Abstract
Pleomorphic adenoma is a benign tumor and the most common salivary gland neoplasm. Metastasizing pleomorphic adenoma shares histological characteristics with pleomorphic adenoma but exhibits malignant behavior, including local lymph node involvement and/or distant metastasis. Several potential risk factors for metastasizing pleomorphic adenoma have been identified, some of which are associated with incomplete tumor clearance due to inadequate surgical techniques used in the treatment of primary pleomorphic adenoma. Here, we present a rare case of metastasizing pleomorphic adenoma originating from the parotid gland and describe its clinical features. The patient was a 75-year-old female with a surgical history of enucleation of a pleomorphic adenoma in the left parotid gland. Total parotidectomy and left neck dissection were performed for the left parotid tumor this time, and the patient attended one postoperative outpatient visit but subsequently was lost to follow-up.
Introduction
Pleomorphic adenoma (PA) is a benign tumor and the most common salivary gland neoplasm, though it carries a risk of malignant transformation into carcinoma ex pleomorphic adenoma (CXPA). 1 Rarely, PA may exhibit malignant behavior, such as local lymph node involvement and/or distant metastasis, known as metastasizing pleomorphic adenoma (MPA). 2 A systemic review analyzed 81 cases published between 1942 and 2014. The report identified potential risk factors for developing MPA, including younger age at initial presentation of PA, incomplete tumor resection, local recurrence, and tumor spillage. 3 Here, we present a case of a left parotid tumor with neck lymph node metastasis. Total parotidectomy and left neck dissection were performed. Final pathology revealed recurrent and metastasizing PA. This case is presented to describe its clinical features and to raise awareness of this disease.
Case presentation
A 75-year-old female had history of hypertension, type II diabetes mellitus, and pulmonary tuberculosis, which was treated from 2012 to 2013. In 2012, she underwent enucleation of a left parotid PA. Recently, she noticed a left infra-auricular mass that had persisted for 6 months and sought evaluation at the ENT outpatient department of Kaohsiung Veterans General Hospital for surgical intervention. Neck sonography revealed a left parotid mass measuring 1.4 cm × 0.8 cm and suspected metastatic lymph nodes at the left neck level III. Fine needle aspiration of the parotid tumor yielded cytopathology results indicative of a salivary gland neoplasm of uncertain malignant potential. CT scan showed some heterogeneously enhancing soft tissue involving the inferior pole of the left parotid gland with downward extension along the left sternocleidomastoid muscle (SCM) and regional skin (Figure 1(a) and (b)). These findings were likely associated with post-aspiration change or/and metastatic lymph node involvement. Additionally, two enhancing nodules were noted within the left parotid gland, suspected to be metastases or reactive lymph nodes (Figure 1(c) and (d)). Subsequently, she underwent total parotidectomy and neck dissection (Left levels II-Va; Figure 2(a)). A SCM rotation flap was used to fill the resulting space for cosmetic purposes (Figure 2(b)). Microscopic examination revealed multiple nodules with predominantly myoepithelial cell proliferation, admixed with stromal components containing myxoid areas (Figure 3). The lymph node sections from the left neck level II showed scant lymphoid tissue replaced by myoepithelial cells with spindle and focal plasmacytoid features, admixed with stromal components containing myxoid and chondroid areas (Figure 4). Focal lipomatous differentiation was also identified, and the epithelial component was inconspicuous. The pathology report confirmed recurrent and metastasizing PA. Following the surgery, the patient attended one postoperative outpatient visit but was subsequently lost to follow-up.
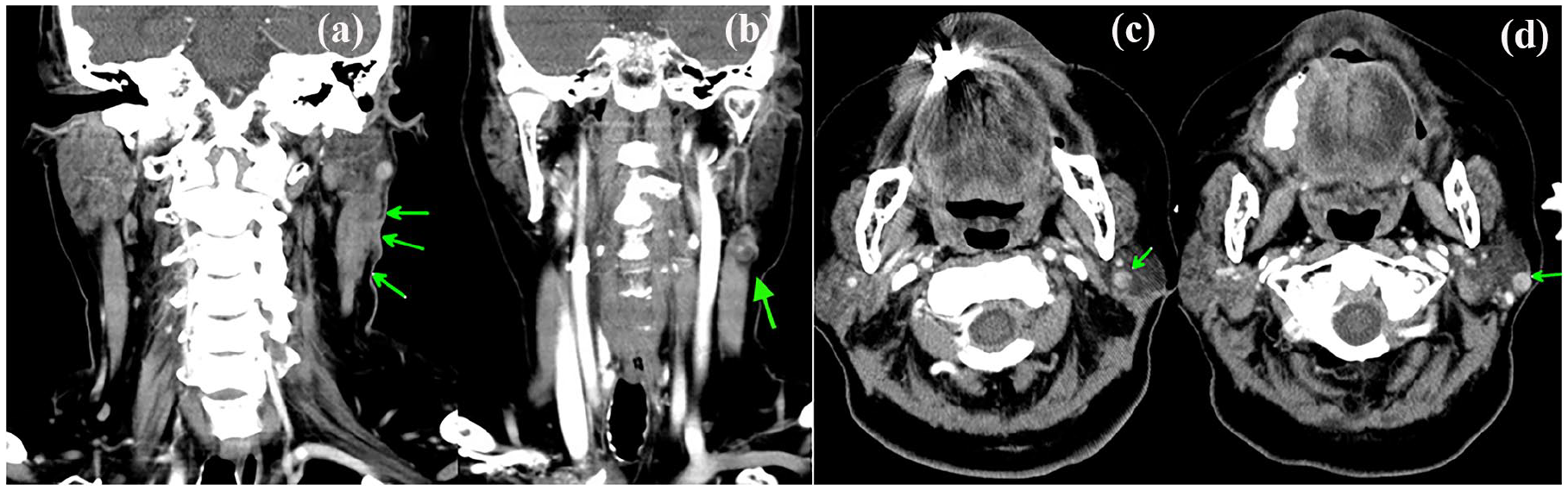
Contrast CT: (a) enhancing soft tissue involving the left parotid with extension along the left sternocleidomastoid muscle and skin (green arrows); (b) one heterogeneously enhancing nodule over the left neck level II, suspected metastatic lymph node (green arrow); ((c) and (d)) two enhancing nodules (green arrows) over the left parotid gland, suspected metastases or reactive lymph nodes.

(a) Left total parotidectomy and neck dissection were performed, and the left facial nerve and its branches (yellow arrows) were well preserved. (b) The sternocleidomastoid muscle rotation flap (yellow arrow) filled the defect after the parotidectomy.

Microscopic examination of the specimen of the parotid gland showed (a) multiple nodules; (b) stromal components containing myxoid areas (green arrow); ((c) and (d)) positive immunohistochemical staining for calponin and CK 14 respectively, indicating myoepithelial cells.

Microscopic examination of the specimen of the lymph nodes from the left neck level II showed (a) stromal components containing myxoid areas (green arrow); (b) positive PLAG1 immunohistochemical staining suggesting pleomorphic adenoma; (c) positive SOX-10 IHC staining suggesting myoepithelial cells.
Discussion
PA, a benign and slow-growing tumor, is the most common salivary gland tumor, with incidence rates ranging from 2.4 to 4.9 per 100,000 persons per year. 1 The current primary treatment for PA is surgical excision, aimed at correcting cosmetic deformities caused by the tumor, diagnosing and treating potential undetected malignant neoplasms, and preventing malignant transformation. The risk of malignant transformation of PA to CXPA is estimated to be 1.5% within the first 5 years of diagnosis if left untreated, 10% if left untreated for more than 15 years, 1 and between 0% and 23% for recurrent PA. 4
The case had previously undergone enucleation of the left parotid PA and later presented as recurrent and metastasizing PA. MPA is rare and characterized by benign histology but malignant behavior. The 2023 classification of head and neck tumors by WHO describes it as follows: “Benign metastasizing PA is histologically and molecularly indistinguishable from a benign tumor at a primary location. No histological or molecular features can reliably predict metastasis.” 5 In the past, the International Classifications of Diseases for Oncology (ICD-O) assigned a benign behavioral code “/1” that corresponded to neoplasms of uncertain and unknown behavior. 6 The latest ICD-O-3.2 assigns a behavioral code “/3” that corresponds to malignant tumors. 5 Knight et al. conducted a systematic review of MPA and identified 81 case reports from 1942 to 2014. 3 The mean age of the 81 cases at the diagnosis of MPA was 49.5 years, and the most common metastatic sites were bone (36.6%), lung (33.8%), and neck lymph nodes (20.1%). About 74.1% of MPA cases originated from primary parotid PA, while 14.8% originated from the submandibular gland. Out of 51 cases of benign MPAs with reported survival data, 9 patients (17.6%) died due to the disease, while 41 (80.4%) were alive at 1 year. Multiple metastases are associated with a poor prognosis.3,5
PA commonly presents in individuals with an average age of 43–46 years. While in patients who develop MPA, the average age of presentation for PA is 34.3 years, with 62.9% of cases occurring before the fifth decade. 3 Hence, a younger age at presentation of PA may serve as a potential risk factor for the development of MPA. Other risk factors for MPA include local recurrence and tumor spillage. The literature indicates that 72.8% of cases reported local recurrence before developing MPA, with 37% demonstrating multiple local recurrences. 3 Additionally, incomplete tumor clearance is strongly associated with local recurrence,3,4,7 and thus the recurrence rate varies depending on the surgical approach utilized. Simple enucleation of PA is associated with high recurrence rates, ranging from 8% to 45%. This rate is reduced to less than 5% with superficial parotidectomy and further decreases to 0.4% with total parotidectomy.3,4,7–9 One proposed hypothesis regarding the mechanism of MPA suggests that it may occur iatrogenically during surgical resection, with disrupted tumor cells seeding into the bloodstream. 3 Therefore, thorough tumor resection with adequate margins should be performed, and enucleation should be avoided. The case involved a 75-year-old female who had undergone enucleation of the left parotid PA at around 64 years of age. Although the previous specimen is not available for examining the surgical margins, it is possible that enucleation as the surgical approach for the primary PA contributed to the recurrence and metastasis of PA.
Due to the extended latency between PA resection and subsequent MPA, it is recommended that patients with incomplete excision, surgical spillage, or local recurrence undergo long-term follow-up for MPA. 3 Some suggest that patients with local recurrence should be investigated for MPA using Positron Emission Tomography (PET). 10 Regarding the treatment of MPA, total surgical resection is considered the treatment of choice as long as the tumor is operable. There is no meaningful evidence of improved survival with postoperative adjuvant radiotherapy. For some cases with inoperable tumor, tamoxifen treatment alone or radiotherapy alone has been reported. 3 The case we reported should be regularly followed up after surgical intervention for other potential missed MPA elsewhere in the body and even investigated by PET if she was not lost to follow-up.
Conclusion
In summary, MPA is a type of benign tumor with malignant behavior. Potential risk factors contributing to MPA include younger age at presentation of primary PA, incomplete tumor resection, local recurrence, and tumor spillage. To prevent the development of MPA, complete tumor resection with adequate margins should be performed instead of enucleation as the primary treatment for PA.
Footnotes
Author contributions
The authors confirm contribution to the paper as follows: conception and design of the case report: Wan-Hsun Tsai, Bor-Hwang Kang; data collection: Wan-Hsun Tsai; analysis and interpretation of the case: Wan-Hsun Tsai Bor-Hwang Kang; draft manuscript preparation: Wan-Hsun Tsai, Bor-Hwang Kang. All authors reviewed the results and approved the final version of the manuscript.
Data availability
All data underlying the results are available as part of the article and no additional source data are required.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for anonymized patient information to be published in this article.
