Abstract
The aerosol box was widely used to shield healthcare providers from exposure to COVID-19 during single-lumen intubation procedures. However, it has not previously been evaluated for its use in double-lumen tube intubations. This report presents the case of a 25-year-old COVID-19-positive male with a fever who required an emergency thoracotomy for a mediastinal abscess. During the rapid-sequence induction of general anesthesia, an attempt to use the aerosol box for double-lumen tube intubation was made. The attempt faced unique challenges due to the aerosol box’s restrictive dimensions and the double-lumen tube’s physical characteristics, such as length and flexibility, resulting in an unsuccessful first attempt. Consequently, the aerosol box was removed, and a successful intubation was achieved without it. Postoperatively, the patient remained intubated, was transferred to the intensive care unit, and was extubated on the second postoperative day, followed by intensive care unit discharge. This experience suggests that the standard aerosol box size (50 cm wide, 40 cm deep, and 50 cm tall) may not be suitable for double-lumen tube intubations. This highlights the importance of assessing the feasibility of each aerosol box before its clinical use in such procedures.
Introduction
The aerosol box, also known as the intubation box, is designed to prevent exposure of healthcare providers to the virus when intubating patients who are positive, or who are suspected to be positive, for COVID-19. Numerous studies have examined these devices,1–4 and hundreds of them were distributed and used in clinical practice worldwide.5–8 However, the aerosol box has only been studied for single-lumen tube (SLT) intubation, and the feasibility of its use for double-lumen tube (DLT) intubation is unknown. Here we report our intraoperative experience with the DLT intubation using the aerosol box.
Case presentation
A 25-year-old COVID-19-positive male (height 178 cm, weight 91 kg) with leukocytosis and fever (39.6°C) was found to have a mediastinal abscess (Figure 1) and scheduled for emergency thoracotomy for mediastinal washout. The plan was to use a left-sided DLT (39 Fr, Broncho-Cath™, Covidien, USA) to facilitate lung isolation. Standard American Society of Anesthesiologists (ASA) monitors were applied in the operating room, and the aerosol box (50 cm wide, 40 cm deep, and 52 cm tall) was placed over the patient’s upper body, including the head, as described in previous reports.2,6–8 An assigned senior anesthesia resident was the primary operator for airway management, and the attending cardiothoracic anesthesiologist was bedside, prepared to assist with airway management. The DLT and a videolaryngoscope (Storz C-MAC® 4 blade) were placed inside the aerosol box and prepared for use. Adequate pre-oxygenation was followed by rapid-sequence induction, and mask ventilation was avoided to minimize the risk of virus exposure. After satisfactory muscle relaxation was achieved and the vocal cords were fully visualized via the videolaryngoscope, the operator attempted to place the DLT. However, the operator struggled to engage the tube tip into the patient’s mouth, as the distance between the patient’s mouth and the ceiling of the aerosol box was shorter than the length of the DLT. This resulted in the proximal end of the DLT constantly contacting the box ceiling during tube manipulation (Figure 2(a)). To overcome this, the DLT was bent sharply so that the tip of the DLT was able to pass through the patient’s oral cavity (Figure 2(b)). Despite this, further advancement of the tube toward and through the vocal cords was not feasible because the hyper-angulated tube could not be aligned with the pathway to the vocal cords. As the patient started desaturating, the procedure was abandoned, and mask ventilation was initiated. At this point, we felt that using the aerosol box would make DLT intubation too challenging. The box was subsequently removed, and the patient was successfully intubated with the DLT on the second attempt without utilizing the intubation box. The surgery was completed without any significant events. Postoperatively, the patient remained intubated and was transferred to the intensive care unit (ICU). The patient was extubated on postoperative day 2 and subsequently discharged from the ICU.

Chest computed tomography with intravenous contrast. There is a diffuse increase in soft tissue attenuation mixed with air scattered throughout the mediastinum, representing a large mediastinal abscess measuring 5.6 cm × 5.1 cm.
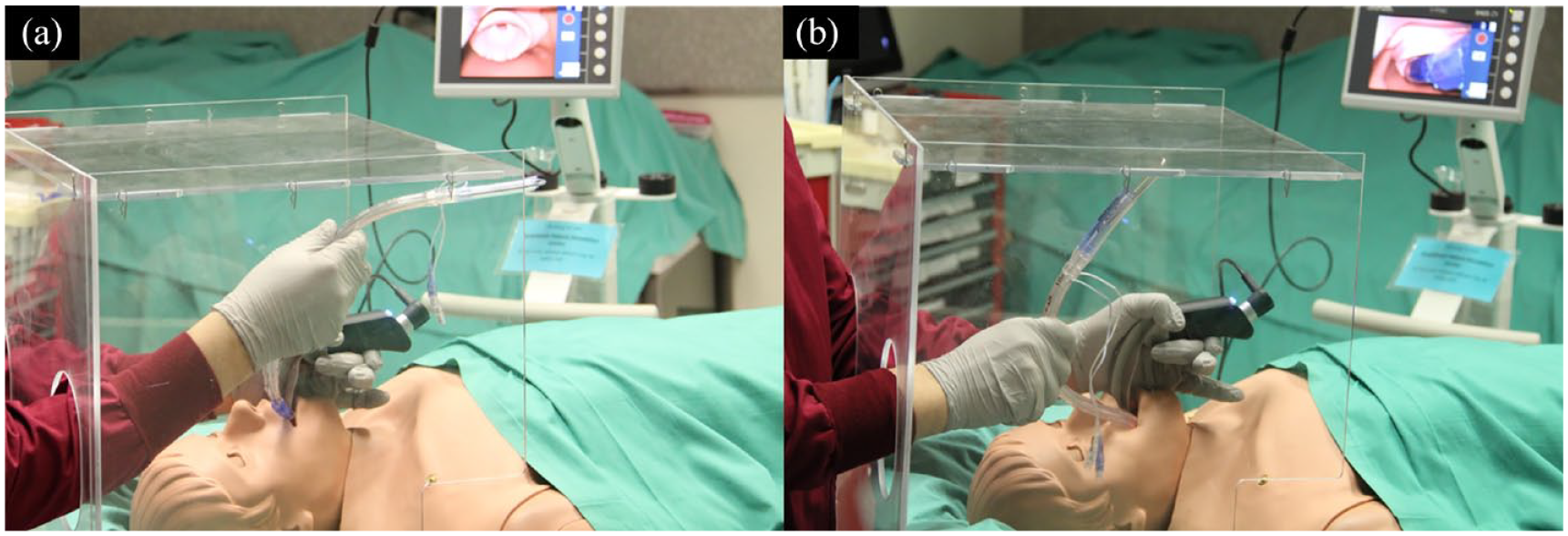
Simulation of double-lumen tube intubation (left-sided 39 Fr, Broncho-Cath™, Covidien, USA) using a manikin with the aerosol box. (a) The tube does not fit within the aerosol box (50 cm wide, 40 cm deep, and 52 cm tall). (b) The tube has to be acutely bent during intubation to accommodate within the aerosol box, making the intubation a real challenge.
Discussion
To our knowledge, this is the first report on the use of the aerosol box for DLT intubation. When COVID-19-positive patients require one-lung ventilation, the DLT can be used. However, anesthesia providers at our institution only had an opportunity to practice SLT, but not DLT, intubation in a manikin using the aerosol box. Without proper practice prior to this emergency surgery, we assumed that the aerosol box would also work for the DLT. In fact, the longer length and limited flexibility of the DLT increased the difficulty of its manipulation inside the box. In addition, we realized that the aerosol box did not provide adequate room for removal of the stylet even if DLT intubation had been successful in this case. Of note, during the DLT intubation, the attending cardiothoracic anesthesiologist, possessing substantial experience, was positioned next to the operator, who was a senior resident. However, the usual intervention and assistance by the attending anesthesiologist were hindered by the barrier created by the aerosol box. Based on our experience, we believe that the original size of the box (50 cm wide, 40 cm deep, and 50 cm tall), which was first described by a Taiwanese doctor 2 and which is almost identical to our box, does not fit appropriately for the DLT. DLT intubation should be avoided using this aerosol box as it would most likely result in intubation failure. Failure or delay of intubation not only increases the risk of hypoxia in respiratory compromised COVID-19-positive patients but also the risk of unnecessary virus exposure. While this single case report does not confirm the reproducibility of the traditional aerosol box (50 cm wide, 40 cm deep, and 50 cm tall) being unsuitable for DLT intubation, subsequent observations using simulations also yielded similar results (Figure 2). It is suggested that enlarging the box size may be necessary for DLT intubation; however, this might interfere with the entire intubation process. Currently, different shapes and sizes of aerosol boxes are available at various locations worldwide3–6,8; thus, the feasibility of each box for DLT intubation must be assessed prior to clinical application. Without a proper size of aerosol box available, the use of personal protective equipment or shields for healthcare providers should be applied during DLT intubation.
Conclusion
This report presents a case of a COVID-19 patient who required DLT intubation for an emergency thoracotomy. Using the aerosol box, DLT intubation was found challenging due to the box’s limited dimensions and the DLT’s longer size and reduced flexibility. Based on this experience, we conclude that the standard-sized aerosol box may not be suitable for DLT intubation and recommend that the compatibility of each aerosol box for DLT intubation be assessed before its clinical use.
Footnotes
Acknowledgements
Not applicable.
Author contributions
N.B.C., Z.D., A.S., S.S., E.V.A., and S.H. contributed to manuscript writing. D.A. wrote the initial draft in addition to contributing to manuscript writing. All authors approved the final version of the manuscript and ensured the integrity and accuracy of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
