Abstract
Objectives:
Treatment options for preventing vaso-occlusive crises among sickle cell disease patients are on the rise, especially if hydroxyurea treatment has failed. This economic analysis is conducted to assess the comparative clinical effectiveness, safety, and acquisition cost of
Methods:
We conduct a decision-tree model, where we compare the clinical and economic outcomes of two novel Food and drug administration (FDA)-approved medications which are available in Qatar;
Results:
Study results showed that both treatment modalities’ costs were the main driver of this analysis, with an average annual cost of the treatments per patient being $189,014 for crizanlizumab (5 mg/kg), $143,798 for crizanlizumab (2.5 mg/kg), and $74,323 for
Conclusions:
Crizanlizumab (2.5 mg/kg) may be a cost-effective intervention, yet it is not the approved dose for preventing vaso-occlusive crises in adolescents and adults with sickle cell disease. Crizanlizumab (5 mg/kg) was more cost-effective than the approved
Introduction
Sickle cell disease (SCD) is a hereditary disease that is caused by autosomal recessive gene fault in the beta (β) allele of the hemoglobin (Hb) gene. As a result, sickled cells are characterized by easy and abnormal hemolysis with resultant varying degrees of anemia. 1 Globally, the incidence of SCD is estimated to reach 400,000 persons per year, and in the United States alone, for example, the prevalence estimation is approximately 100,000 patients. 2 Possible clinical presentations of SCD may come from different pathophysiologic mechanisms; the disfiguration of the RBC with subsequent loss of function can lead to vascular occlusion and a short lifetime of these RBCs that leads to hemolysis. The consequence of function loss is a vascular blockage and can cause vascular lesions. 3 The most severe and serious manifestation of SCD is the recurrent acute pain, or better known as vaso-occlusive crisis (VOC).4,5 Additionally, other clinical manifestations that SCD patients may show are acute complications such as acute chest syndrome (ACS), recurrent infections, kidney necrosis, and stroke. 6 Such complications may affect multiple organs and can result in early death. 6 Acute pain crisis is another common complication of SCD and is usually managed with pain medications, especially opioids.6,7
In terms of SCD management, there is no universal curative treatment for SCD. Nonetheless, few medications are available for its symptoms and complication management. Of these medications in the market, hydroxyurea was the first approved medication for SCD management.
8
Later, other drugs were approved and are indicated for the prevention of VOC, namely
In relation to treatment costs, SCD management costs are high, with an estimated economic burden of $2.98 billion per year in the United States, with approximately 57% due to inpatient-based costs, 38% incurred by outpatient-based costs, and a remaining 5% as an out-of-pocket cost. 14 Furthermore, the minimum treatment options, added to the stigma caused by the continuous need for pain management, make coping with SCD problematic. 15
Of the drugs mentioned, two new therapies have become available for patients with SCD in the state of Qatar,
Methods
Model structure
Clinical data were abstracted from landmark randomized controlled trials (RCTs), based on which drugs were granted approval. Additionally, treatment modalities included in data analysis has input from sickle cell treatment guidelines at the National Center for Cancer Care and Research (NCCCR) in Qatar to reflect the current practice in the country.
A conventional decision-tree model was structured to generate the clinical pathways followed by SCD patients (Figure 1). The model alternatives were three possible treatment strategies: crizanlizumab (5 mg/kg), crizanlizumab (2.5 mg/kg), and

A schematic representation of the decision analysis model of study drugs.
Patients were initially differentiated based on success and failure. Success is to have no pain crisis, with/without adverse events (AEs). Failure is to develop pain crisis, ACS, or death. After the first pain crisis, a second may also develop. After the second pain, an additional pain crisis may develop. Second crisis is defined as the development of a second pain incident following a first pain episode. Based on the RCT sources of data, the duration of the model follow-up is 1 year. Figure 1 presents the decision-tree structure of study comparators.
Success is defined as having no pain crisis, death, ACS, or serious AEs. According to RCTs, a pain crisis is defined as pain resulting in receiving a parenterally administered narcotic or ketorolac in an emergency department or outpatient treatment center, or during hospitalization. Pain begins suddenly and lasts several hours to several days, can be mild to severe, and can last for any length of time.16,17 Death is defined as all-cause death. An AE is defined as any undesirable effect that is probably associated with the use of a medication in a patient. 18 A serious AE, on the other hand, is defined as AE that may result in disability, hospitalization, or death. 18 The ACS is defined as life-threatening and should be treated in a hospital. Signs and symptoms include chest pain, coughing, difficulty breathing, and fever.17,18 An expert panel of hematology consultants who are based at the NCCCR validated the structure of the model and its consequences.
Sample size calculation
Unlike clinical trials, where sample size calculations are essential to ensure sufficient statistical power for detecting differences in primary endpoints, in our cost-effectiveness analysis, we simulate the progression of disease, treatment effects, and associated costs over time. These models allow for the exploration of various scenarios and assumptions, rendering traditional sample size calculations less applicable.
Clinical inputs
Model probability inputs in relevance to events associated with crizanlizumab 5 mg and crizanlizumab 2.5 mg, including pain crises averted, AEs, ACS, and death were primarily obtained from the SUSTAIN trial by Ataga et al. 21 Model clinical events, their probabilities, and sources of data can be seen in Table 1.
Clinical outcomes probabilities of different modalities.

The SUSTAIN trial is a phase 2, multicenter, randomized, placebo-controlled, double-blinded trial to assess the safety and efficacy of crizanlizumab (2.5 and 5 mg, administered 14 times, intravenously, over 1 year) with or without hydroxyurea in SCD patients—ClinicalTrials.gov Identifier: NCT03814746.
21
The Niihara et al. study is a phase 3, multicenter, randomized, placebo-controlled, double-blinded, to evaluate the efficacy of
Study perspective
The study was conducted from the NCCCR hospital’s perspective. The NCCCR is one of the hospitals under Hamad Medical Corporation (HMC), the main healthcare provider in Qatar, including 13 major specialized public hospitals.
Cost inputs
Direct medical costs of resources consumed in the management of SCD were calculated (based on 2020–2021 prices). The cost data were obtained from the Finance and Costing Department of HMC, and included costs of medications acquisition by dispensing pharmacy, hospitalization, managing serious AEs, laboratory, and screening tests (Table 2). The medical care component of Qatar’s Consumer Price Index was used for cost inflation. All costs were expressed in 2021 Qatari Riyal (QAR) and then were converted to United States Dollar (USD). No discounting was applied as outcomes were not projected beyond a 1-year time horizon.
Total cost of resources per patient.

Outcome measurement
The outcome of the study was the incremental cost-effectiveness ratio (ICER) in terms of QAR per additional case of pain crisis avoided. The outcome endpoint was basically the annual rate of pain crises, defined as the development of acute incidents of pain that resulted in either an urgent medical visit to the hospital or treatment with oral or parenteral narcotic agents or with a parenteral nonsteroidal anti-inflammatory drug (NSAID). Of note, no crisis means that patients finish the 1-year study with no pain episodes. Cost-effectiveness was determined based on a willingness-to-pay threshold of USD 150,000 (547,500 QAR) per outcome.
Sensitivity analysis
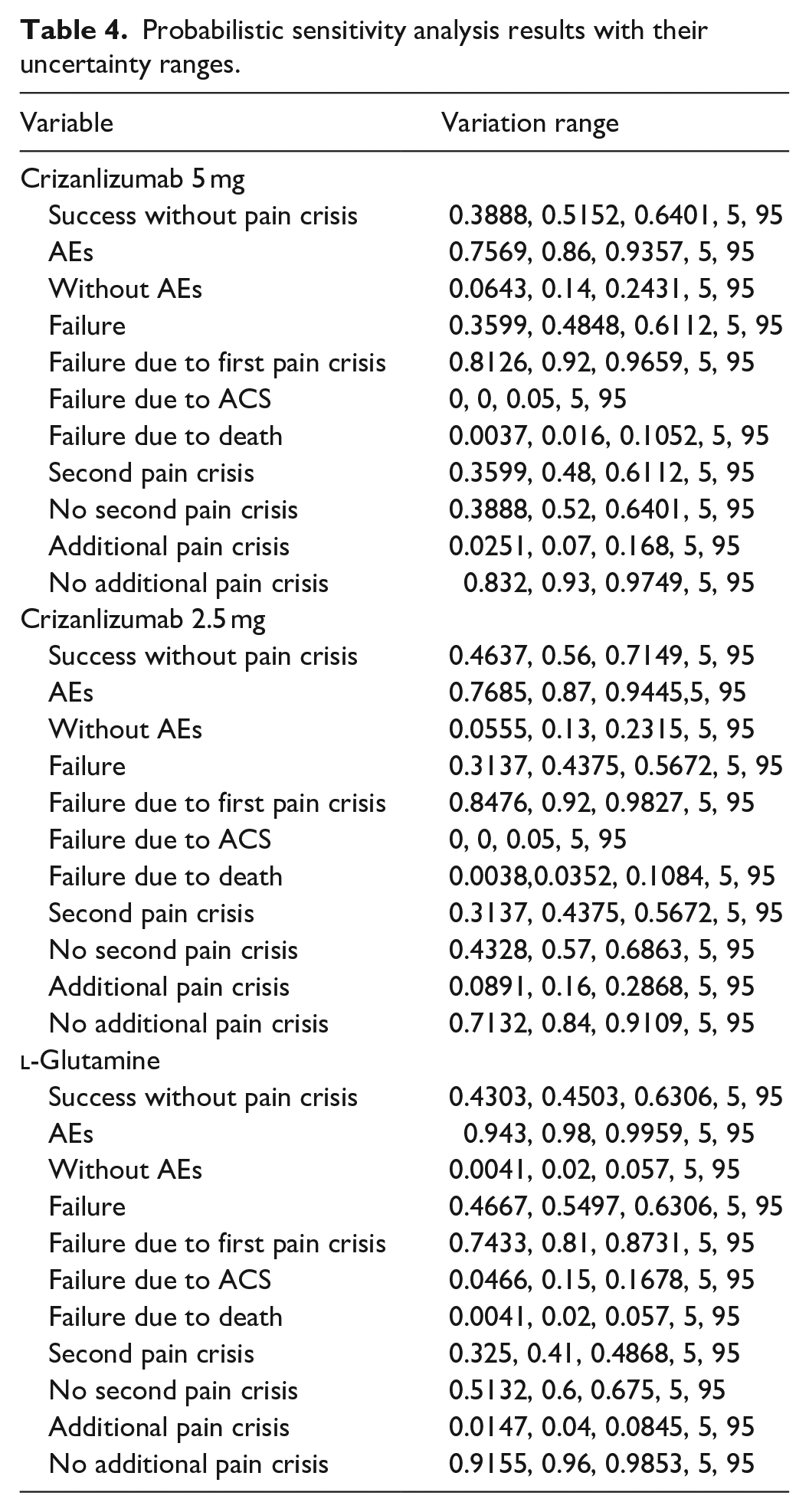
A one-way sensitivity analysis was first conducted by assigning a ±15% uncertainty range to the cost of medications, using a triangular type of distribution. A probabilistic sensitivity analysis was conducted by introducing uncertainty to the base-case clinical events. A ±95% confidence interval (CI) uncertainty range of the base-case value was applied to clinical events using Trigen distribution. All sensitivity analyses were performed via the Monte Carlo simulation approach using @Risk-7.5® (Palisade Corporation, Ithaca, NY, USA), with 5000 iterations. 24
Results
Base-case analysis
The 5 mg crizanlizumab achieved a success rate of pain crisis averted of 0.5152 compared to 0.4503 with
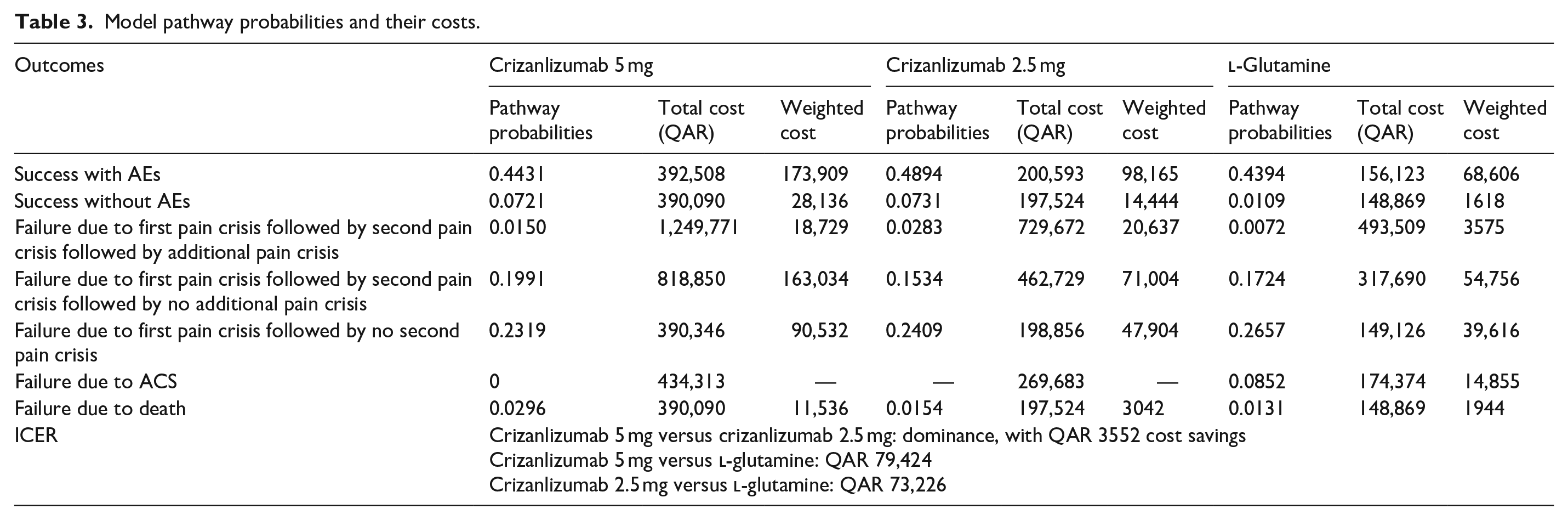
Model pathway probabilities and their costs are as seen in Tables 3 and 4.
Model pathway probabilities and their costs.
Probabilistic sensitivity analysis results with their uncertainty ranges.
Sensitivity analysis results
One-way sensitivity analysis
Crizanlizumab 5 mg versus l -glutamine
Cost inputs in one-way sensitivity analysis, and their uncertainty distributions, are presented in Table 5. The model was insensitive to changes in all cases.
One-way sensitivity analysis on cost input, distributions—crizanlizumab 5 mg versus

Crizanlizumab 2.5 mg versus l -glutamine
Cost inputs in one-way sensitivity analysis, and their uncertainty distributions, are presented in Table 6. The model was insensitive to changes in all cases.
One-way sensitivity analysis on cost input, distributions—crizanlizumab 2.5 mg versus

Crizanlizumab 5 mg versus crizanlizumab 2.5 mg
Cost inputs in one-way sensitivity analysis, and their uncertainty distributions, are presented in Table 7. The model was insensitive to changes in all cases.
One-way sensitivity analysis on cost input, distributions—crizanlizumab 5 mg versus crizanlizumab 2.5 mg.

Probabilistic sensitivity analysis
Model cost inputs and their plausible ranges are presented in Table 6. The ICER probability curve showed that crizanlizumab 5 mg was cost-effective in nearly 90% of simulated cases and was dominant in less than 10% of the cases (Figure 2).

ICER acceptability curve of crizanlizumab 5 mg.
Based on the multivariate uncertainty analysis, a tornado regression analysis of the different study outcomes revealed that success with AEs with crizanlizumab 5 mg was the input that had the main effect on the outcome, while success without AEs with
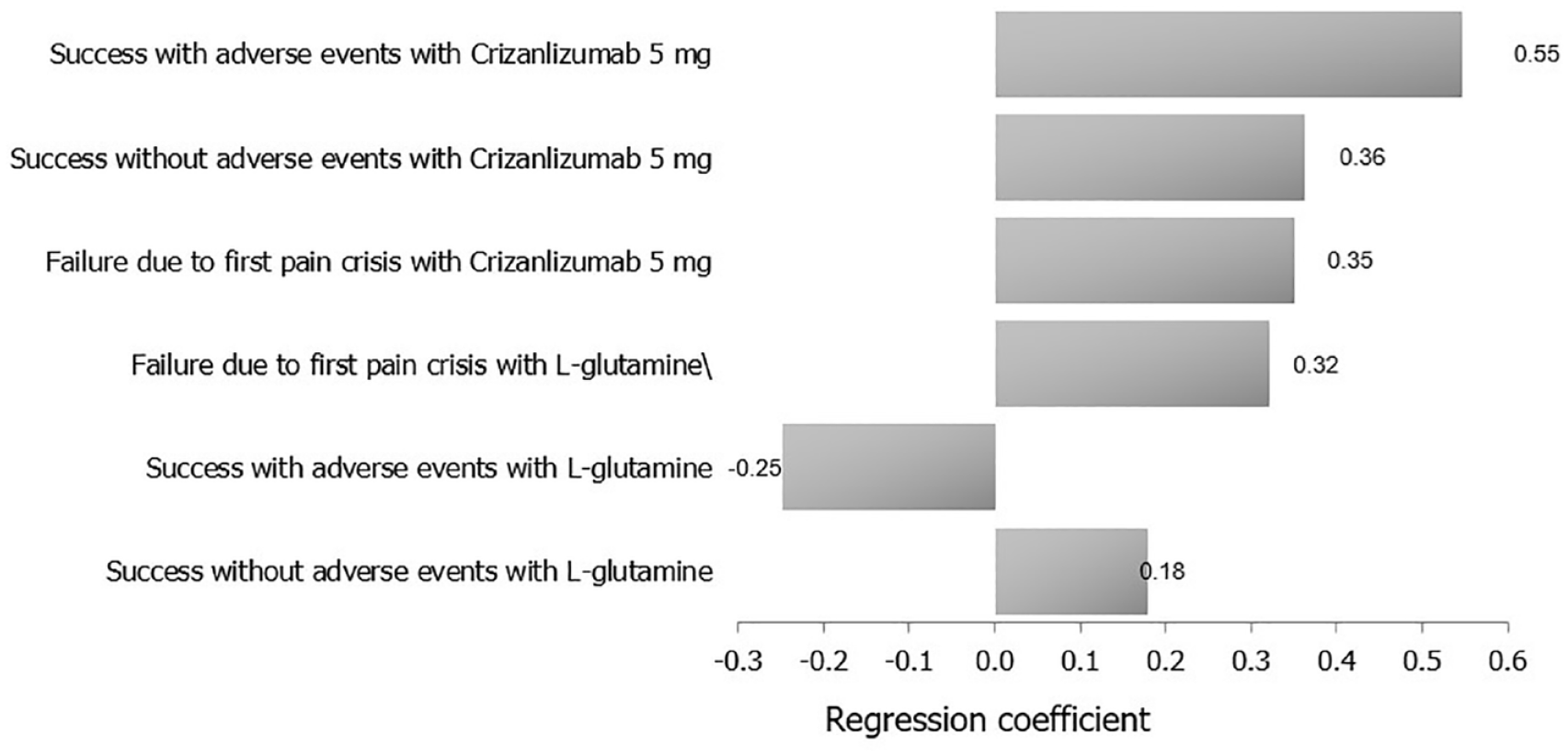
A tornado analysis of the study’s clinical outcomes and their costs on ICER (crizanlizumab 5 mg versus
When comparing crizanlizumab 2.5 mg versus

ICER acceptability curve of crizanlizumab 2.5 mg.

A tornado analysis of the study’s clinical outcomes and their costs on ICER (crizanlizumab 2.5 mg versus
Finally, when comparing the two doses of crizanlizumab, model cost inputs and their ranges are presented in Table 6. Crizanlizumab 2.5 mg dominated the 5 mg regimen at the estimated WTP (Figure 6). Results of tornado diagram shows that success with and without AEs with crizanlizumab 2.5 mg were the key drivers of the outcome while failure due to additional pain crisis with crizanlizumab 2.5 mg was the least driver (Figure 7).

Cost saving acceptability curve of crizanlizumab 2.5 mg.

A tornado analysis of study medications costs on ICER (crizanlizumab 2.5 mg versus crizanlizumab 5 mg).
Discussion
Based on previous systematic reviews and meta-analyses of SCD treatment modalities, hydroxyurea showed that it is effective in reducing VOC rates.19,20 Nevertheless, SCD patients who are receiving hydroxyurea can go on to have pain crises, subsequent organ damage, and higher mortality rate.
21
Crizanlizumab and
To the best of our knowledge, this is the first cost-effectiveness evaluation of crizanlizumab 5 mg, crizanlizumab 2.5 mg, versus
Our study, with a follow-up duration of a 1-year span, showed that the 5 mg crizanlizumab achieved a success rate of pain crisis averted of 0.5152 compared to 0.4503 with
The sensitivity analyses confirmed the robustness of our findings and showed that the probability of success with AEs with crizanlizumab 5 mg had the main effect on the outcome in relation to the comparison between crizanlizumab 5 mg and
Of note, the acquisition cost of both drugs was the biggest cost driver in model analysis, followed by hospitalization costs.
While there are no similar literature models to compare to, our study model is focusing on the main outcome targeted for SCD management (i.e., VOC) followed, where the VOC averted was the outcome of interest since VOCs decrease the quality of life (QoL) and are the main cause of hospital visitations in SCD and the increase in the risk of death. It is essential to emphasize the model’s narrow focus, primarily centered on the single outcome of acute vaso-occlusive pain. It’s important to note that the available data did not afford us the opportunity to model other critical acute outcomes frequently associated with SCD, such as ACS, which stands as a leading cause of mortality in SCD. Additionally, the limitations of the data hindered us from modeling major chronic complications of SCD that carry substantial morbidity, economic burden, and a significant impact on the QoL, including conditions like stroke, chronic renal failure, and avascular necrosis. New therapies that reduce SCD hospitalizations are desirable given the potential to impact healthcare utilization, but also to reduce disease burden and decrease mortality and morbidity.25,26
Limitations
Our economic evaluation of crizanlizumab and
The presence of potential heterogeneity among patient subpopulations, particularly in a multinational country like Qatar, underscores the need for further investigation into diverse patient demographics, including factors such as race and ethnicity.
Additionally, our study is limited by the age restrictions of the RCT data sources, which may impact the generalizability of our findings to the pediatric SCD population. We anticipate that ongoing trials, such as the assessment of crizanlizumab in pediatric patients as young as 2 years of age, will provide valuable insights to enhance the accuracy of our economic model for this specific population.
While our study primarily focused on clinical and economic outcomes, we acknowledge the absence of an assessment of the QoL due to the lack of comparative QoL data from the RCTs. Moreover, the potential impact of drop-out rates and real-world medication compliance further emphasizes the need for cautious interpretation of our findings, emphasizing the importance of considering practical aspects of medication use beyond clinical efficacy.
It is important to acknowledge that our analysis has not incorporated potential additional benefits of these medications that have been presented in analyses such as Boshen Jiao et al. 27 Furthermore, we recognize the need to clarify the model’s assumption regarding the reduction of ACS events with crizanlizumab. The report of zero ACS events in the pivotal trial by Ataga et al. does not necessarily indicate the complete elimination of ACS. Therefore, further investigation into the impact of crizanlizumab on ACS should be considered in future studies to refine the model’s assumptions. 28
We also acknowledge the potential impact of glutamine on reducing blood transfusions, as indicated by Zaidi et al. 29 The subjectivity and lack of uniformity in the decision to administer blood transfusions pose a challenge in assessing their precise impact. Addressing the variability in clinical practice and its implications on the economic evaluation is an important consideration for future studies. 29
Furthermore, distinguishing between patients on and off hydroxyurea treatment is a valid concern. Addressing subgroups like pregnant women and those with renal failure, who may not be on hydroxyurea, should be a focus for future studies to comprehensively understand medication effectiveness and economic impact within these populations. 29
Additionally, the limitations imposed by the availability of model inputs from RCT studies by Niihara et al. and Ataga et al. Nevertheless, it highlights the importance of incorporating a wider range of estimates in sensitivity analyses where feasible from other studies on both drugs.
Lastly, the differences in reporting criteria for adverse effects, as highlighted in the Niihara et al. and Ataga et al. studies, underline the need for future studies that standardize AEs reporting criteria. This standardization will enable meaningful comparisons and a comprehensive assessment of adverse effects and their associated costs.
Conclusion
Our baseline analysis suggested that each of the 5 and 2.5 mg/kg doses of crizanlizumab reduces pain crises at a higher rate than
Footnotes
Acknowledgements
We acknowledge hematology and clinical pharmacy departments for their significant contribution.
Author contributions
All authors equally contributed to writing and editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Will be supported by Qatar National Library if accepted (no grant number).
Data availability statement
The original contributions presented in this study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics approval
Informed consent
Informed consent was not sought for the present study because human material or data were not used.
