonly prescribed for non-infectious scleritis. Recently, there have been reports regarding the efficacy of tumour necrosis factor-α inhibitors or anti-CD20 agents in the treatment of adult scleritis. 2 However, to the best of our knowledge, the treatment of non-infectious scleritis in children has not been reported. Herein, we present a novel therapeutic approach for a 13-year-old boy with non-infectious scleritis.
Case
A 13-year-old boy (height = 162 cm; weight = 60 kg) initially presented to our hospital in February 2021 with a pain in his right eye following an upper respiratory tract infection (URTI). At infancy, he received Bacillus Calmette–Guérin; hepatitis B; trivalent oral polio; whole-cell diphtheria–pertussis–tetanus; measles; diphtheria and tetanus toxoids; Japanese encephalitis; group A meningococcal; groups A and C meningococcal; measles, mumps and rubella; and hepatitis A vaccines. Neither his classmates nor family members experienced a similar situation. He had been referred to Xijing Hospital, Beijing Tongren Hospital professional ophthalmology 1 month prior, but unfortunately, some specific laboratory findings are unknown. He received steroids, non-steroidal anti-inflammatory drugs and tobramycin, which altered the course of the disease. However, because of a recurrent URTI, the patient presented to our hospital with complaints of severe ocular pain and tenderness, redness, oedema, photophobia and blurred binocular vision. Eye examination at a provincial eye hospital before admission revealed pressures of 16 and 15 mmHg in the right and left eyes, respectively. The right eye demonstrated a clear cornea with 2+ flare in the anterior chamber, while the left eye exhibited conjunctival and episcleral vascular congestion, a clear cornea, and 3+ flare in the anterior chamber. Fundus B-scan ultrasound findings for both eyes are as shown in Figure 1.

Ocular photographs of the patient at the beginning of the inpatient treatment regimen in February 2021 (a, right; b, left). Both eyes presented optic discs with well-defined margins, pale colouration and oedema, more pronounced in the left eye.
He was diagnosed with non-infectious scleritis, after excluding streptococci, tuberculosis, respiratory pathogens and Epstein–Barr virus infections, and was recommended consultation with the rheumatology and immunology departments. In our department, he underwent blood investigations for rheumatoid factor, antinuclear antibody, antineutrophilic cytoplasmic antibody, anti-citrullinated protein antibody, human leukocyte antigen B27 levels, treponema pallidum haemagglutination, hepatitis virus, human immunodeficiency virus and mycoplasma antibodies, and tuberculosis; all of which were negative. However, his C-reactive protein level was 23.1 mg/L and erythrocyte sedimentation rate was 72 mm/h. With the cooperation of ophthalmologists, the patient was started on topical steroids and systemic steroid therapy (80 mg methylprednisolone daily reduced by 40 mg prednisolone after a week) and azathioprine tablets (2 mg/kg daily), which showed beneficial effects. Visual acuity improved significantly, and he had no adverse and unanticipated events after a week. The steroid’s dose gradually decreased out of hospital. However, prednisone could not be tapered below 15 mg daily without a disease flare as of October 2021. Upon tapering of prednisone to 5 mg daily, both eyes exhibited disease recurrence accompanied by ocular manifestations including congestion, photophobia, tearing and/or decreased vision. Fundoscopic examination revealed the following characteristic findings: (1) the vitreous cavity demonstrated punctate echoes; (2) a curvilinear band-like echo originated from the optic disc; and (3) Posterior ocular wall echoes showed diffuse thickening with sparse reflectivity, culminating in the pathognomonic “T-sign” (Figure 2). Details regarding the macular fovea are shown in Figure 3.
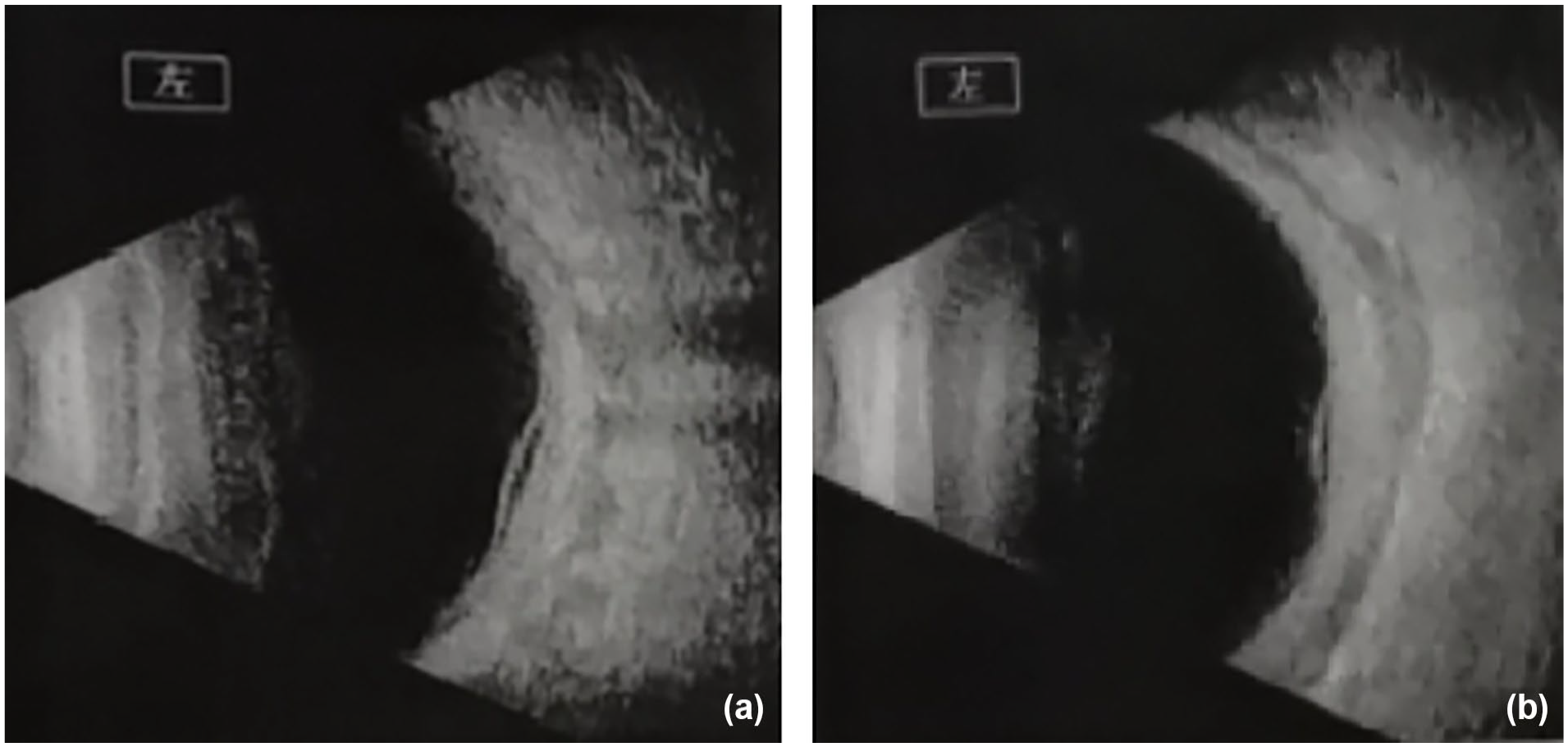
Ocular ultrasonography demonstrated the following characteristic findings (a, right; b, left): (1) diffused punctate echoes within the vitreous cavity bilaterally; (2) abnormal thickening of the posterior ocular wall with heterogeneous reflectivity; and (3) irregular hypoechoic zones in the retrobulbar space, particularly marked in the left eye.

Optical coherencetomography as of October 2021 (a, right; b, left). The macular fovea of the right eye was 170 µm, with no obvious abnormality. While the left eye macular fovea was 666 µm, with widespread neuroepithelial detachment in the macular area, granular layer reflection (exudation) and retinal detachment (The “Green lines” indicates the direction of the B-scan corresponding to the fundus scanning line in the OCT images).
IL-22, 3 IL-6, and IL-2 4 have been highly expressed in the blood of patients with non-infectious scleritis. Tocilizumab, an IL-6 receptor antagonist, is used to treat systemic juvenile idiopathic arthritis, but has not been reported to treat scleritis. However, in China, only the intravenous form is available, and it is expensive. After obtaining parental consent, we administered tocilizumab following the drug instructions (8 mg/kg for body weight ⩾30 kg). For this patient, Tocilizumab 480 mg twice weekly was then implemented with efficacious results (when prednisone was reduced to 2.5 mg/day, there was no recurrence). The patient reported remarkable improvement in his symptoms, and the scleral inflammation completely resolved. Figure 4 shows the macular fovea improvements.

Ocular photographs following treatment with tocilizumab and tofacitinib as of January 2022 (a, right; b, left). The macular fovea of the right eye reduced to 145 µm, and that of the left eye to 150 µm, with no obvious abnormalities (The “Green lines” indicates the direction of the B-scan corresponding to the fundus scanning line in the OCT images).
After the fourth tocilizumab infusion in January 2022, the patient presented with no signs of inflammation. Despite the positive efficacy and lack of adverse reactions of tocilizumab, the family complained of the cost and intravenous route of administration, expressing the desire for a more economical, convenient and effective drug for maintenance treatment. Tofacitinib, a first-generation Janus kinase (JAK) inhibitor, primarily blocks the JAK1/JAK3 pathway, thereby inhibiting cytokine release, including IL-6,5,6 was considered due to its oral route of administration and low cost. Subsequently, the patient was started on tofacitinib tablets (5 mg twice daily) based on the available pharmacokinetics data. 7 Steroid therapy was reduced to 2.5 mg and discontinued in February 2022. The patient followed the doctor’s prescription, and there was no recurrence. Tofacitinib was safely discontinued in July 2023. Presently, the patient has no discomfort in both eyes, normal vision, normal development and his height has increased by 13 cm in ~2 years (Figure 5).

Timeline of this case report.
Discussion
Anti-TNF drugs have been effective in the treatment of ocular inflammatory diseases such as non-infectious scleritis,8,9 but there are no reports on their use in paediatric scleritis. Some patients do not tolerate or respond to these treatment regimens, putting them at risk of persisting morbidity and losing vision. Tocilizumab and tofacitinib work through different pathways and have different usage methods and prices.
In a previous report of tocilizumab treatment in a 60-year-old female patient with necrotising scleritis, neither local steroid therapy, systemic steroid therapy (40 mg methylprednisolone daily, reduced by 10 mg prednisolone on a weekly basis), nor anti-TNF-α (infliximab) therapy altered the disease’s course. However, the combination of anti-IL-6 therapy (tocilizumab, 480 mg monthly) and a steroid bolus (prednisolone, 500 mg for 3 days and 250 mg for 2 days followed up by a slow reduction) proved to be effective. After the patient’s sixth tocilizumab infusion, she presented with no inflammatory signs at follow-up. Steroid therapy was reduced and subsequently discontinued after 3 years without a disease flare. However, they could not differentiate whether tocilizumab alone, steroid bolus therapy or a combination of both was the driving mechanisms mediating the cure in this case report. 10 Additionally, a previous case series described 10 cases of scleritis that were treated with tocilizumab for 9 months, 11 in which successful control of inflammation via retaining steroids (corticosteroid-sparing success was defined as inactive inflammation after tapering prednisone to ⩽5 mg daily) was achieved in 50% of the patients. This case series confirmed that IL-6 antagonist, tocilizumab, is effective in treating scleritis. However, in that report, tocilizumab was discontinued in four patients (24%) because of serious side effects.
The present case was that of a child diagnosed with scleritis. After four doses of tocilizumab treatment (480 mg), the patient’s scleral inflammation improved. His macular oedema and retinal detachment disappeared, and his vision was substantially recovered without any adverse reactions, suggesting that tocilizumab is effective in the treatment of non-infectious scleritis (Figure 4).
At his parents’ request, considering the method of administration and price, we switched to oral tofacitinib. A previous case report profiled a 40-year-old woman who was diagnosed with scleritis by a local ophthalmologist. 12 Steroid-sparing therapy with methotrexate or mycophenolate as a monotherapy regimen was ineffective. Tofacitinib was added to methotrexate following which her scleritis resolved within 1 week. Over the subsequent 9 months, steroid-sparing combination therapy with tofacitinib and methotrexate-maintained disease remission. A similar result was reported in a 65-year-old man presenting with redness and pain associated with active necrotising scleritis in the left eye. 13 He was started on mycophenolate mofetil and oral corticosteroids. However, the disease course remained unaltered after 4 weeks, following which he was treated with tofacitinib, causing a dramatic improvement in symptoms. Researchers have speculated that tofacitinib can more effectively cross the blood–water or blood–retinal barrier compared with other pharmaceutical compounds and may thus be superior to other biological agents used to treat scleritis.
Tocilizumab or tofacitinib has been approved for the treatment of some autoimmune diseases.
To the best of our knowledge, this is the first report of the use of tocilizumab followed by tofacitinib to successfully treat non-infectious scleritis in a child. Given the efficacy of this regimen in this case report, we hope it can be applied to non-infectious scleritis and autoimmune diseases. Future research is necessary to evaluate more clinical evidence to reach a definitive conclusion and more comprehensively inform medical guidelines.
Footnotes
Acknowledgements
We would like to thank the patient for their consent to use their data for publication.
Ethical considerations
The Ethics Committee of The second Clinical Medical College of Shanxi Medical University waived the need for formal approval in this case. This study was performed in accordance with the Helsinki Declaration of 1964 and its later amendments.
Consent to participate
The guardian of the patient has verbally agreed and actively cooperated with the conception of this report.
Consent for publication
The guardian of the patient has provided consent to publish this report.
Author contributions
Xiaoxiang Xie acquired the data and finished the article and was a major contributor in writing the manuscript. Pan Jiang and Xuxu Li performed the statistical analysis and participated in its design. Jinli Ru participated in acquisition, analysis or interpretation of data and made critical revision of the manuscript. All authors read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data used for this study, though not available in a public repository, will be made available to other researchers upon reasonable request.
