Abstract
Janus kinase (JAK) inhibitors are novel molecules increasingly prescribed for various dermatologic conditions. However, the Food and Drug Administration recently reported increased risks of malignancy in patients taking this class of medication. To shed more light on this potential adverse effect, we present a patient with cutaneous T-cell lymphoma possibly associated with his treatment with a JAK inhibitor for atopic dermatitis. To our knowledge, there are no reported cases of cutaneous T-cell lymphoma in association with JAK inhibitors in the literature. We highlight the importance of remaining cautious when prescribing this new class of medication, especially in patients with risk factors for malignancy. Moreover, when faced with atypical presentations of atopic dermatitis, we stress the need for a biopsy to make the correct diagnosis prior to treatment. Lastly, we encourage further studies to better characterize the malignancy risk associated with JAK inhibitors.
Introduction
JAK inhibitors are novel molecules that are gaining traction in dermatology. They are being increasingly prescribed for various dermatologic conditions. Moreover, there are several studies currently underway to assess their effectiveness in the treatment of multiple other diseases, which will likely expand their use in the near future. Although there is no denying that this drug is showing a lot of promise, its associated risks are still being studied. A recent Food and Drug Administration (FDA) report regarding the increased risks of malignancy associated with this class of medication emphasizes the importance of remaining cautious when prescribing this new drug. 1 Herein, we present a case of cutaneous T-cell lymphoma that is possibly associated with the use of JAK inhibitors.
Case report
A 44-year-old otherwise healthy male with no prior history of atopy in childhood started experiencing localized pruritus on his flanks in 2013. In 2020, he developed worsening pruritus and new erythematosquamous plaques on his abdomen, arms, and face. He was evaluated by a dermatologist, who made a clinical diagnosis of severe generalized eczema. No biopsy had been performed at the time.
He was initially treated with topical steroids and phototherapy, but his symptoms did not improve. He thus began upadacitinib in March 2022 with partial relief of his symptoms. While on upadacitinib, he was diagnosed with recurrent episodes of bullous impetigo. Upadacitinib was therefore stopped in October 2022.
He noted rapid deterioration of his symptoms in November 2022. He experienced diffuse hair thinning, development of leonine facies, debilitating generalized pruritus and redness, weight loss, and night sweats. He was assessed by a dermatologist in November 2022, who biopsied his left cheek. The biopsy revealed pilotropic mycosis fungoides, which prompted a referral to our cutaneous lymphoma clinic.
At the time of our initial evaluation in February 2023, the patient had been on oral methotrexate since January 2023 and multiple steroid creams, with minimal improvement in his symptoms. He had also recently finished a course of oral prednisone. Physical examination revealed leonine facies with facial erythema, loss of eyebrows, diffuse hair thinning, and scalp hyperkeratosis (Figure 1(a) and (b)). He was also noted to have keratosis pilaris-like perifollicular papules on around 80% of his body surface area, which coalesced to form generalized erythematous infiltrated plaques (Figure 1(c)). Palpable lymphadenopathy of 1 cm in diameter was appreciated in the occipital, axillary, and inguinal regions.
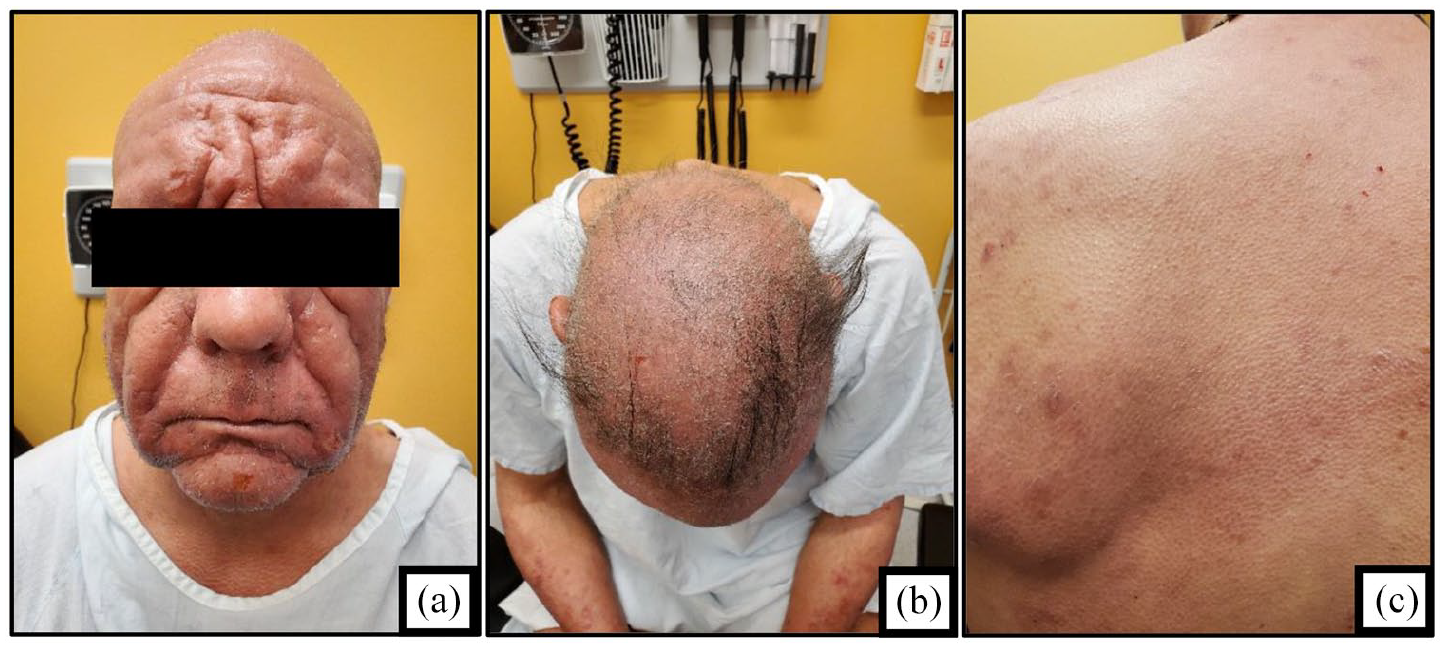
Presentation of the patient’s cutaneous T-cell lymphoma. (a) Leonine facies. (b) Alopecia and scalp hyperkeratosis. (c) Keratosis pilaris-like perifollicular papules coalescing into erythematous infiltrated plaques.
The patient’s blood workup did not show anemia or lymphocytosis. Additional blood tests for lymphoproliferative disorders revealed an increased CD4/CD8 ratio of 22:1 and TRBC1 monoclonality. These results were discussed with hematology-oncology, who stated the patient did not meet the criteria for Sézary syndrome due to the low number of clones. He underwent computerized tomography scans of his thorax and abdomen, which revealed no evidence of lymphoma.
In May 2023, he noted partial relief of his pruritus with weekly subcutaneous methotrexate, aprepitant, hydroxyzine, and pregabalin. He was also followed by hematology-oncology and radiation oncology for human leukocyte antigen (HLA) typing ahead of a possible bone marrow transplant and consideration for electron beam therapy, respectively.
Discussion
Atopic dermatitis treatments are constantly evolving. JAK inhibitors are novel molecules that show promise in the treatment of atopic dermatitis and are becoming more widely prescribed by dermatologists. However, in 2021, the FDA issued a warning that JAK inhibitors may be associated with an increased risk of lymphomas and other malignancies. 1 This report was based on the ORAL surveillance study, which compared rheumatoid arthritis patients over 50 years of age treated with tofacitinib with those treated with a tumor necrosis factor inhibitor during a 4-year follow-up period. 2
There have been no reports of cutaneous T-cell lymphoma induced by a JAK inhibitor at this time. However, considering the recent FDA warning, we believe the development of cutaneous T-cell lymphoma in this patient could possibly be associated with his prior treatment with a JAK inhibitor, upadacitinib. As JAK inhibitor use becomes more widespread and more data is collected, the association between this drug and malignancies, including cutaneous lymphomas, can be better understood.
It is important for dermatologists to carefully choose which patients to prescribe JAK inhibitors to. They should also consider other therapies first. Risks and benefits should be discussed, including the risks of malignancy. This is especially important in patients who are at increased risk of malignancy (e.g., personal or family history, smoking, and older age). 1 As they are a recent drug, there is still a lot of research needed to properly assess their safety and risks. As such, clinicians must be particularly cautious when prescribing JAK inhibitors.
It is important to note that since no biopsy was performed at his initial evaluation, it is impossible to establish if this patient presented with severe atopic dermatitis in 2020, which then progressed to cutaneous T-cell lymphoma, or already had cutaneous T-cell lymphoma in 2020. The two entities can be difficult to differentiate clinically. 3 Hence, it is possible that upadacitinib had no role in the development of cutaneous lymphoma in this patient if this condition was simply undiagnosed at initial presentation. We, therefore, stress the importance of making the correct diagnosis prior to treatment. Atypical presentations of atopic dermatitis, such as a first presentation at an older age without a history of atopy or severe disease not responding to therapy, should prompt the clinician to exclude other diagnoses by taking a biopsy. 4
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
The patient has provided informed written consent to publish this report and the associated images.
