Abstract
We recently performed the mini-loop technique on a 69-year-old woman with idiopathic isolated tricuspid valve prolapse. Although the tricuspid valve leaflets showed no abnormalities, we noted a severely enlarged annulus and posterior leaflet prolapse due to rupture of the posterior papillary muscle. The procedure included four pairs of artificial chordal reconstruction using the loop technique with GORE-TEX® suture CV-4 and the insertion of the 28-mm CE Physio Tricuspid ring. Postoperatively, tricuspid regurgitation was well-controlled below trivial levels. Although the loop technique is typically used in mitral valve plasty, it may also be beneficial for tricuspid valve plasty.
Introduction
The loop technique using expanded polytetrafluoroethylene (ePTFE) is commonly employed for mitral annuloplasty, 1 has been reported as a useful procedure with good long-term results. 2 Although ePTFE is proven to be effective for the reconstruction of artificial neochordae in tricuspid valve plasty,3–5 there have been few reports on the use of the loop technique for this purpose.
Case
The patient was a 69-year-old woman who started experiencing occasional heart palpitations at 33 years of age. At 67 years of age, a heart murmur was detected during a physical examination, while an echocardiogram indicated severe tricuspid regurgitation (TR); however, she had no symptoms of heart failure and underwent follow-up every 6 months with echocardiography. At the age of 69, her heart palpitations became frequent, and an electrocardiogram showed paroxysmal atrial fibrillation. She had no symptoms during sinus rhythm, but the right ventricular diameter was enlarging, prompting a referral for surgery. A holosystolic murmur (Levine II/VI) was noted, which was strongest at the left margin of the fourth intercostal sternum; however, there was no evidence of the Rivero-Carvallo sign,6,7 and hepatosplenomegaly was not palpable. Right heart catheterization showed no pulmonary hypertension, and transthoracic echocardiography revealed an enlarged right ventricular diastolic diameter of 49 mm; however, the right ventricular function was within normal limits with a right ventricular fractional area change of 0.46. The diameter of the tricuspid valve annulus was 50 × 49 mm, and severe TR due to posterior tricuspid valve leaflet prolapse was observed on transesophageal echocardiography (Figure 1).

The tricuspid valve annulus diameter was 50 × 49 mm, and a severe TR due to posterior tricuspid valve leaflet prolapse was observed using transesophageal echocardiography.
Intraoperative findings revealed a normal tricuspid valve, but a severely enlarged annulus and posterior leaflet prolapse due to rupture of the posterior papillary muscle (Figure 2(a)). The stump of the ruptured posterior papillary muscle appeared scar-like (Figure 2(b)). At the beginning of the operation, a single GORE-TEX® suture CV-4 (W. L. Gore & Associates, Inc.; Flagstaff, Ariz) was used to make a single 8-mm mini-loop, and both ends of the needle were threaded through the pledget. Suture needles were passed through the tip of the posterior papillary muscle and tied over a second pledget. Subsequently, they were passed through the anteromedial portion of the chordae tendineae attachment of the posterior leaflet, and a tourniquet was applied. Free loops were fixed to the prolapsing leaflet segment using an additional CV-4. One CV-4 was added to one of the chordae tendineae attachment sites on the posterior leaflet near the septal leaflet, and a tourniquet was applied. Thereafter, a 28-mm CE Physio Tricuspid ring (Edwards Lifesciences) was inserted. A water test was performed, and the tourniquet was tightened. The optimal position to control the reverse flow was identified, and the CV-4 was ligated. Repeated water tests were conducted, and three additional CV-4 were used to reconstruct all the identified chordae tendineae attachment sites (Figure 2(c) and (d)). In addition, pulmonary vein isolation and left atrial appendage resection were performed. Intraoperative transesophageal echocardiography showed no TR. One year postoperatively, transthoracic echocardiography showed trivial TR and the right ventricular diastolic diameter had improved to 30 mm. The patient was symptom-free and well.
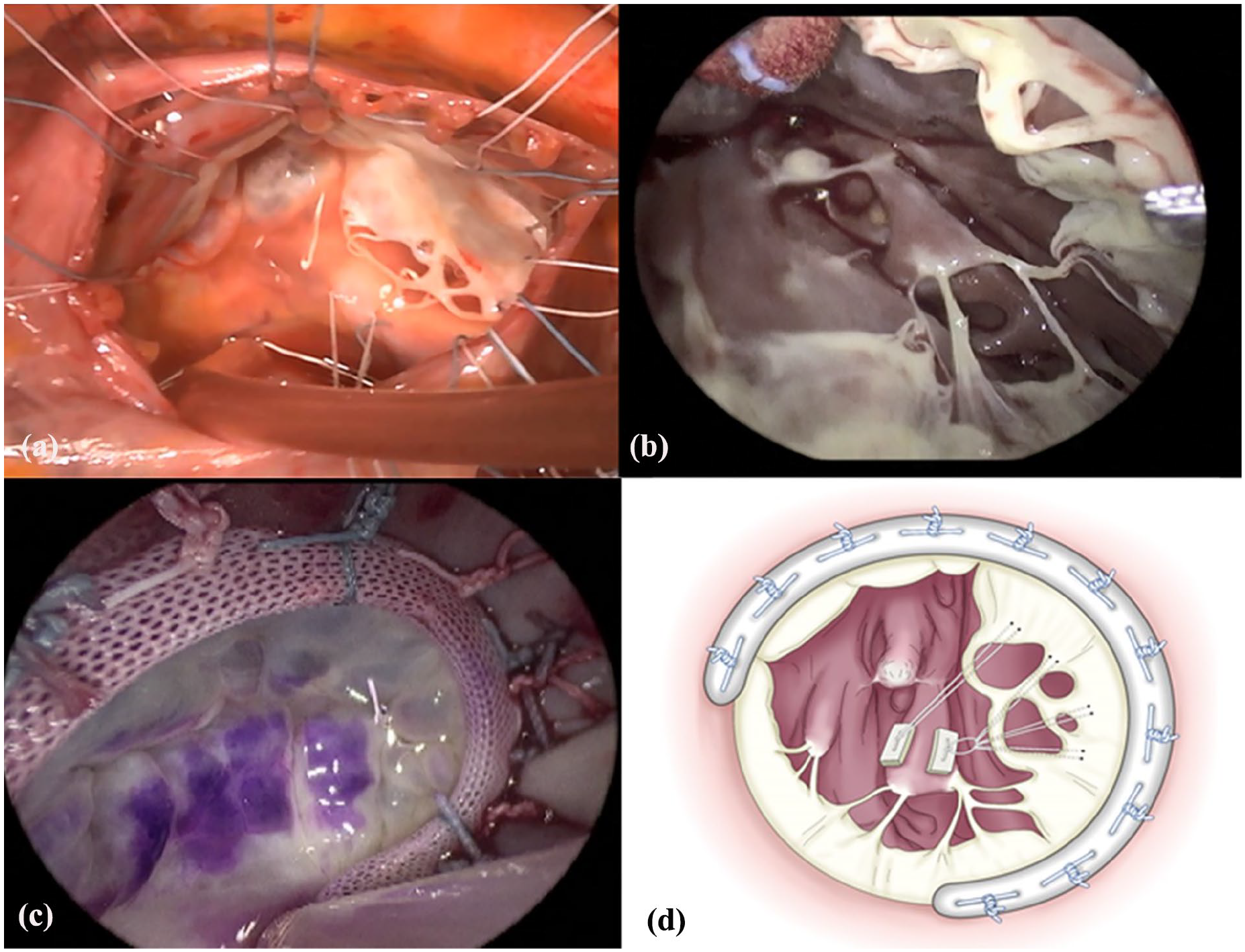
Intraoperative findings. (a) The tricuspid valve was normal, and a severely enlarged annulus and posterior leaflet prolapse due to rupture of the posterior papillary muscle. (b) The stump of the ruptured posterior papillary muscle was scar-like. (c) The procedure included four pairs of artificial chordal reconstruction using loop technique with GORE-TEX® suture CV-4 and the insertion of the 28-mm CE Physio Tricupid ring. (d) Artificial neochordae were fabricated at four locations.
Discussion
The prevalence of tricuspid valve prolapse is estimated to be about 0.1 to 5.5%, 8 however most cases are associated with mitral valve prolapse. Common causes of primary tricuspid prolapse include Ebstein's malformation, traumatic, infectious, post-radiotherapy, drug use, and myxomatous degeneration, while idiopathic isolated tricuspid valve prolapse is rare.
Histopathology was not performed to determine the cause of papillary muscle rupture in this case; however, surgical findings showed no myxomatous degeneration of the tricuspid valve. The patient had no history of trauma, and although she experienced palpitations during childbirth at 33 years of age, there was no evidence linking them to papillary muscle rupture. Therefore, idiopathic isolated tricuspid valve prolapse was suspected.
In this case, the patient underwent valvuloplasty with artificial neochordae reconstruction using a mini-loop technique with ePTFE and annuloplasty. ePTFE has been shown to be effective for the reconstruction of artificial neochordae in tricuspid valve plasty,3–5,9–12 with early- and mid-term results reporting a 30-day reoperation rate of 5.2% and a cumulative risk of reoperation of 7.2% at 3 years. 5 However, few studies have employed the loop technique for this purpose. 13 During the procedure, we did not observe any degeneration of the papillary muscle tissue, except for those in the regions affected by chordae tendineae rupture. The leaflets and remaining papillary muscle tissue were sufficiently robust. Therefore, we determined the loop technique would allow for an anatomically natural reconstruction. Consequently, a single CV-4 was used to make an 8-mm mini-loop, and both ends of the needle were threaded through a pledget. Free loops were fixed to the prolapsing leaflet segment using an additional CV-4. Finally, artificial neochordae were fabricated at four locations.
Conclusion
The mini-loop technique is a feasible and effective approach for artificial neochordae reconstruction in tricuspid valvular plasty. Further studies are required to evaluate the long-term outcomes and broader applicability of this method in patients with tricuspid valve plasty.
Footnotes
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing. We also thank Y. Sato, SAIKOU, Inc. (![]() ) for the assistance provided in figure illustration.
) for the assistance provided in figure illustration.
Author contributions
Y.M. collected the clinical data and drafted the manuscript. S.I. supervised the writing of case report as the corresponding author and critically reviewed the manuscript. Both authors approved the final version of the manuscript and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
