Abstract
Myoepithelioma is an extremely rare salivary gland neoplasm with diverse histological variants. Understanding myoepithelioma’s clinical, radiological, and molecular features is crucial for accurate diagnosis and management. We present a case of a 40-year-old Iranian male with a painless, firm, solitary exophytic mass on the upper lip. Surgical excision was curative, with no recurrence at 36 months. Histopathological analysis confirmed myoepithelioma with spindle and plasmacytoid cell variants. Myoepithelioma’s rarity is reflected in epidemiologic studies and its features are mainly recognized by the information in the case reports. Majorly it appears at the site of major salivary glands and occasionally the palate. The occurrence at the site of the upper lip is extremely rare and only appeared in a few case reports. Seeking the history of previous exposures to possible etiologic factors might not be a facilitator. Differential diagnosis includes pleomorphic adenoma, myoepithelial carcinoma, and basal cell adenoma. The main diagnostic approach is histopathological analysis. Following that, the tumor’s nature, cell variants, and possible transformation could be evaluated. While molecular studies supplement diagnosis, their routine clinical use is limited and their necessity is debated. The application of artificial intelligence could be helpful when uncertainty arises, or for analyzing microscopic images. Surgical excision with healthy margins is curative, and follow-up is essential.
Introduction
Neoplastic transformations of the salivary glands (SGs) give rise to various types of salivary gland tumors (SGTs), which are categorized based on the differentiation of the involved cells, determining their benign or malignant potential. However, such lesions remain infrequent, with a global annual incidence estimated at 0.4–13.5 cases per 100,000 individuals. 1 When neoplasms display benign behavior and differentiation toward myoepithelial cells (MCs), they are termed myoepithelioma (ME). Initially proposed by Sheldon, ME was formally recognized as a distinct pathological entity by the World Health Organization (WHO) in its 1991 classification of SGTs.2,3 ME constitutes a rare lesion, accounting for approximately 1%–1.5% of all SGTs. 4 Diagnosis primarily relies on histopathological evaluation, yet the intricate histological subtypes of ME have led to varying interpretations among pathologists, resulting in disparate classifications. This divergence occasionally leads to misinterpretations, particularly when ME lesions resemble other entities like pleomorphic adenoma (PA). Given the relative scarcity of ME cases and the lack of consensus regarding its histopathological subtypes, meticulous documentation and continual updates regarding this lesion’s characteristics are imperative, particularly when confronted with unconventional scenarios.
This case report is notable as, in this patient, ME is situated in an atypical anatomical location, exhibits unconventional histopathological features, and has occurred in an Iranian population that has a low incidence of ME.
Case presentation
A 40-year-old Iranian male patient was referred to the dental faculty of Semnan University of Medical Sciences. A written consent was obtained from the patient. The patient’s chief complaint was the presence of an upper lip swelling for the last 6 months. The head and neck lymph nodes were inspected for pain, swelling, asymmetry, or erythema. No abnormal finding was observed. Upon physical examination, an isolated exophytic mass was identified on the upper lip. The mass appeared smooth in texture, painless, and exhibited a firm consistency. Notably, it had well-defined borders and natural surface coloration. The patient was referred to a surgeon for comprehensive treatment. Excisional biopsy and histopathological evaluation were planned. Given the dimensions, clinical features, and location, further assessment through radiological modalities was deemed unnecessary for treatment planning or diagnosis.
Subsequently, under local anesthesia, the patient underwent the procedure. The excision included a 5 mm intact rim and a suspicious lesion of submucosal SGTs. An artificial dermis covered the surgical defect. The specimen, encapsulated and intact, was sent to the Pathology Department of Kowsar Hospital at Semnan in a 5% formalin solution. Macroscopic examination unveiled an oval, one-piece, firm tissue with white-brown hues, measuring approximately 0.8 × 0.3 × 0.5 cm. Cross-sectional sections were prepared and subjected to hematoxylin and eosin staining. The histopathological analysis of the specimen disclosed characteristic features consistent with SG neoplasms with myoepithelial differentiation (Figures 1–5).
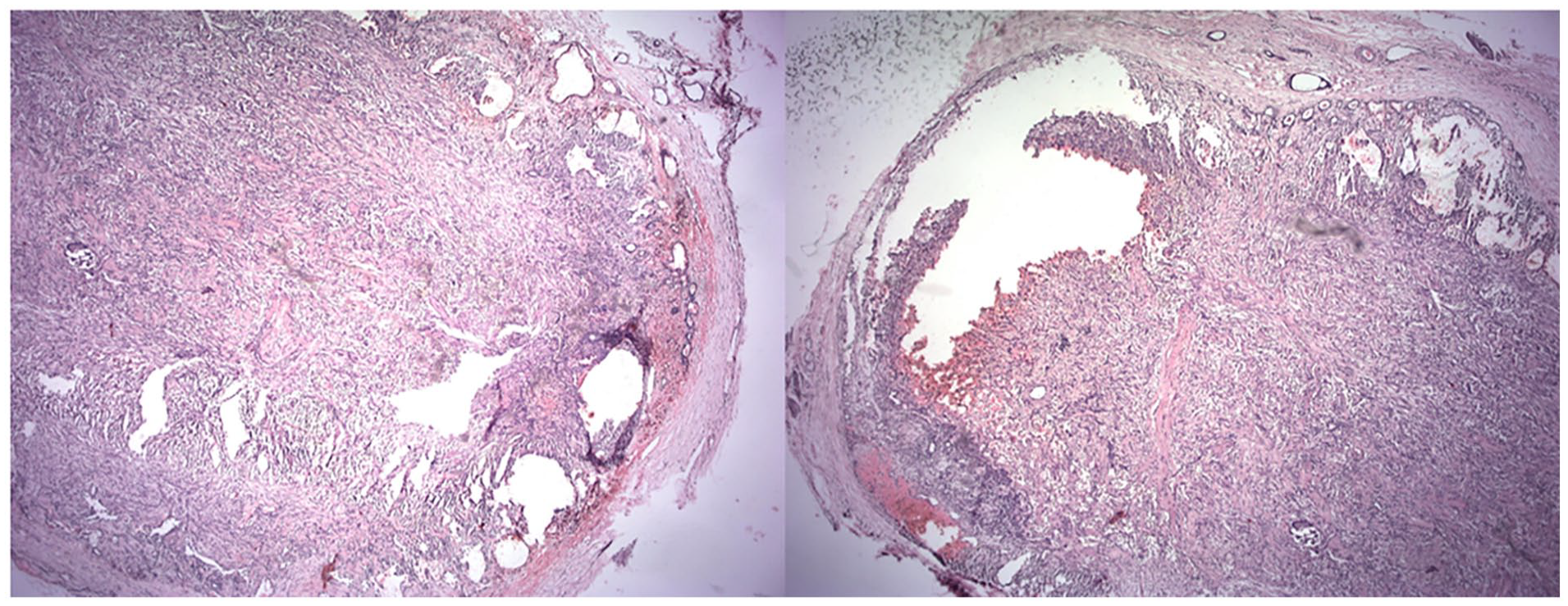
H&E staining ×4 magnification.

H&E staining ×10 magnification.

H&E staining ×40 magnification: Epithelioid cells (left) and clear cells (right).

H&E staining ×40 magnification: plasmacytoid cells.
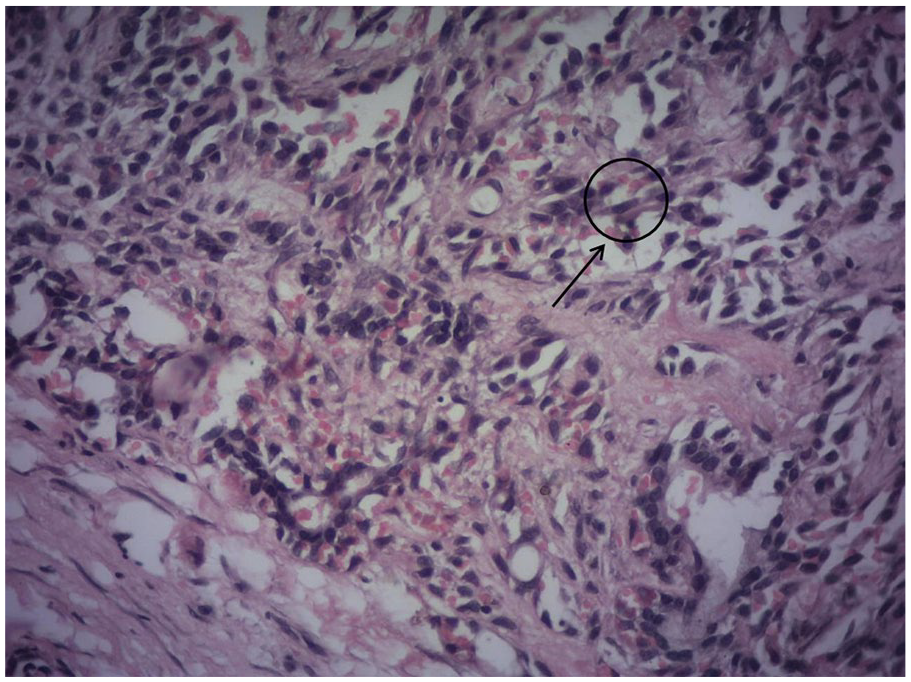
H&E staining ×40 magnification: spindle cell.
Notably, the lesion exhibited no malignant attributes or invasive behavior. Microscopic slides show a benign SGT revealed cells with myoepithelial differentiation, encompassing a diverse morphological spectrum ranging from spindle to plasmacytoid variants. Infrequently, regions with clear cells were discernible. Ductal structures were observed sporadically, while no evidence of osseous or chondroid differentiation was detected. The lesion exhibited encapsulation by fibrous tissue, with the surrounding margin displaying entirely unremarkable tissue.
Given the definitive histopathological diagnosis of ME, additional molecular investigations were not pursued. Continuous monitoring was scheduled, and at the 36-month follow-up mark, no signs of recurrence were evident.
Discussion
ME is a rare lesion often encountered in case reports. A multinational study spanning from 2006 to 2019 evaluated the prevalence of SGTs globally and identified a mere 62 cases of ME, constituting only 1% of all tumors. 5 ME, recognized as a benign ectodermal neoplasm primarily composed of differentiated MCs, necessitates precise clinical and pathological delineation to distinguish it from similar lesions. Before its classification as a distinct entity (ICD-O: 8982/0), 3 ME was considered a variant of PA. The WHO has underscored the differentiation between these entities based on distinction in their growth patterns, more aggressive behavior, and the absence of ductal differentiation. 3 Additionally, some authors believe that ME is clinically important due to its more aggressive nature, higher transformation potential into myoepithelial malignancy, 3 and its effect on SGTs. MCs are shown to play a role in suppressing or developing SGTs6,7; hence, their malfunction might be concerning. However, the diagnosis is challenging for several reasons such as variable histopathology, non-pathognomonic clinical appearance, non-specific biopsy, and rarity.5,8,9 Here, the diagnostic approaches to ME are briefly reviewed.
Etiologic approach
Physiologically, MCs reside in the basal and peripheral regions of intercalated ducts, facilitating secretion expulsion through their contractile capabilities. 7 Despite their antitumor function, MCs may also undergo a neoplastic process resulting in SGTs such as ME.7,9 Additionally, studies have suggested stem cells with the potential to differentiate into both epithelial and MCs as the likely origin of ME. 10 Interestingly, Argyris et al. reported the emergence of ME in the buccal cheek after detecting a low-grade pleomorphic adenocarcinoma of the upper lip in a female child, suggesting the possibility of common or related etiologic factors. However, given the scarcity of cohort or longitudinal studies, the exact etiology and specific risk factors of ME remain elusive. Thus, seeking a history of specific etiologic factors may not be strategic.
Clinical approach
Classically, ME presents as described in this case. Usually, it appears in the SG regions as a solitary, smoothly-surfaced swelling with a broad base. ME tends to be asymptomatic, non-tender, well-defined, and firm in consistency. Though the core may exhibit coloration ranging from brown to yellow,11,12 the covering mucosa may remain unaltered. The tumor progresses slowly over months to years, which possibly but not commonly can reach sizes of up to 5 cm. 8 However, the presence of a clear cell variant, although benign, can indicate an unpredictable course. While its age of involvement varies, the incidence appears to peak around 30–40 years. Although ME lacks gender or specific racial predilections, limited reports are available concerning the Iranian population. A recent systematic review of 2870 SGT cases in this population revealed no report of ME. 13 However, some sporadic case reports of ME as an SGT are reported from this population. While MCs exist in all SGs, the parotid gland (40%) is the primary site of origin for salivary ME, followed by minor SGs (predominantly the palate) and submandibular glands respectively.14,15 In rare instances, involvement of buccal mucosal glands has been documented. 16 Occurrence in lips, as reported here, is exceedingly uncommon. A recent review of 2280 SGT global case reports only revealed three cases of ME located in the upper lip. 15 As none of the mentioned features are exclusive, the clinical approach is not diagnostic and may confirm the presence of an SGT at most, but not specifically.
Radiologic approach
Radiologic modalities help determine the extent, origin, spread, and relation to adjacent structures, or guide clinicians in biopsy. 17 Although CT scans are well established for SGTs, including ME, MRI could be preferred due to superior soft tissue contrast.18,19 However, CT scans may be resealable especially for lesions located in the hard palate due to the risk of invasion to the bone 9 or ruling out osseous origin. Wang et al. described radiologic features of ME in the parotid gland as well-circumscribed, enhancing mass lesions with smooth or lobulated margins, typically located in the superficial lobe of the parotid gland and abutting the capsule which may contain enhancing nodules and non-enhancing areas, such as linear bands, slit-like structures, or cysts. 18 In brief, radiologic approaches may be helpful in cases where uncertainty arises, or be needed for presurgical evaluation, though they fail to distinguish ME specifically.
Cytopathologic approach
Fine needle aspiration (FNA) is investigated for diagnosing SGTs like ME. FNA smears can show variable morphologies mirroring the ME variant. 20 However, due to this variety and stroma composition diversity, the diagnosis could be challenging, and more evaluation might be needed. 21 Some misdiagnoses are also reported such as extra-skeletal myxoid chondrosarcoma. 20 Hence, the cytopathologic approach could be beneficial but not sufficient. Also, when the excisional biopsy of a small lesion is planned, as in the presented case, further evaluation utilizing FNA might be difficult to justify.
Histopathologic approach
This is the most efficient approach as the lesion’s definition relies on it. Although it was initially defined as a neoplasm that is only composed of MCs, the WHO classification also considers the absence of chondroid or myxochondroid stroma. 9 The histopathological diagnosis and classification of ME are based on the morphological attributes of neoplastic cells, which are classified into three variants: spindle, plasmacytoid, and clear cell. An additional oncocytic variant has been proposed, resembling oncocytoma. Two or three cell types are typically observed, with the dominant variant determining the lesion’s variant. Occasional reports of mixed-form ME with coexisting morphological diversity have also been presented. 22 In contrast to the presented case, spindle variants are the most common subtype and are more prevalent in parotid glands, while plasmacytoid variants dominate minor SGs of the palate.
The spindle variant comprises ample cellular content with minimal stromal background. Stromal elements may consist of myoepithelial, fibrous, or hyalinized cells, with myxoid and lipomatous stroma being rare. Contrary to the presented case, a thin capsule is often present in parotid presentations but not in minor SG involvement. Notably, ME lacks ductal structures within the lesion, differentiating it from PAs. While some pathologists reject any ductal structure in ME, others accept up to 5% presence, categorizing higher levels as PA.
Molecular and genetic approach
Evaluating markers independently of histopathology cannot definitively diagnose ME due to the existence of various overlapping conditions, the broadness of reported markers, and the molecular profile diversity of soft tissue MEs. However, they can be beneficial as supplementary tools to determine cell differentiation status, distinguishing other possible diagnoses, or for research purposes. Nayak et al. suggested immunohistochemical evaluations to distinguish ME, PA, and malignant myoepithelial carcinoma as ME occurrence might be underestimated based on non-immunohistochemical evaluation. 23 Several immunohistochemical markers have been proposed for evaluation, with the most notable being S-100, cytokeratin, GFAP, and Vimentin. However, the immunophenotype of cells may vary either among different tumors or the cellular population of a single tumor. 24 Moreover, there have been reports of certain genetic profile alterations in the involved cells, but these changes seem to lack clinical significance and diagnostic value. On the other side, it is supported that soft tissue MEs may exhibit heterogeneous genetic profiles, 25 which can complicate molecular differential diagnosis.
It’s worth mentioning that there are some reports of similarities in the genetic alterations between ME and other myoepithelial lesions, including PA.25,26 Also, considering MCs of some myoepithelial lesions as part of a spectrum within PA due to shared genetic anomalies has been proposed in some articles. 8 These data may raise concerns about diagnostic challenges and the accuracy of our understanding of myoepithelial tumors’ nature and relations. Furthermore, it may reinforce the concept of dynamic changes in differentiation and cellular morphology in myoepithelial tumors.
Artificial intelligence assistance
Artificial intelligence (AI)-assisted models for oral cancer diagnosis are currently still developing as the number of cases at the same diagnostic center is limited. Studies have shown that deep learning algorithms outperform professionals in some cases. Currently, it is believed that AI can be an effective assistant to the pathologist in a variety of digital pathologist aspects. For the analysis of histopathological slides, an important feature to be developed in deep learning is that it enables computers to automatically extract features from images and build a recognition algorithm. Deep learning methods, mainly convolutional neural networks, have been used as computational pathology techniques to analyze images of different tumors. These techniques can also be integrated into the assessment of demographic and molecular data and prognostic and therapeutic implications.28-30
To diagnose SGTs, the major applications of AI are processing microscopic and radiographic images. The assistance of AI has made it possible to accurately interpret technical information such as determining the percentage of each morphological cell type and its related variants, distinguishing between malignant and benign lesions, and evaluating the ratio of each positive marker in a more precise and quantitative manner. Hence, the utilization of AI is increasing in modern pathology, resulting in a rise in the number of AI-equipped laboratories. For instance, Alsanie et al. established a classifier based on Qupath to distinguish malignant and non-malignant SGTs. 27 However, the application of AI tools for histopathological evaluation of SGTs is still under development and needs more investigations to become a key approach. Currently, in the case of ME, AI can make the diagnosis process smoother by analyzing cell morphology, variant typing, marker analysis, and facilitating differential diagnosis when it comes to complexities and dilemmas. 31
Differential diagnosis
In cases where the lesion is located within an SG, lesions primarily composed of MCs raise differential diagnosis considerations, including the most significant ones such as PA, myoepithelial carcinoma, and basal cell adenoma. Fibromyxoid stroma and the presence of ductal structures are indicative of PA. Myoepithelial carcinoma consists of differentiated MCs but is a malignant lesion with malignant features and invasive behavior. Basal cell adenoma may share cellular attributes with ME but typically exhibits ductal structures. Depending on the variant of the lesion, the predominant cellular composition guides the differential diagnosis. In such cases, if a myoepithelial phenotype is identifiable, pathologists will face fewer challenges. However, in cases of uncertainty, marker evaluation could be helpful. For example, clear cell carcinomas of the kidney are negative for S100 protein, and ME is also negative for cytokeratin.
Treatment, prognosis, and recurrence
The primary treatment approach for ME is akin to that of PA, involving complete excision of the neoplastic tissue, often accompanied by enucleation. Despite the benign nature of ME, surgical resection with a clear margin is recommended, as recurrence, although infrequent, could occur and carry a risk of malignancy. 30 Hornick and Fletcher studied 101 soft tissue myoepithelial tumors, showing predominance of benign fashion in the case of benign or low-grade lesions with a 20% risk for local recurrence. 32 However, some other authors considered benign ME as a rare condition that may rarely be recurrent. 33 If the pathologist does not identify any signs of malignancy, there is currently no logical recommendation to add chemotherapy or radiotherapy to the treatment plan. The prognosis for ME is generally favorable, and it typically responds well to treatment. Nevertheless, leaving the patient untreated or the cured ones unfollowed is not without risk, as they may present with malignant transformation and recurrence, respectively. 34
Conclusion
ME, particularly as a neoplasm of minor SGs, presents challenges in differential diagnosis due to limited information. Our case, unique in its presentation in the upper lip, encapsulation contrary to expectations, and a combination of multiple cell types—predominantly spindle which is not common when appears in minor SG—highlights the need for reevaluation of ME characteristics. More attention to distinguishing ME, recognizing its diversity and nature, and updating our knowledge is warranted based on this and similar cases.
Footnotes
Acknowledgements
Thanks to the pathology department of Semnan Kowsar Hospital, who accepted a patient who was a 40-year-old Iranian man.
Author contributions
S.B.: project administration; conceptualization; writing—review & editing; supervision, formal analysis. F.G.: project administration, writing—original draft; writing—review & editing, data curation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Practical/clinical implications (key finding statement)
This is especially pertinent considering the critical need for precise diagnoses and the continuous refinement of classifications, particularly in situations deviating from the norm.
