Abstract
Management of sickle cell disease complications in the setting of the coronavirus disease 2019 (COVID-19) pandemic is complicated with little published pediatric data. We report the first documented case of a 9-year-old boy with sickle cell disease, presenting with fever, cough, and shortness of breath, diagnosed to have acute chest syndrome and coronavirus disease 2019 (COVID-19) pneumonia with inflammatory storm requiring ventilation, exchange blood transfusion, immunomodulatory agents, and prophylactic anticoagulation. The patient responded satisfactorily to the management of the acute illness and was found to be well at the next visit to the pediatric hematology outpatient department following hospital discharge.
Keywords
Introduction
As the coronavirus disease 2019 (COVID-19) pandemic continues to wax and wane, the impact of patient’s comorbidities on disease presentation and clinical course has generated considerable interest. Patients with sickle cell disease (SCD) who are infected with COVID-19 may have a significant risk of developing acute chest syndrome (ACS), a potentially life-threatening complication. To date, SARS-CoV-2 infection has been reported to cause ACS mostly in adult patients with SCD. 1 Viral infections are well-established triggers to the acute manifestations of SCD1,2 and infection with the new coronavirus might follow the same course.
Case presentation
A 9-year-old boy, known to have SCD and on maintenance therapy of folic acid and oral antibiotic prophylaxis (phenoxymethylpenicillin), presented to our emergency room (ER) during the early phase of the pandemic with complaints of high-grade fever (39°C), cough, shortness of breath, and chest pain for 2 days. He had visited the ER in the preceding week for similar complaints and was discharged home on oral antibiotics. As the visual triage score for acute respiratory infection was high, he was admitted to the isolation ward with a provisional diagnosis of SCD with COVID-19 pneumonia. A nasopharyngeal swab for COVID-19 was performed along with a collection of blood and urine cultures and an empirical course of azithromycin and Co-Trimoxazole started (the patient being allergic to ceftriaxone). Chest X-ray (CXR) showed infiltrates in both lower lung fields (Figure 1). Twenty-four hours later, his COVID-19 RT-PCR test result was reported to be positive, and hydroxychloroquine was prescribed.
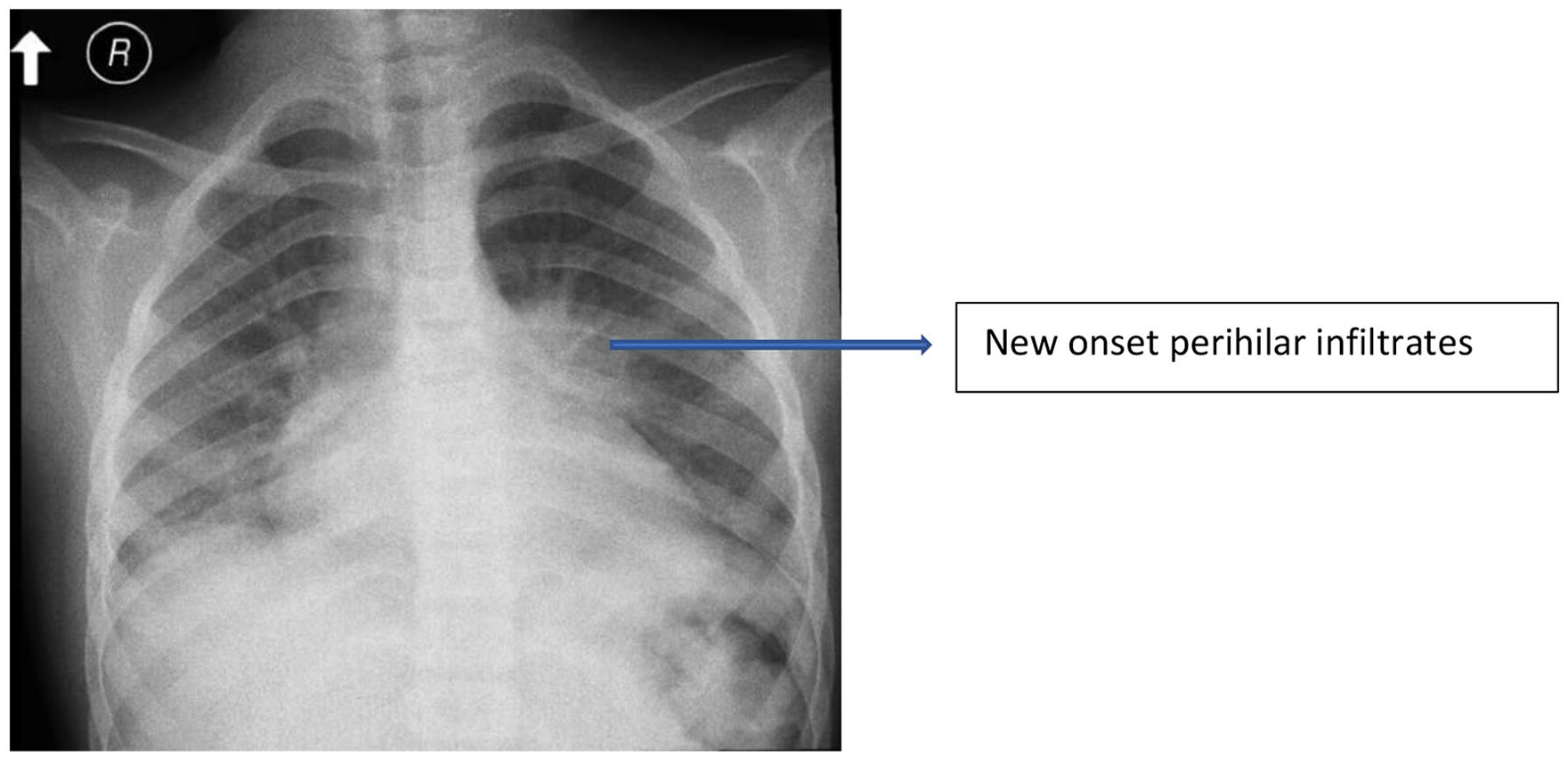
Initial CXR showing infiltrates in both lower lobes.
A review of his past medical history showed that he was diagnosed with SCD at the age of 4 months and had multiple admissions since then for acute respiratory infections and splenic sequestration, ultimately requiring splenectomy when he was 3 years old. He had been previously admitted to the pediatric intensive care unit (PICU) once before with pneumonia but he did not require any assisted respiratory support.
The patient was conscious upon admission, with a high-grade fever of 39°C and although tachypneic with a respiratory rate (RR) of 35/min, his peripheral oxygen (O2) saturation was maintained at 96% on 2 L/min O2 via nasal cannula. The respiratory examination revealed bilateral equal air entry with crackles in both lower lung fields.
Hospital course
The patient’s condition deteriorated the next day with increased work of breathing and progressive hypoxemia on 10 L/min O2, so he was admitted to the PICU and was intubated due to type 2 respiratory failure. Chest X-ray was repeated (Figure 2) which showed an ill-defined large left lung opacity. Antibiotic therapy was switched to levofloxacin and vancomycin. The patient was continued on hydroxychloroquine as per national COVID-19 guidelines. A simple blood transfusion was initiated for ACS as the patient’s hemoglobin (Hb) was 6.1 g/dL, which then increased the Hb to 8.4 g/dL. Chest computed tomography scan (CT scan) could not be performed at the time due to the patient’s unstable condition. Arrangements for urgent automated exchange transfusion were begun.

CXR on day 2 demonstrates a large left lung opacity.
His condition worsened the next day, requiring inotropic support. Intravenous immunoglobulin (IVIG) and methylprednisolone were started to control the inflammatory process. Meanwhile, the patient developed mild renal impairment as S. Creatinine rose to 78 umol/L, but his urine output was still adequate (>1 ml/kg/h). An automated exchange transfusion was initiated. HbS testing was not immediately available prior to the exchange transfusion, but the HbS during his previous admission was 55%. Inflammatory markers were found to be elevated (D-Dimer: 4.4 mg/l, CRP: 178 mg/dl, S. Ferritin: 367.8 mg/ml) and the patient was started on prophylactic anticoagulation due to the prothrombotic nature of both SCD and COVID-19. Post-blood exchange transfusion Hb electrophoresis revealed HbS of 16.5%.
The patient’s condition started improving gradually on day 4 of the PICU stay and inotropes could be tapered off. Improvement was evident in serial CXR (Figure 3). Blood gases had begun to stabilize.

CXR on day 3 shows significant improvement in aeration.
By day 4 of PICU admission, the child was in stable condition; CXR had further improved with only a small irregular opacity in the left middle lung field (Figure 4). The patient was gradually weaned off ventilation and extubated successfully the next day. Blood, urine, and tracheal aspirate cultures were negative. The patient was continued on levofloxacin and vancomycin for 7 days as advised by the Infectious diseases team due to the critical condition of the patient. The total length of hospital stay was 11 days, whereas the length of PICU stay was 7 days, of which the patient spent 4 days on ventilatory support. A summary of the patient’s PICU course including the relevant investigations is provided in Table 1.

CXR on day 4: nearly normalized with a small remnant opacity in the left middle lung field.
Summary of PICU course including relevant investigations.

NRM: non rebreathing mask; PICU: pediatric intensive care unit; PRVC: pressure regulated volume control.
Discussion
The etiology of ACS in children with SCD is often multifactorial, combining increased adhesion of sickle red cells to pulmonary microvasculature, pulmonary fat embolism, infarction, and infection, with an excessive inflammatory lung injury response in the presence of a damaged lung microvasculature. 3 The recurrent sickling process in SCD causes tissue hypoxemia and micro-infarcts, resulting in end organ damage. Infections are particularly common in children and a frequent cause of ACS. 4
The most frequently involved infectious agents are bacteria such as
Besides, monocyte activation has been reported in SCD and is responsible for enhanced production of pro-inflammatory cytokines such as IL-6, IL-1β, and TNF-α, which contributes to vaso-occlusion by promoting endothelial activation. Many cytokines are elevated during steady-state in SCD, and some of them, especially IL-6, are further increased during vaso-occlusive crises.
Data are still emerging on how the pandemic may complicate SCD in children. Pediatric studies have not demonstrated clear correlations between SCD and COVID-19. A prospective multicentric registry in Brazil studied 25 children known to have SCD, who concurrently developed COVID-19, and found that three of them required ICU care of whom only one patient developed ACS (managed conservatively); but all three were noted to be obese. 9
Martin et al report the result of a 1-year review of 55 pediatric SCD patients with COVID-19 and found that among the 47% (
Both ACS and COVID-19 pneumonia may present with similar features. The pro-inflammatory state of SCD may be augmented further by the hyper-inflammatory state caused by the COVID-19 infection. In such patients, we recommend prompt admission to the intensive care unit with initiation of blood exchange transfusion, along with invasive or noninvasive ventilatory support as needed, in addition to treatment of the COVID-19 pneumonia and the impending cytokine storm to prevent life-threatening complications like ACS. Although hydroxychloroquine was administered to our patient based on evidence available in the early phase of the pandemic showing in vitro antiviral effects hypothesized to decrease the SARS-Co-V2 viral load, 12 subsequent systematic reviews and meta-analyses have failed to demonstrate a clear benefit. 13 We have not discussed in detail regarding cytokine storm/multisystem inflammatory syndrome in children (MIS-C) as our patient did not manifest the typical features of the condition, but administration of IVIG, high dose of IV mehylprednisolone pulse therapy, and use of biologic agents such as anakinra in severe cases are implemented as standard regimens for its management. 14 MIS-C has been reported to occur in a few pediatric SCD patients, 15 including a fully vaccinated child, 16 but they were all successfully managed with the aforementioned therapy. Prophylactic anticoagulation with low molecular weight heparin and/or aspirin 17 may be required as SCD and COVID-19 are both thrombogenic states.
Although the child was found to be well at the next visit to the pediatric hematology outpatient department (OPD) following discharge, unfortunately, our patient was lost to follow up subsequently, so we were unable to trace his long-term progress making it a limitation to our report. Since data on patients with SCD infected with the new coronavirus are scarce, especially in the pediatric group, it is necessary to obtain more information to reduce the uncertainties still present in medical practice. 18
Conclusion
As patients with SCD generally have a higher tendency to develop ACS and respiratory complications when triggered by viral infections, it is possible that they are also at higher risk of such complications from COVID-19. This is the first reported case of ACS in a pediatric patient with SCD infected with SARS-CoV-2 virus which required exchange transfusion and mechanical ventilation. Hence, our case report gives some insight as to how the SARS-CoV-2 pandemic may lead to increased rates of ACS in sickle cell patients. This case report highlights that all hospitalized sickle cell patients should be monitored closely for the development of ACS and that if this occurs, exchange transfusion and other supportive treatment should be promptly initiated to achieve favorable outcomes.
Footnotes
Acknowledgements
We would like to acknowledge the pediatric hematology/oncology department and the PICU staff of King Saud Medical City, Riyadh, Saudi Arabia.
Authors’ note
The first draft of the case report was written by the co-author Dr Mohammad Raza. No honorarium, grant, or other form of payment was given to anyone to produce the manuscript.
Author contributions
All authors contributed equally to writing the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case was obtained from the King Saud Medical City Institutional Review Board with reference number: H2RI-30-Oct22-02.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
