Abstract
Presenting as a large vessel vasculitis, giant cell arteritis (GCA) manifests with various symptoms, including fever, myalgias, headache, and jaw claudication. Although the precise pathogenesis of GCA remains incompletely elucidated, there is speculation about the involvement of environmental factors and infectious agents like bacteria and viruses in its development. Nevertheless, data on the potential link between influenza infection and GCA are limited. In this report, we present the case of an 88-year-old patient diagnosed with GCA following a severe influenza A infection.
Introduction
Giant cell arteritis (GCA) and polymyalgia rheumatica (PMR) are prevalent inflammatory rheumatic diseases observed primarily in individuals aged 50 and above. 1 The pathogenesis of GCA is complex, involving intricate interactions between cells of both the innate and adaptive immune system. While the exact cause of GCA remains elusive, there is speculation regarding the potential involvement of environmental factors and infectious agents, including bacteria and viruses, in its development. 2 However, limited data exist regarding influenza infection as a possible trigger for GCA. Here, we present the case of an 87-year-old patient diagnosed with GCA following a severe influenza A infection.
Case description
An 87-year-old male presents in our emergency department exhibiting asthenia, fever, and a cough-producing brown sputum. He had a smoking history of approximately 20–30 pack-years, having ceased smoking 15 years ago. His comorbidities include Parkinson’s syndrome, which has been documented for the past 1.5 years. Physical examination revealed no significant findings. Laboratory analysis showed elevated C-reactive protein (CRP) levels of 27 mg/L alongside a normal white blood cell count (Table 1). Arterial blood gas analysis indicated type I respiratory failure with partial oxygen pressure at 9.6 kPa. Furthermore, the polymerase chain reaction test for influenza A was positive. Consequently, the patient was admitted to our acute geriatric ward to manage symptomatic influenza A infection. With oxygen supplementation at 2 L/min necessitated, a specific antiviral therapy involving oseltamivir was initiated for a duration of 5 days.
Laboratory parameters on admission, hospital day 12, hospital day 15 and discharge home.

ALAT: alanine aminotransferase; ALP: alkaline phosphatase; ASAT: aspartate aminotransferase; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; GGT: gamma-glutamyl transferase; Hb: haemoglobin.
Upon suspicion of a bacterial superinfection on hospital day (HD) 10, characterized by a drop in oxygen saturation to 88%, persistent productive cough and new infiltrates observed in the left lower lung lobe on chest X-ray, antibiotic therapy commenced with ceftriaxone at 2 g/day. In addition, the patient began experiencing symptoms of neck stiffness and right-sided headaches radiating to the right hip, unresponsive to basic analgesia. Similar headaches or joint pains were not mentioned in the detailed medical history. Even questioning relatives (the patient’s wife and daughter) did not reveal any significant headaches or joint pain. Clinical examination revealed underlying Parkinson’s syndrome with hypomimia, right-sided rigour, compromised posture control and mild cognitive impairment. A Grade I cerebral microangiopathy was identified through brain computed tomography (CT) imaging, and therapy with levodopa was initiated. Persistent productive cough and evident lung obstruction prompted the initiation of prednisone at 40 mg/day for 4 days (HD 12). Subsequent improvement in pulmonary symptoms was observed alongside alleviation of neck stiffness and right-sided headaches. However, upon cessation of prednisone, symptoms recurred.
Laboratory assessments on HD 15 revealed a significantly elevated CRP of 137 mg/L and an increased erythrocyte sedimentation rate (ESR) of 73 mm/h (Table 1). Cerebral magnetic resonance imaging (MRI) ruled out fresh ischaemia, calcium pyrophosphate deposition and spondylodiscitis. Suspecting PMR and GCA, duplex sonography identified partial vasculitis-like findings, notably at the origin of the left internal carotid artery (ICA), leading to 75%–99% stenosis. Moreover, formal findings suspicious of vasculitis were found in the distal common carotid artery of both sides and the left subclavian artery at the level of the lateral clavicle. Overall, the diagnosis of GCA seemed possible based on the duplex sonographic examination. Importantly, the immunological tests, including antinuclear antibodies, antineutrophil cytoplasmic antibodies and anti-double stranded DNA (anti-ds-DNA), were negative. A biopsy of the right temporal artery was conducted, yielding no pathological findings. During the biopsy, a vascular strand measuring 1.2 cm long and 0.2 cm in diameter was removed. A lumen with a diameter of 0.2 cm was visible on the section. However, the presence of possible skip lesions cannot be ruled out. Additionally, MRI angiography of intra- and extracranial vessels revealed a short signal void in the proximal segment of the left ICA, post-carotid bifurcation, without impacting downstream vessel signal, indicative of severe stenosis (Figure 1). Positron emission tomography-computed tomography (PET-CT) confirmed the diagnosis of GCA (Figure 2). Interestingly, the patient did not exhibit typical clinical signs and symptoms of GCA in his personal history, leading us to consider the likelihood of pre-existing GCA/PMR to be unlikely. Moreover, we observed no visual disturbances occurred during the hospital stay. Prednisone was reintroduced at 60 mg/day, resulting in rapid pain regression. Upon discharge, laboratory findings demonstrated decreased CRP (22 mg/L) and reduced ESR (8 mm/h), indicative of treatment efficacy (Table 1). A gradual reduction of the steroid dose was planned after discharge. In addition, we prescribed prophylaxis with trimethoprim-sulfamethoxazole as long as the steroid dose was above 20 mg daily. In the case of visual disturbances (e.g., double vision), we recommended immediate emergency presentation. A follow-up examination at the rheumatology office 4 weeks after discharge was scheduled. However, the patient did not attend the follow-up examination.
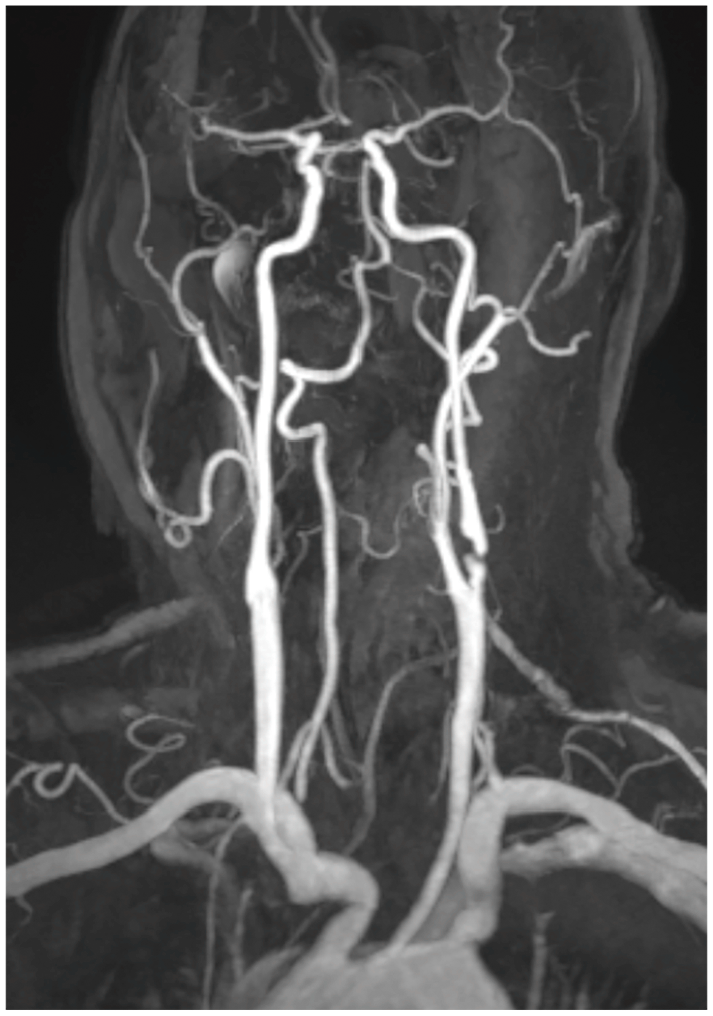
In the MR angiography, short signal gap of the left ICA in the proximal vessel segment shortly after carotid bifurcation, but without impairment of the downstream vessel signal.

PET-CT scan demonstrating the thoracic aorta with a moderate excess of fluorodeoxyglucose uptake, indicating increased metabolic activity, with only mild atheromatosis present (a and b).
Discussion
GCA is characterized by a non-necrotizing granulomatous vasculitis affecting large and medium-sized vessels, particularly the cranial branches originating from the carotid artery.1,3 This condition is relatively common among individuals older than 50 years, with an estimated incidence of 1 per 10,000 person-years.1,4 Initially, there was a prevailing belief that GCA solely affected the temporal artery, leading to the designation of temporal arteritis. However, it became evident that GCA can also affect larger vessels. 5 Indeed, there are two primary patterns of vascular inflammation that share common clinical findings. The first, known as cranial GCA, was initially characterized by Horton in 1932. It entails inflammation of the extracranial branches of the carotid artery, predominantly affecting the temporal artery. The second pattern, termed large-vessel GCA, involves inflammation of the aorta and its proximal branches, including the axillary, subclavian and proximal brachial arteries. 6 PMR is found in up to 50% of patients with GCA and may precede, occur simultaneously with, or follow the clinical manifestations of GCA, 7 indicating a potential continuum or different spectrums of the same disease process between GCA and PMR. Vasculitis triggers luminal occlusion, leading to ischaemic complications like ischaemic optic neuropathy, culminating in vision loss in approximately 10% to 15% of individuals. 8 Additionally, aortitis can lead to complications, including dissection and the formation of aneurysms. 9
GCA, commonly observed in individuals with a genetic predisposition, is often precipitated by various epigenetic and environmental factors. 2 The pathogenesis of GCA is complex, involving intricate interactions between cells of both the innate and adaptive immune systems. These interactions culminate in the formation of granulomas, marked by the presence of giant cells, a defining characteristic of the disease, alongside arterial tertiary follicular organs. 2 Furthermore, cells within the vessel wall, including vascular smooth muscle cells and endothelial cells, actively engage in vascular remodelling processes. These processes are pivotal in the development of vascular stenosis and the onset of ischaemic complications associated with GCA. 2
Although the aetiology of GCA remains elusive, there is speculation that environmental factors and infectious agents may contribute to its pathogenesis. GCA is considered an antigen-driven disease; however, the specific autoantigens or antigens responsible have not yet been definitively identified. Fluctuations in GCA incidence and the presence of an epidemic-like cyclical pattern have prompted an investigation into the potential involvement of specific infectious agents as triggers. 10 As mentioned above, research has concentrated on the involvement of cellular components in the elusive pathophysiology of GCA. Essentially, activation of dendritic cells in the adventitia is initiated by an unidentified antigen within the vascular wall. This activation subsequently leads to the recruitment and activation of CD4 T cells and macrophages, culminating in vascular inflammation and remodelling. Like with other forms of vasculitis, there is speculation that GCA may be triggered by an antigen of infectious origin interacting with specific genetic predispositions in susceptible individuals. 11 Despite that numerous infectious agents, including viral and fungal agents, or microbial antigens, have been reported in the literature in association with vasculitis, a definitive causal relationship has only been firmly established in specific instances of vasculitis. For example, chronic hepatitis-B-virus-associated polyarteritis nodosa and hepatitis-C-virus-related cryoglobulinaemic vasculitis are among the few cases where such a relationship has been clearly established. These entities are classified as ‘vasculitis associated with probable etiology’ according to the 2012 Chapel Hill Consensus Conference on the Nomenclature of Vasculitides. 12 Regarding GCA, certain reports have underscored the potential role of specific bacteria and viruses, like cytomegalovirus, parvovirus B19 or varicella-zoster, along with Mycoplasma pneumoniae, Chlamydia pneumoniae and influenza vaccines in precipitating GCA in individuals predisposed to the condition.10,13
Influenza vaccination has been associated with GCA and/or PMR onset. Research suggests that up to 3% of newly diagnosed cases of GCA may be triggered by receiving a seasonal influenza vaccination. Furthermore, the HLA DRB1*13:01 genotype has been proposed as a potential factor implicated in the development of post-influenza vaccination GCA. Unlike PMR, GCA can represent a serious complication following influenza vaccination. Both GCA and PMR may be considered as part of the autoimmune/inflammatory syndrome induced by adjuvants, highlighting the complex interplay between vaccination and autoimmune reactions in susceptible individuals. 14
Influenza infection as a trigger for GCA is, in our opinion, considered exceptionally rare, with conflicting data in the literature. To date, to the best of our knowledge, only one study has provided evidence of a potential association between influenza infection and GCA. In this study, which included 1005 patients with biopsy-confirmed GCA and 10,050 controls, infections were found to be more prevalent among patients subsequently diagnosed with GCA compared to controls. Specifically, acute upper respiratory tract infection, influenza and pneumonia, as well as unspecified infections, were associated with GCA. However, no correlation was observed between GCA and skin or gastrointestinal infections. 10 Conversely, earlier investigations have indicated that the seroprevalences of both A and B influenza viruses were comparable between cases and controls. 15
Conclusion
Our case suggest a potential association between influenza infection and GCA; however, further research is needed to establish a definitive causal relationship. Moreover, the absence of our patient at the scheduled follow-up appointment poses a limitation in assessing long-term outcomes and continuity of care in this case study.
Footnotes
Acknowledgements
We would like to thank Dr. med. Helmut Rasch for his assistance in selecting and describing the images.
Author contributions
The study was designed by M.Z. D.K.M., P.P. and M.Z. were involved in the diagnosis and management. D.K.M. and P.P. searched the articles and drafted the manuscript, to which M.Z. contributed and revised. All authors read and approved the final manuscript.
Data availability
Data sharing is not applicable to this article as no data sets were generated or analysed during the present study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
The project did not meet the definition of human subject research.
Informed consent
Written informed consent was obtained from the patient for his anonymized information to be published in this article.
