Abstract
A 67-year-old male with positive signals for neuroendocrine and head/neck cancer on a commercially available multicancer detection test underwent an extensive negative diagnostic workup at a local hospital. Referred to our medical center for advanced imaging and endoscopic procedures ultimately led to the diagnosis of an early-stage seminoma with eventual orchiectomy. This case highlights challenges in evaluating positive multicancer detection results, emphasizing the need for a broader understanding of how to interpret these testing results to fully evaluate complex cases, as well as the need for protocolized follow-up approaches to a positive multicancer detection test.
Background
Cancer is the second leading cause of death in the United States, with over 600,000 deaths in 2023. 1 Since 1991, the overall cancer mortality rate has declined, averting more than 4 million deaths due to reduced smoking, earlier detection, and improved treatments. However, incidence rates for major cancers—such as breast, prostate, uterine, pancreatic, and liver—are rising, with the latter two having no reliable screening test.1–3 Promising new methods have been introduced to expand cancer screening efforts.
A blood-based multicancer detection (MCD) test (Galleri Multi-Cancer test, GRAIL Menlo Park, CA) that measures circulating methylated DNA changes in cancer cells has been available for the past few years. 4 The recently commercialized MCD test evaluates 50 types of cancer, identifying the cancer signal of origin (CSO), and has great potential to improve early detection of cancers that lack reliable screening tests (e.g., pancreatic, liver, and ovarian). 4 There is high demand for a biomarker test that addresses screening gaps for these cancers. Despite its potential, numerous challenges exist in the widespread implementation of MCD testing in clinical practice, such as understanding the benefits and risks of these tests, obtaining a reliable diagnostic algorithm to evaluate positive signals, and determining the appropriate management of false-positive tests. 5 In the present report, we describe a patient with positive MCD and negative cancer workup locally for the CSO. However, the MCD test prompted a thorough evaluation leading to the revelation of a previously undiagnosed cancer in the patient.
Case report
A 67-year-old male had MCD testing as recommended by his local primary care physician in a rural town in December 2022. A positive signal for a neuroendocrine tumor (NET) with a secondary positive signal for head and neck cancer was reported. At that time, he completed an evaluation with a CT Chest, abdomen, and pelvis which was unrevealing. He also had a brain MRI and laryngoscopy for the head and neck signals which were negative. In addition, laboratory testing for 5-HIAA and metanephrines was negative.
In March 2023, he had repeat MCD testing which again demonstrated a positive neuroendocrine signal. This time, there was no secondary head and neck signal, but rather a secondary stomach and esophagus signal. He was then referred to our DMC for further diagnostic imaging as he had exhausted his local hospital’s capabilities. At our institution, all previous evaluations were reviewed with the patient including the MCD testing results, negative imaging, and other test results. A detailed history and physical examination were completed and was unremarkable except for an enlarged left testicle.
Due to the positive NET signal, a Dotatate PET scan was obtained, which was negative but did not visualize the distal pancreas well. The decision to use a Dotatate PET in NETs is due to its high sensitivity and specificity in detecting somatostatin receptor-expressing tumors (NETs often overexpress somatostatin). 6 An esophagogastroduodenoscopy (EGD) and colonoscopy were obtained, which were negative other than a 5 mm hyperplastic colon polyp (given the high frequency of NETs in the GI tract). The EGD showed LA grade A esophagitis, a 15 mm submucosal lesion at 25 cm from the incisors, and bulging of the major papilla. After discussion with the proceduralist, an EGD with endoscopic ultrasound was ordered to further characterize the submucosal lesion, the distal pancreas, and the major papilla. On endosonographic evaluation, no true abnormality was found. The submucosal lesion found on EGD was pulsatile and consistent with the arch of the aorta. The ampulla was prominent but normal. The only other incidental findings were a fatty liver and pancreas. No areas concerning a NET were identified.
To investigate the enlargement of the left testicle discovered, an ultrasound (US) was obtained. The decision to choose the US is because it is considered the first-line imaging modality to confirm the presence of a testicular mass, determine whether it is intra- or extratesticular, and assess the contralateral testis. 7 This showed a focal solid hypoechoic slightly vascular mass measuring 2.1 cm. Serum tumor markers should be assessed both before and after orchiectomy to aid in prognosis and staging. 7 Beta-hCG was elevated (2.2 IU/L; normal <1.4 IU/L) but alpha fetal protein and prostate-specific antigen were normal. CT chest/abdomen and pelvis had previously been obtained and were unremarkable as mentioned above. A urology referral was placed. An orchiectomy was performed a few days later with a preoperative differential diagnosis of primary testicular cancer, lymphoma, or metastatic disease. The surgical specimen showed a fleshy 2.1 cm mass (Figure 1). Pathology was consistent with seminoma confined to the testis. Given the lack of abnormal lymph nodes on imaging obtained over the course of the evaluation, no lymphadenectomy was performed.
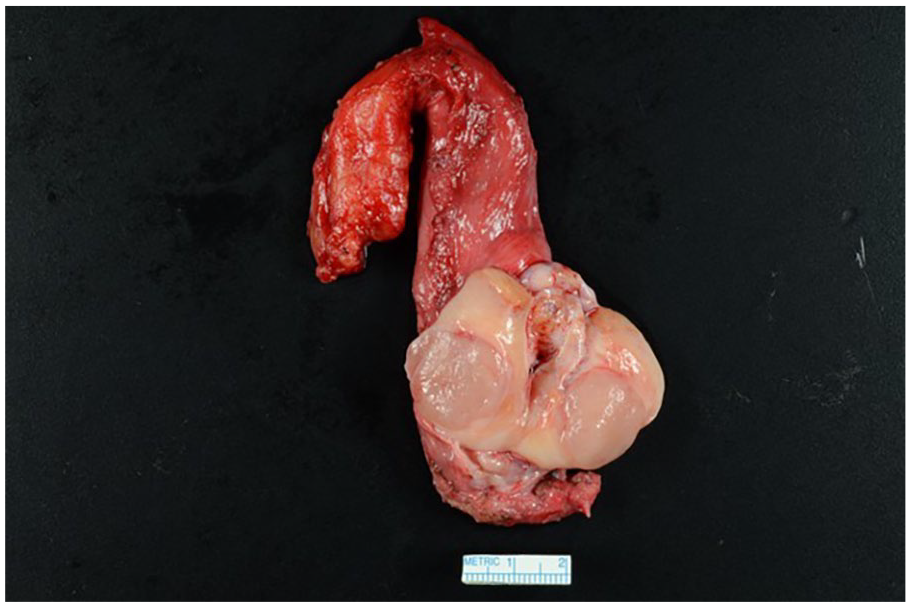
Left orchiectomy gross specimen.
Three months following the orchiectomy, the patient returned for surveillance. Surveillance CT scans were negative and beta-hCG had returned to undetectable (<0.7 IU/L). The repeat MCD test was also negative (no signal detected) after having two previously positive tests both with the neuroendocrine signals.
Discussion
This report highlights a patient with a positive MCD test with CSO NET origin whose initial workup at his local institution was negative. However, a thorough clinical evaluation, prompted by the persistence of positive MCD signal on repeat testing, led to the identification of an early-stage seminoma. Using specialized imaging and procedures, we evaluated the positive MCD signal and identified the underlying cancer that was suspected to have triggered the positive test.
A core aspect of this evolving technology is that MCD is used as a screening tool for asymptomatic individuals and presents a result known as a CSO. While an emerging topic of investigation, several studies have shown that the accuracy of the CSO identified in one MCD test was around 90%.5,8 As MCD testing is increasingly utilized in clinical practice, clinicians will need to be fully aware of the intricacies of ordering and following up MCD testing, as well as capable of educating patients on the nuances this testing brings to cancer screening and evaluation.
One of the challenges of widescale implementation of MCD testing is the lack of protocolized approaches to positive test signals. 5 With the concerns about the low positive predictive value of the test for numerous cancer types, it is unclear what constitutes a false positive and what follow-up is indicated. As seen in this presented case, false positives can occur when pertaining to the CSO, as there was no evidence of Head and Neck cancer or NET. A detailed evaluation however did identify a yet undiagnosed cancer. Interestingly, a review of the literature revealed previous studies indicating that cell-free DNA methylation patterns can serve as biomarkers for testicular germ cell tumors and seminomas. 9
A systematic approach to the workup is necessary before labeling an MCD test as a false positive. In this case, the consecutive persistently positive test report warranted a repeat evaluation with a thorough history and physical exam and more detailed, protocolized diagnostic testing. Of note, repeating MCD testing after the ensuing treatment of an identified cancer is not its intended use. MCD testing is intended to be used as a screening test for cancer among asymptomatic individuals, but not to be used as a biomarker for monitoring patients after cancer treatment. Ongoing pragmatic trials will further the knowledge and improve the future management of patients with positive MCD tests.
Conclusion
With the widespread adoption of MCD testing, expertise in the management of complex medical situations and diagnostic dilemmas will be key to completing an evaluation of positive test signals. Ensuring that providers at all levels in healthcare are equipped with the knowledge of how to effectively manage and follow up MCD testing will be paramount as this technology evolves or referral to centers with such expertise will be necessary. Future studies of larger populations undergoing MCD testing, along with longer duration follow-ups, will help enhance understanding of the role of MCD as a screening tool in the future.
Footnotes
Acknowledgements
We want to thank our patient for providing consent and permission to publish this case and share his story.
Author contributions
E.A.G., A.K.G., S.S., I.T.C., and R.T.H. all authors were involved in the writing, review, and approval of the content of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
