Abstract
Opsoclonus-myoclonus syndrome is a rare neurological condition characterized by opsoclonus, myoclonus, ataxia, irritability, and sleep disturbances. In pediatric patients, symptoms usually start between 16 and 18 months of age; opsoclonus-myoclonus syndrome presentation in children under 6 months is rare. Approximately 50% of cases are associated with neuroblastoma. We report an early onset presentation of opsoclonus-myoclonus syndrome in a previously healthy, 3-month-old female infant. The diagnostic workup revealed no abnormalities. The patient underwent monthly cycles of dexamethasone pulses and intravenous immunoglobulin with a favorable response. After a few months, the patient presented intermittent opsoclonus before the next scheduled pulse so from the 9th cycle onwards, the intravenous immunoglobulin dose was increased to 2 g/kg. After 9 months of treatment, she was diagnosed with a latent Mycobacterium tuberculosis infection. Due to this infection, dexamethasone pulses were discontinued, and intravenous immunoglobulin treatment was maintained with clinical improvement The patient received 18 intravenous immunoglobulin cycles, leaving her with a score of one on the Mitchell-Pike scale. Developmental milestones have been attained according to age. Despite the range of therapeutic options for managing opsoclonus-myoclonus syndrome described in the literature, the efficacy of these available therapies needs to be better established. A modified upfront approach with dexamethasone and intravenous immunoglobulin could be an option in settings where rituximab is unavailable.
Keywords
Introduction
Opsoclonus-myoclonus syndrome (OMS) is a rare autoimmune neurological disorder, with an approximate incidence of 0.18–0.6 cases per million per year in the pediatric population.1,2 It is characterized by opsoclonus (erratic eye movement), myoclonus, ataxia, irritability, and sleep disturbance. 3 About 50% of cases are associated with neuroblastoma, followed in frequency by post-infectious OMS.4,5 Several studies show that the median age of onset of symptoms in children is between 16 and 18 months, with no gender preponderance.1,4–7 Treatment options include corticosteroids, human immunoglobulin (IVIg), cyclophosphamide, rituximab, and plasmapheresis. 3 However, despite immunotherapy, patients present with various neurodevelopmental sequelae during follow-up.3,5,8 We report an early onset presentation of OMS in a previously healthy, 3-month-old infant.
Case report
A previously healthy 6-month-old female patient was admitted to our hospital with a 3-month history of abnormal eye movements and sporadic myoclonus. The patient initially exhibited intermittent abnormal eye and upper eyelid movements, approximately 15–20 times daily. By 4 months of age, the abnormal eye movements were constant and erratic.
Medical history indicated that the patient was born preterm at 36 weeks, with adequate weight and a good Apgar score. She was hospitalized at birth for a week due to transient tachypnea of the newborn. Her mother had preeclampsia during pregnancy, she was taking oral contraceptives pills (OCP) and received ivermectin and hyoscine during gestation, before being aware of the pregnancy, at about 25 weeks of gestational age. There is a family history of epilepsy; the patient’s father has idiopathic generalized epilepsy.
Upon admission, she had a heart rate of 110 beats/min, respiratory rate of 22 breaths/min, temperature of 36.5°C, oxygen saturation of 99%, and a BP of 110/62 mmHg. The patient presented continuous, multidirectional opsoclonus. Motor function evaluation revealed symmetrical strength and reflexes, with slightly increased muscle tone in extremities. She had truncal ataxia while sitting. Cranial nerve assessment demonstrated abnormal fixation and following movements, facial symmetry, normal hearing responses to auditory stimuli, intact palate elevations and gag reflexes, and normal tongue movements. The rest of the physical exam was normal. The diagnostic workup revealed a normal brain computed tomography, brain magnetic resonance imaging (MRI), and whole-body positron emission tomography (PET) scan (Figures 1 and 2). The cerebrospinal fluid (CSF) was normal. TORCH serology and COVID tests were negative. Urine catecholamines were also in the normal range.
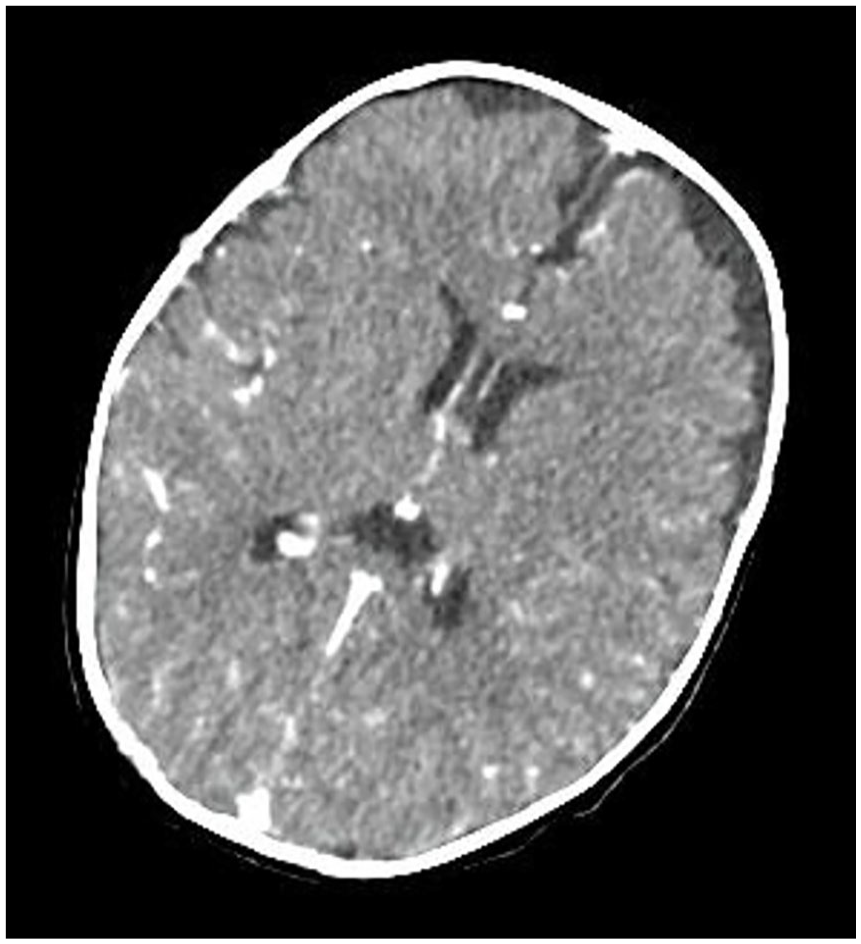
Axial brain computed tomography scan showing normal contrast enhancement and physiological enlargement of the subarachnoid space.

Brain PET-CT scan showing normal cerebral metabolism.
She was diagnosed with idiopathic OMS and received pulses of dexamethasone and IVIg with an initial dose of 20 mg/m2/day of dexamethasone for three consecutive days plus 2 g/kg of IVIg, followed by monthly pulses of 1 g/kg of IVIg with dexamethasone. Myoclonus and ataxia resolved quickly, but after a few months, the patient presented intermittent opsoclonus before the next scheduled pulse (four points on the Mitchell-Pike scale 3 ), so from the 9th cycle onwards, the IVIg dose was increased to 2 g/kg. After 9 months of treatment, she was diagnosed with a latent Mycobacterium tuberculosis infection due to a positive Mantoux test and a negative chest X-ray and received chemoprophylaxis with isoniazid for 6 months. Due to this infection, dexamethasone pulses were discontinued, and IVIg treatment was maintained, achieving clinical improvement (score of 1 on the Mitchell-Pike scale). The patient received IVIg for a total of 18 cycles, leaving her with a score of 1 on the Mitchell-Pike scale. Visual evoked potentials (VEP) at 1 year and 9 months showed bilateral dysfunction of visual pathways of probable demyelinating cause. Fundus examination was normal. Eye fundoscopy remained normal. Follow-up brain MRI at 2 years of age was normal (Figure 3). Developmental milestones have been attained according to age.

Follow-up brain MRI at 2 years of age. Axial T1-weighted image (a) with corresponding T2-weighted image (b) and Fluid-attenuated inversion recovery (FLAIR) image (c) showing no signs of inflammatory lesions.
Discussion
OMS has an incidence of 0.18–0.6 cases per million per year in the pediatric population.1,2 Approximately 50% of cases are associated with peripheral neuroblastic tumors, with neuroblastoma being the most frequent type of tumor (73%), followed by ganglioneuroblastoma (22%) and ganglioneuroma (4%).3–5 Although rare in adolescents, an association with ovarian teratomas has been reported. 3 OMS has also been reported in association with malnutrition, 9 infections such as SARS-CoV-2, 10 and drugs. 11 We have not found a specific relation with the use of prenatal hyoscine or OCP.
The presence of neural antigens in neoplasms and the associated inflammatory reaction have been determined as the probable cause of the relatively pathognomonic clinic of this disease. The exact immunopathogenesis is unclear; however, an abundant lymphocytic inflammatory infiltrate associated with tumors has been reported. CSF studies detected elevation of cytokines and chemokines, presence of oligoclonal bands, participation of B lymphocytes, and, to a lesser extent, T lymphocytes. 12
There are some case reports of pediatric patients with OMS in Latin America. In Colombia, three cases were reported in patients aged 13 months, 15 months, and 14 years, with thoracic ganglioneuroblastoma, paravertebral ganglioneuroblastoma, and ovarian teratoma, respectively.13–15 Despite the active search for a neoplasm, there were no relevant findings in our patient’s MRI, computed tomography, and PET scans. It is important to highlight that the finding of neoplasia can be detected before, during, or after the clinical expression of the disease, therefore an active search for a tumor is essential during follow-up.
In Argentina, a 2-year-old boy with OMS was treated with human gammaglobulin for neuroblastoma that required surgical resection. 16 In Brazil, a 17-month-old child with OMS was reported, and in Uruguay, another 28-month-old child with OMS was reported.17,18 In these cases, patients required surgery, corticosteroids, and immunomodulatory therapy. Additionally, in Peru, OMS secondary to neuroblastoma was reported in a 1-year-old patient.17–19 Like previous studies, OMS was secondary to a paraneoplastic neurological disorder that required surgery. In contrast, no neoplasia was found in our patient. She received corticoids and IVIg monthly to control symptoms. She continues to be monitored, with an active search for neoplasia.
Symptom onset is usually after 6 months, mainly between 16 and 18 months of age.2,4,6,7,20,21 A prospective study in the United Kingdom described a patient with an onset of symptoms at 3 months of age, but, unlike our patient, this case was associated with thoracic neuroblastoma. 1
The most frequent clinical manifestations are opsoclonus and myoclonus, followed by ataxia and irritability. Additionally, about 30% of patients present with speech disturbance, head titubation, tremors, and sleep disturbances. Less frequent manifestations include dysmetria, loss of appetite, fatigue, vomiting, dystonia, hyperactivity, drooling, and hypotonia. 22 A diagnosis of OMS requires at least three of the following four criteria: opsoclonus, myoclonus or ataxia, behavioral changes or sleep disorders, and neuroblastoma. 3
OMS usually responds to immunotherapy, though the most effective drug has not been established. 21 Corticosteroids are the first line of therapy, but they have long-term sequelae, and OMS relapses still occur. Because of this, several multimodal treatment regimens have been proposed. For example, a regime of rituximab, adrenocorticotropic hormone (ACTH), and IVIg seems an effective option as rescue therapy, according to an observational study by Pranzatelli et al. 23 In addition, a retrospective study by Pranzatelli et al. 24 showed that combined immunotherapy with IVIg, dexamethasone, and rituximab reduced markers of neuroinflammation and improved clinical scores, though this study had several limitations such as a small sample size and prior treatment in some patients.
A recent consensus proposed two approaches: an aggressive upfront approach and a gradual approach. The upfront approach includes ACTH or corticosteroids, plus plasmapheresis or IVIg, plus rituximab or cyclophosphamide. 3 Examples of corticosteroid regimens include oral dexamethasone (20 mg/m2/day for three consecutive days as pulses 3–4 weekly and IV pulse methylprednisolone (30 mg/kg/day for 3–5 days, maximum 1 g/d) followed by oral prednisone or prednisolone (1–2 mg/kg/day). IVIg dose is 2 g/kg over 2–5 days followed by 1–2 g/kg every 4 weeks. Rituximab is usually given in doses ranging from 375 mg/m2/dose for 2–4 weekly doses to 750 mg/m2/dose for two biweekly doses. Cyclophosphamide dose is 750 mg/m2/dose monthly for 6 months. 3 The gradual approach starts with monthly cycles of dexamethasone at 20 mg/m2/day for three consecutive days; if the response at 3 months is inadequate, rituximab or cyclophosphamide is added. 3 With either approach, timely diagnosis and treatment are recommended, with a rapid escalation if there is an insufficient response.
Our patient was treated with a modified upfront approach therapy with only IVIg and dexamethasone; rituximab was not administered due to drug unavailability and since she achieved clinical remission no further escalation was needed.
Several factors can influence OMS prognosis, including the underlying etiology, age at onset, the severity of symptoms, and response to treatment.3,5,8,25 De Grandis et al. described children with an interval between symptom onset and OMS diagnosis longer than 2 months that showed a higher frequency of neurological sequela than children diagnosed earlier, although statistical significance was not reached. 7 Several neurodevelopmental and neurodevelopmental sequelae are observed in patients with OMS, such as cognitive problems, attention deficits, visual-motor difficulties, speech abnormalities, sleep disorders, and behavioral issues.3,5,8 A study by Yildirim et al. reported that approximately 43.5% of children with OMS achieved complete remission and 39% experienced relapse. 22 Similarly, a retrospective study of 13 patients by Urtiaga-Valle et al. reported that 46% had a full recovery, and 38% had neurodevelopmental disorders, primarily attention deficit disorder. 5 Abnormal VEP have been described by Sakuma et al., observing a correlation between the severity of the clinic and the intensity of abnormalities in the VEP. 26
Conclusions
This case contributes to the characterization of the OMS clinical spectrum and highlights the challenges in its management. When rituximab is unavailable, a modified aggressive approach with a combination of dexamethasone and IVIg could be a valid option. Because of the risk of neurological sequelae, timely diagnosis and treatment are essential to optimize the outcome of children with OMS. Response to treatment is variable, and further research is needed to establish optimal treatment regimens.
Footnotes
Acknowledgements
To the Department of Pediatrics from Hospital Nacional Edgardo Rebagliati Martins.
Author contributions
R.E.C.N., M.D.A.D.G., and K.J.R.D. participated in the conception and design of the article. R.E.C.N. and M.D.A.D.G. conducted the bibliographic search and participated in writing the article’s initial version. R.E.C.N., M.D.A.D.G., and K.J.R.D. participated in the critical review of the article and approval of the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case was obtained from the Institutional Review Board of Hospital Nacional Edgardo Rebagliati Martins (PROVEIDO N° 804-OFlyD-GHNERM-GRPR-ESSALUD-2022).
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
