Abstract
Mature cystic teratoma of the ovary (MCTO) is a type of common disease in clinic. However, the malignant transformation of MCTO occurred rarely, with many unexplained questions. To the best of our knowledge, owing to the rarity of squamous cell carcinoma (SCC) in MCTO, even though there are some previous research works concerning about this rare disease, further exploration and discussion is still necessary to reveal unknown aspects. We present a case of pathologically confirmed SCC in MCTO in a 54-year-old female patient, who underwent surgery and subsequent adjuvant chemotherapy. After the treatment, she recovered well and was followed up until now. SCC in MCTO occurred rarely; clinicians should abandon habitual concepts and make targeted management individually.
Introduction
Mature cystic teratoma of the ovary (MCTO) is the most common ovarian germ cell tumor.1,2 The malignant transformation occurrence of MCTO is 0.17%–2%,1–4 with the prominent subtype of malignant transformation being squamous cell carcinoma (SCC).1–4 The precise diagnosis of ovarian tumor pre-operation is difficult, so is the MCTO. Although the ultrasound and imaging radiology such as computed tomography (CT) and magnetic resonance imaging (MRI) are widely used in clinic, with laboratory tests just like cancer tumor biomarkers accompanied, it still proves to be difficult to distinguish benign tumors from malignant ones. Furthermore, the main symptoms of MCTO are short of specificity and sensitivity. Thus, MCTO is diagnosed by pathological exam terminally. Also, owing to the rarity of SCC-MCTO, there is no standard and unified management scheme for its treatment and survival, despite that there exist some consensus and researches previously.1–4 Here, we present a case of pathologically confirmed to be SCC arising from MCTO in a 54-year-old female.
Case presentation
The patient was a 54-year-old postmenopause woman, who complained of intermittent lower abdominal pain for almost 2 months. She was suspected to have a left adnexal tumor after pelvic-vaginal physical examination. Vaginal ultrasonography implied a mass sized 7.8 × 8.1 × 5.4 cm and situated in the left pelvic cavity, containing irregular high echo and non-pure liquid dark area (Figure 1(b) and (c)). Subsequent abdominopelvic CT scan confirmed this, showing that there was a large cystic mass (78 × 63 mm) located in the left adnexal area (Figure 1(a)). The mass contained lipid contents and displayed slightly higher ultrasonic echo, with the cyst wall calcified. She reported no family history of tumor, and the elementary physical examination, including blood pressure, pulse rate, and temperature, was normal. Of course, the patient underwent specialized gynecological examination, including cervical cancer screening, after admission to hospital. The cervix was atrophic but without bleeding and neoplasm. The results of routine hematologic and urinary tests were insignificant. However, the serum level of cancer antigen 199 (CA199) was exceedingly elevated, showing as >1200.0 U/ml (normal <37 U/ml), whereas other tumor biomarkers, such as cancer antigen 125 (CA125), alpha fetoprotein, carcinoembryonic antigen, were negative.

Imageological examinations. (a) Enhanced CT scan showed a large mass with cyst wall calcified in left pelvic area; (b) ultrasound exam showed the calcified site of the cyst wall; (c) ultrasound exam showed the cyst.
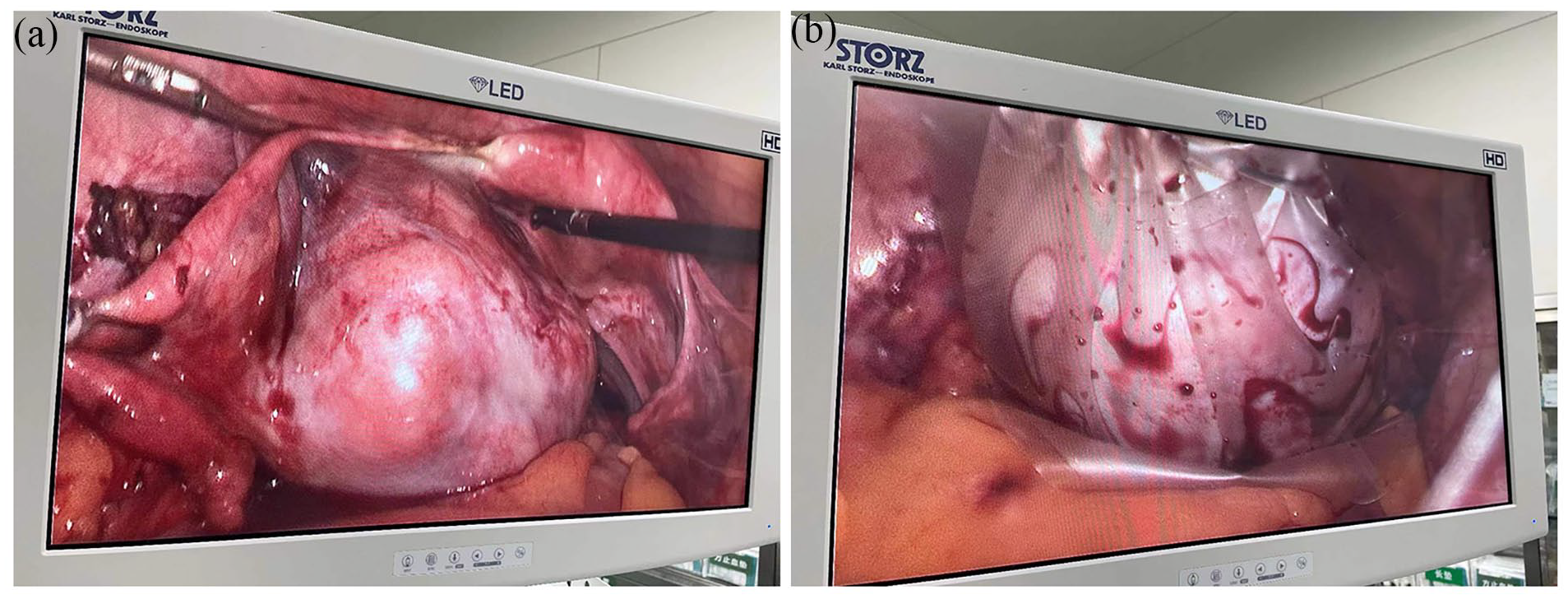
After obtaining written informed consent, the patient underwent exploratory laparoscopic abdominal surgery. During the surgery, on gross examination, as shown in Figure 2, we found that the surface of bilateral annex was smooth, and both ovaries were complete without rupture. The left adnexa were densely adherent to the pelvic wall, and the left ovary was enlargement containing a mass measuring about 8.0 × 8.0 cm. The uterus was atrophic but even. The left fallopian tube, the right adnexa, appendix, omentum, pelvic wall, peritoneum, liver, diaphragm, and bladder were free of tumor. There was no ascites fluid for cytological examination. The left adnexa were resected completely with special specimen bag (HSD-80; TKBAG, Guangzhou, China, www.TK-MEDICAL.com) packaged, and we took the bag out from the laparoscopic incision. Then, the specimen was cut open for the first and quick check and sent to fast frozen pathologic analysis.

Images of tumor. (a) Gross examination of tumor pre-operation; (b) the tumor was resected and packed in special specimen bag.
The fast frozen pathologic examination read that the cystic mass had been broken macroscopically, and it measured 7 cm × 5 cm × 1 cm with 2 cm × 1.5 cm × 1 cm solid cephalic segment inside. Moreover, greasy hair was observed in the cyst and the attached left fallopian tube appeared usually. Microscopically, there was malignant transformation of mature cystic teratoma, and squamous cells were observed, as shown in Figure 3. Finally, it concluded pathologically that it was SCC of mature cystic teratoma in the left ovary, and the left fallopian tube was free of tumor. Then, we performed a laparoscopic total hysterectomy and omentectomy with right annex resected. We defined the ovarian cancer as stage 1a according to the International Federation of Gynecology and Obstetrics guideline.
Representative histological images. (a) The segmental area was covered with squamous epithelial cancer, and infiltrated into the interstitial (HE40). (b) In the stroma, the cells of SCC grew in a nest-like infiltrate pattern. The nuclei were large and deeply stained, the nuclear division was observed, the atypia was obvious, and the intercellular bridge was visible, suggesting squamous cell differentiation (HE100).
The patient discharged 1 week after the operation with no complications. Three weeks after her discharge, she returned back to hospital and received adjuvant chemotherapy with paclitaxel—carboplatin scheme, which demonstrated paclitaxel 175 mg/m2 intravenously over 3 h and carboplatin 450 mg intravenous drip. The dose of the chemotherapeutic drugs was calculated according to her weight and height. Each time before the chemotherapy, she received examination of blood hematology analysis, electrocardiogram, liver, and kidney function test. Assessments included, but were not limited to these check items. She underwent six courses of standard adjuvant chemotherapy after the surgery.
The patient was followed up after the completion of the chemotherapy until now. Fortunately, there was no recurrence detected through routine exam such as chest radiology, abdominal-pelvic CT scan, and so on.
Discussion
MCTO is quite common in clinic, accounting for nearly 20% of all ovarian cancers. It often occurs in young women during reproductive age.2,5,6 The condition of malignant transformation in MCTO is rare and often observed in postmenopausal women,2,5 with the average age ranging from 33 to 56 years old. 7 The most common pathological type of malignant transformation in MCTO is SCC, followed by adenocarcinoma, sarcoma, and other subtypes.5,8 The clinical characteristics of the patient in our case were accordance with these studies.
The etiology of SCC in MCTO remains unclear. Iwasa et al. found that dysplasia of squamous cells was the cause of the malignance. 9 However, there are also other researches demonstrating that human papillomavirus (HPV) infection was pathogenic factor. 10 In our case, the HPV test and Thinprep Cytologic Test of the patient were negative; combining with vaginal examination, we could exclude cervical cancer metastasis. Otherwise, other studies reported that genetic factors also associated with the malignance.11,12 Therefore, the role of HPV in SCC arising from MCTO remains uncertain. The mechanism of this disease is complex and still needs to further investigated.
The clinical manifestations of malignance of MCTO are nonspecific and variable. 3 In the case we presented, the patient displayed chronic abdominal pain for several months. In fact, many patients were asymptomatic when diagnosed incidentally. 13 With the progress of the disease, some other patients presented with abdominal pain and distention. Some patients also complained of nausea, vomiting, and abdominal pressure. 13 Furthermore, ovarian torsion or rupture presenting as a gynecological emergency was also reported.12,14,15
Owing to the atypical clinical symptoms of malignant transformation of MCTO, the definite pre-operative diagnosis is difficult. As mentioned above, many patients showed no discomfort before examination. Physical examination and imageological examination, such as ultrasound, CT scan, or MRI, may display pelvic mass. However, all the results of checks before surgery were just raised suspicion of ovarian tumor; the final result must be confirmed by the pathology.
Because of the rarity of SCC of MCTO, the management methods of this disease are puzzle and controversial. Most patients diagnosed with SCC-MCTO underwent surgical treatments and adjuvant chemotherapy subsequently. It was reported that radiology therapy displayed no benefit to the prognosis.2,3 Conservative surgery was conducted when young patients who required to reserve fertility ability, without tumor invasion or metastasis. Otherwise, concerning about old patients who were postmenopause, total debulking surgery was performed, including hysterectomy, omentectomy, and bilateral adnexectomy. After surgery, to improve the prognosis and enhance the survival rate, the chemotherapy was on schedule. Although there were alternative options, paclitaxel–carboplatin scheme was commonly used in clinic. 16 With the development of genomic analysis and gene test of the disease, the immune checkpoint inhibitor, poly ADP-ribose polymerase (PARP) inhibitors, and targeted drugs demonstrated wide prospect in the therapy.1,2,12,17 Despite alternative managements, the prognosis of SCC-MCTO remains poor. Gadducci et al. reported that 5-year overall survival rate ranged from 28% to 66.7%, 16 whereas Li et al. revealed that the rate was 85.8%, 39.1%, 26.2%, and 0 for stages I, II, III, and IV, respectively. 11 Although there was no consensus of prognosis, many studies revealed that elder age, tumor size, tumor stage, and serum level of tumor biomarkers were prognostic factors.3,6,18,19 For instance, some studies indicated that the fact tumor size ⩾10 cm was a predicator of malignant transformation. However, in our case, the maximal diameter was only 8.1 cm, which renewed our perception.
In our case, some points were alike with other reviews, such as the examinations after admission, general treatment program with adjuvant chemotherapy after operation. Of course, we ourselves also realized that the chemotherapy regimens existed difference. Some patients underwent platinum-based therapy, whereas some others received Alkylating drugs or combination of bleomycin, etoposide, and Platinol plan. Until now, there is still no standard management program all over the world about SCC-MCTO, even though previous clinical practices exhibited different therapy protocols. However, with the understanding of this disease, the blank with be fulfilled in the not far future. Despite that the only case presented is provided only by a single institution, it will be helpful for all of us to understand this disease.
Conclusion
Overall, although SCC in MCTO occurred rarely, many related fields, including carcinogenesis, progression, and prognosis and so on, still called for further discovered.
Footnotes
Acknowledgements
Not applicable.
Author contributions
G.X. and C.X. carried out the study, collected the case, and written the manuscript. L.Y. analyzed and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case was obtained from Institutional Review Board of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
