Abstract
Atopic dermatitis is a chronic inflammatory skin disease that may progress to erythroderma in severe cases. Biologic agents such as dupilumab have recently become the mainstay of systemic treatment for moderate-to-severe cases, yet many patients remain refractory to therapy. Here, we present a case of erythrodermic atopic dermatitis, resistant to prednisone and dupilumab, with remarkably rapid achievement of remission following treatment with upadacitinib, an oral selective Janus kinase 1 inhibitor.
Introduction
Atopic dermatitis is a prevalent inflammatory skin condition, typically manifesting as chronic, itchy, erythematous papules, and plaques, with a substantial impact on quality of life. 1 The pathophysiology of atopic dermatitis involves a multifactorial interplay of genetic predispositions, environmental triggers, and immune dysregulation leading to skin barrier disruption and heightened inflammatory responses. 2 A severe and rare manifestation of atopic dermatitis is erythroderma where over 90% of the body’s surface area is affected and is associated with severe morbidity, risk of infection, electrolyte disturbances, and elevated mortality. 3
The management of moderate-to-severe atopic dermatitis often necessitates systemic treatments, which have historically included corticosteroids, immunosuppressants like cyclosporine, and methotrexate. 1 However, biologic medications have transformed the therapeutic landscape. Dupilumab is a monoclonal antibody that inhibits interleukin-4 (IL-4) and interleukin-13 (IL-13) signaling, is indicated for atopic dermatitis, and has demonstrated efficacy in improving skin disease and pruritus. 4 Janus kinase (JAK) inhibitors have emerged as an alternative to injectable biologics, with a fast onset of action and considerable efficacy in controlling disease activity. 1 The use of JAK inhibitors in moderate-to-severe atopic dermatitis has shown promise, offering high-affinity modulation of immune pathways involved in atopic dermatitis and the potential for therapeutic resolution in cases refractory to traditional treatments. 5 We present a case of a patient with erythrodermic atopic dermatitis resistant to both prednisone and dupilumab who achieved rapid resolution with upadacitinib, an oral selective JAK1 inhibitor.
Case report
A 26-year-old male with atopic dermatitis had progressed to erythroderma. They were of African-Canadian background with a darker skin phototype, and the extent of their disease had been chronically underestimated likely due to less appreciable erythema. They had presented to a rapid access dermatology clinic due to worsening atopic dermatitis, chills, rigors, weight loss, and diffuse reactive lymphadenopathy. He was previously diagnosed with atopic dermatitis in childhood and had been well controlled with topical therapy alone. He had no previous history of hospitalization or skin infections requiring systemic antibiotics. He is otherwise healthy and has allergies to tree nuts. Initially, due to his systemic involvement and diffuse lymphadenopathy, skin biopsies were obtained to rule out cutaneous T-cell lymphoma. Appropriate systemic work-up prior to starting immunosuppressive therapy was also performed.
He was initially given a loading dose of dupilumab 600 mg subcutaneous injection in the clinic, followed by 300 mg injections subcutaneously every 2 weeks with no improvement after 1 month. In addition, he was given a 10-day course of cefadroxil 500 mg twice daily for secondarily infected atopic dermatitis. When he did not respond to dupilumab injections, he was started on a prednisone taper beginning at 35 mg once daily as a bridging therapy prior to starting upadacitinib 30 mg once daily for 3 months.
On initial physical examination, he was a skin phototype 6 individual with erythroderma and extensive lichenification and excoriation as well as facial and flexural predominance. His severity scoring was recorded as body surface area (BSA) of 95%, eczema area and severity index (EASI) 72, IGA 4 (Figure 1). He was found to have palpable lymph nodes to the bilateral cervical chains and axillae. His palms and soles were spared and there was notable loss of eyebrow hair. There was little to no improvement with the treatment of dupilumab, prednisone, or betaderm 0.1% ointment. After only less than 1 month of treatment with upadacitinib, the patient’s skin improved to BSA 25% and he was able to resume work, sleep, and other activities of daily living (Figure 2). This further improved to BSA 20%, EASI 13, physician’s global assessment (PGA) 3 after 2 months of treatment with upadacitinib 30 mg once daily and he continued to improve over time.
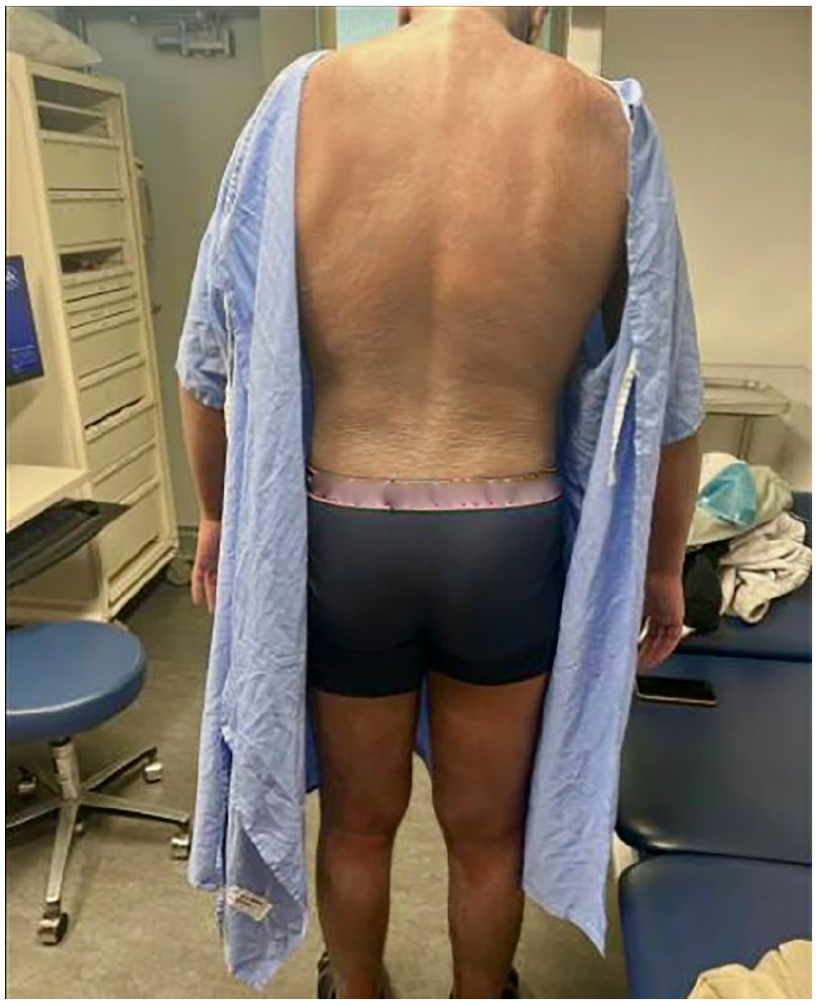
Before treatment with upadacitinib.

One month of treatment with upadacitinib.
Discussion
Here, we illustrate the efficacy of upadacitinib in achieving significant improvement of erythrodermic atopic dermatitis, resistant to conventional therapies including topical corticosteroids, dupilumab, and prednisone. This clinical response was achieved rapidly, with significant improvement after less than 1 month of treatment with upadacitinib, and no associated adverse effects. There has been one other report of improvement with upadacitinib 30 mg daily in a patient with erythrodermic atopic dermatitis which was resistant to oral prednisolone, dupilumab, cyclosporine, and baricitinib. However, this response was achieved over a 5-week treatment period. 6 Our case provides further support for the use of upadacitinib in erythrodermic atopic dermatitis and demonstrates a remarkably rapid onset of skin clearance and symptomatic relief.
Several studies have supported the role of upadacitinib in the management of atopic dermatitis. A recent head-to-head randomized controlled trial comparing dupilumab to upadacitinib in patients with moderate-to-severe atopic dermatitis found that upadacitinib had superior efficacy over a 16-week treatment period, but this different lessend at 24 weeks. Upadacitinib was associated with a more rapid onset of skin clearance compared to dupilumab. Rates of serious infections were higher in patients treated with upadacitinib compared to dupilumab. 4 A recent analysis of pooled safety data from clinical trials for upadacitinib showed no increased risk for venous thromboembolism, major adverse cardiovascular events, or malignancy. 7 Conjunctivitis was the most commonly reported side effect in those who received dupilumab. 4 This is supported by a network meta-analysis which found that JAK-1 inhibitors (upadacitinib and abrocitinib) were associated with the greatest efficacy outcomes compared to dupilumab and baricitinib in moderate-to-severe atopic dermatitis. 1
The efficacy of upadacitinib has also been investigated in patients with an inadequate response to dupilumab. A multicenter prospective cohort study found that the efficacy of upadacitinib in moderate-to-severe atopic dermatitis was similar in dupilumab non-responders and dupilumab responders/dupilumab-naïve patients. In this study, a significant clinical improvement was achieved within the first 4 weeks of treatment with upadacitinib. 8 A multicenter retrospective study also reported a 76.9% treatment success rate with upadacitinib in dupilumab non-responders. 5 Multiple case series have also reported successful treatment of atopic dermatitis with upadacitinib in patients unresponsive to dupilumab.9–11
Several pro-inflammatory cytokines involved in the pathogenesis of atopic dermatitis, including IL-4, IL-13, and IL-31 act via JAK/STAT signaling pathways. 12 The effectiveness and rapidity of therapeutic response with upadacitinib may be due to the upstream inhibition of cytokine signaling pathways and greater suppression of the intracellular cytokines, compared to dupilumab. Thus, there may be a promising role for JAK inhibitors such as upadacitinib in patients with severe, erythrodermic atopic dermatitis requiring rapid resolution of symptoms, as described in this case. Further studies are required to determine the most effective dose of upadacitinib in this patient population.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
The patient provided informed consent for publication of the case report and images.
