Abstract
Acute hemorrhagic leukoencephalitis (AHLE) is a rare neurologic inflammatory disorder characterized by severe and rapidly progressive encephalopathy; typically considered a severe variant of acute demyelinating encephalomyelitis (ADEM). There is limited pediatric data on treatment modalities for refractory illness. We report here the case of an 8-year-old girl who presented with acute concern for encephalopathy, ataxia, and seizures in the context of preceding infectious symptoms and neuroimaging findings highly suspicious for ADEM. Treatment was promptly initiated with high-dose glucocorticoids followed by addition of intravenous immunoglobulin therapy. Despite ongoing treatment and permissive hypernatremia and hyperosmolar therapy to treat signs of increased intracranial pressure, she showed no clinical signs of improvement, so plasmapheresis was initiated. Follow-up magnetic resonance imaging (MRI) brain showed progression of lesions with more hemorrhagic conversion concerning for AHLE, so anakinra was started. With the combination of pheresis and anakinra, the patient began to show signs of neurologic recovery. Her course was complicated by delirium and physical deconditioning, requiring inpatient rehab, although her neurologic function showed daily improvement. Final MRI brain to date reflected these improvements. Our case highlights the importance of rapid escalation of care to improve mortality rates and reduce neurologic sequelae of AHLE. Future studies are needed to address the safety profile and efficacy of anakinra in refractory disease.
Keywords
Introduction
Acute hemorrhagic leukoencephalitis (AHLE) is a rare, severe neurologic inflammatory disorder characterized by rapidly progressive encephalopathy, symmetrical multifocal demyelinating brain lesions, and associated necrosis, hemorrhage, and edema on neuroimaging. 1 Typically considered a severe variant of acute demyelinating encephalomyelitis (ADEM), AHLE carries a high mortality rate of around 70%.2,3 Given the rarity of AHLE, particularly in the pediatric population, treatment protocols remain limited, with sparse data regarding effective therapies for refractory cases. 4 Existing therapies primarily focus on immunosuppression and inflammation control typically involving the use of systemic corticosteroids, intravenous immunoglobulin, and/or plasmapheresis. 5 However, refractory cases pose challenges to this initial approach, underscoring the need for innovative treatments. Herein, we report a pediatric case of AHLE with a favorable outcome following aggressive medical management, including the novel use of biologic therapy (anakinra) after standard care failed to result in any meaningful clinical recovery.
Case description
An 8-year-old girl (JA) with a history of obesity and mild hyperlipidemia presented to our Pediatric Intensive Care Unit (PICU) with acute encephalopathy, ataxia, and seizure-like activity. Symptoms were preceded by 1 week of fever, upper respiratory symptoms, diarrhea, and emesis, as well as a month-long history of intermittent headaches. Notably, domicile family members had similar antecedent infectious symptoms, and the mother endorsed a history of intermittent cold sores with recent recurrence. There was no history of trauma, travel, zoonotic exposures, or new medications. A noncontrast computed tomography (CT) scan of the head revealed multifocal lesions with diffuse cerebral edema.
Shortly after admission, the patient developed anisocoria, right upper and lower extremity clonic seizure, and a decline in the Glasgow Coma Scale (from 15 to 13). She was urgently intubated, and treatment with levetiracetam, lorazepam, and a 3% NaCl bolus was initiated. Pediatric Neurology, Neurosurgery, and Infectious Disease services were consulted. Continuous electroencephalogram monitoring was started, and standard neuroprotective measures were implemented, including elevation of the head of the bed, maintenance of normonatremia, normocarbia, normothermia, and normoglycemia. Empiric antimicrobial therapy was started with meningitic dosing of ceftriaxone (100 mg/kg/day), vancomycin (60 mg/kg/day), acyclovir (30 mg/kg/day), and doxycycline (100 mg twice daily).
Magnetic resonance imaging (MRI) of the brain showed multifocal, bilateral, asymmetric T2, and fluid-attenuated inversion recovery hyperintense lesions involving the frontal, parietal, and temporal lobes, with mass effect and contrast enhancement (Figure 1(a)). These findings were concerning for ADEM, though the differential diagnoses also included infectious etiology and immune-mediated vasculitides. An area of punctate susceptibility raised the suspicion for AHLE due to hemorrhagic changes (Figure 3(a)).
High-dose methylprednisolone (1000 mg/day) was initiated for empiric ADEM treatment. However, there was no clinical improvement, and intravenous immunoglobulin (IVIG) (1 g/kg/day) was started on day 3. The patient, however, began exhibiting signs of increased intracranial pressure (ICP), including bradycardia, hypertension, anisocoria, and fluctuating consciousness, prompting the initiation of permissive hypernatremia (150–160 mmol/L) and hyperosmolar therapy (targeting serum osmolality of 310–320 mOsm/kg). Repeat head imaging in the context of this symptomatology was largely unchanged without signs of impending cerebral herniation.
Despite these aggressive management strategies, her clinical status remained unchanged. Apheresis was initiated on day 4, and IVIG was discontinued after two rounds (2 g/kg). Due to concerns regarding ICP, lumbar puncture (LP) was deferred. To garner additional data regarding the progression of illness and possibly inform prognosis, a repeat MRI brain was done on day 5 of admission, which revealed interval worsening diffuse edema and hemorrhage, and new and enlarging areas of diffusion restriction (Figures 1(b), 2, and 3(b)).
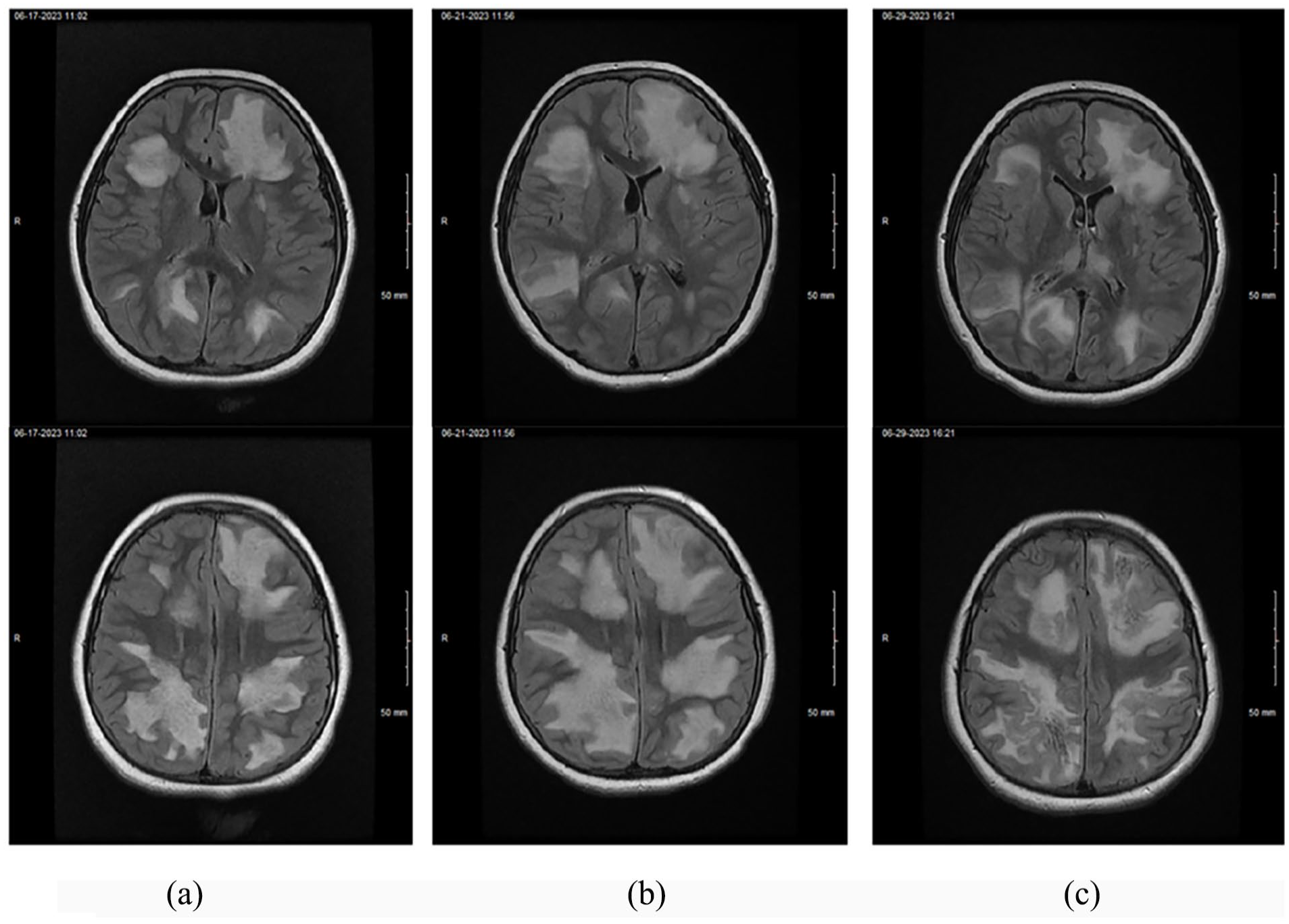
MRI brain with and without contrast: axial T2 fluid attenuated inversion recovery. (a) Day 1 of admission. (b) Day 5 of admission. (c) Day 12 of admission.

MRI brain: axial DWI with corresponding ADC. (a) DWI. (b) ADC.

MRI brain: axial susceptibility-weighted angiography. (a) Day 1. (b) Day 5. (c) Day 12.
Following a multidisciplinary discussion, anakinra (100 mg daily), an interleukin-1 (IL-1) inhibitor, was introduced. Ongoing anakinra therapy, combined with three more plasmapheresis sessions, led to gradual neurologic recovery, including improved wakefulness, resolution of anisocoria, and the ability to follow commands. The patient was also able to tolerate liberalization of serum sodium and osmolality goals previously described. Sedation was weaned, and the patient was successfully extubated after 14 days.
The patient underwent LP postextubation, revealing elevated opening pressure (>50 cmH2O) but normal cerebrospinal fluid analysis (Table 1). An urgent follow-up CT scan showed no signs of cerebral herniation, confirming the absence of clinical deterioration despite the elevated opening pressure.
Results of diagnostic assessment.

CSF: cerebrospinal fluid; LDH: lactate dehydrogenase; ACE: angiotensin converting enzyme; PCR: polymerase chain reaction; MOG: myelin oligodendrocyte glycoprotein; NMO: neuromyselitis optica; JCV: JC/John Cunningham virus; EBV: Epstein–Barr virus.
The remainder of her PICU course focused on rehabilitation, including physical, occupational, and speech therapies, and delirium management. The patient spent 4 weeks in inpatient rehab before being discharged home. Throughout her hospital stay, the patient completed five sessions of plasmapheresis, a 5-day course of pulse steroids (with subsequent taper), and 4 weeks of daily anakinra with a gradual taper over the following 2 weeks.
Long-term follow-up continues, with ongoing monitoring for potential neurologic deficits. Recent MRI results showed significant improvement, with reduced lesion size, mass effect, and diffusion restriction, although small parenchymal hemorrhages persist (Figures 1(c) and 3(c)). Happy to share that her neurologic assessments to date report an overall reassuring exam, with an alert girl answering questions appropriately for age with only slightly weaker left upper and lower extremities on strength testing.
Discussion
AHLE is an acute, rare, and often fatal inflammatory myelinopathy believed to occur in the postinfectious state where there is cross-reactivity between antigens targeting infectious agents and central nervous system tissues.2,6 Although more common in young adults, AHLE has been described in children, with 24 documented cases in the literature.2,4,7,8,9 Given the rare nature of AHLE, pediatric treatment protocols remain underdeveloped. Furthermore, the etiology of AHLE remains unknown, though the usual infectious prodrome suggests an autoimmune entity as noted by Alromaihi 2 and Atherton et al. 6
In accordance with this hypothesis, first-line therapy is glucocorticoids—typically methylprednisolone; with IVIG and plasma exchange/plasmapheresis being reserved as typical second-line agents to help dampen this dysregulated immune response. 5 Outside of these “conventional” therapies, strong recommendations for refractory disease remain unclear. 4 In this case, the patient showed no signs of convincing improvement while on high-dose steroids and IVIG, so plasma exchange was initiated. However, even with this modality, albeit only two cycles, neuroimaging results showed worsening lesions and the patient continued to be profoundly encephalopathic with continued signs of increased ICP. Thus, more esoteric options were explored.
Recent literature highlights the potential role of biologic agents, such as tocilizumab and rituximab, in refractory cerebral autoinflammatory conditions,5,10,11 although the informative value of these adult studies is limited by the small sample size and potential lack of external validity in a pediatric context. Moreover, rituximab (CD-20 monoclonoal antibody) and tocilizumab (anti-interleukin-6 antibody) target the adaptive arm of the immune system (B-cell/T-cell functioning) 11 ; however, there is growing evidence to suggest that dysregulation of the innate immune system accounts for the refractory nature of autoinflammatory diseases. 10 The main effectors of the innate immune response are myeloid cells, like neutrophils and monocytes, and pro-inflammatory cytokines, such as IL-1 and tumor necrosis factor-α (TNF-α). 12
Anakinra, a recombinant human IL-1 receptor antagonist, has shown promise in treating inflammatory diseases by targeting disordered innate immune responses, including those involving monocytes and macrophages. 10 Specifically, IL-1 is a crucial pro-inflammatory cytokine responsible for inducing the further release of other pro-inflammatory cytokines (TNF-α and IL-6) and chemokines; disrupting the blood brain barrier; activating glial cells and releasing neurotoxic substances. 13 Studies on the efficacy of anakinra in neuroinflammatory diseases are still limited, with no documented use in pediatrics, but its success in treating conditions like multisystem inflammatory syndrome of childhood (MIS-C) and chronic rheumatologic disorders has led to increased confidence in its use in pediatric neuroinflammatory disorders.
There are several limitations the authors recognize. First, the simultaneous use of plasmapheresis complicates the attribution of clinical improvement solely to anakinra. Additionally, it is acknowledged that plasmapheresis could theoretically interfere with plasma levels of anakinra, but there are no definitive results to fully elucidate this phenomenon. Nonetheless, to curb this possible confounder, on days plasmapheresis was conducted, anakinra was given after completion of the session. It is also important to discuss the potential diagnostic limitations posed by the timing of CSF analysis. In fact, there were multiple discussions with neurology about the ideal timing of LP—outside of the concerns for raised ICP—given the known potential for IVIG and pheresis to interfere with results. Interestingly though, while the negative NMO and MOG results in the CSF could be challenged, the corresponding serum results, which were obtained prior to IVIG and pheresis, were negative. Finally, the timing of “prognostic” MRI (day 5) may have been premature to assess radiological recovery after only two cycles of plasmapheresis. However, we also know that clinical improvement, particularly in neuroinflammatory conditions, often outpaces radiological changes.10,14 Therefore, while one can argue about the timeline of imaging, it was more so the worsening of the patient’s status, which prompted the multidisciplinary team to proceed with anakinra therapy given lack of response to conventional therapy.
In our case, we postulate that early escalation of therapy, including anakinra, likely contributed to the favorable outcome. Further studies are needed to clarify the role of anakinra in treating AHLE, particularly in pediatric populations, and to explore its safety profile when used in conjunction with other immunotherapies. There is also some educational value for adult cases to extrapolate from this case as anakinra use even in adult AHLE cases are sporadic.
Conclusion
AHLE is a rare, life-threatening neurologic disease with rapidly progressive cerebral lesions and encephalopathy, falling within the spectrum of ADEM. The small number of published cases calls for a high level of suspicion when patients develop signs of cerebral inflammation without a clear etiology. Our case emphasizes the importance of early diagnosis and rapid escalation of care in improving outcomes and reducing long-term neurologic sequelae. Additionally, it underscores the potential therapeutic benefits of immune modulation with anakinra in refractory AHLE, though further research is required to assess its safety and efficacy. We thus hope this case contributes to the growing body of literature on alternative treatment strategies in AHLE and similar neuroinflammatory diseases. To our knowledge, this is the first documented case of anakinra use in pediatric AHLE.
Footnotes
Ethical considerations
Our institution does not require ethical approval for reporting individual cases or case series.
Consent to participate
Written informed consent was obtained from a legally authorized representative (mother) for anonymized patient information to be published in this article.
Author contributions
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
