Abstract
Cerebrovascular stroke caused by skull base meningioma has been rarely reported. A 30-year-old male presented (April 2015) with acute right-sided hemiplegia. His brain neuroimaging (computerized tomography and magnetic resonance imaging) showed left ischemic infarction in the territory of middle cerebral artery. Magnetic resonance imaging also showed a right parasellar solid lesion which extended to the right basisphenoid and cavernous sinus and attenuated the right internal carotid artery. It also had left smaller parasellar extension. The lesion enhanced uniformly and strongly following gadolinium injection. Digital subtraction angiography using selective catheterization of both common carotid and left vertebral arteries (07/13/2015) showed occlusion of both internal carotid arteries and faint visualization of left terminal internal carotid artery and its bifurcation. The right internal carotid artery and its branches were not visualized. Left vertebral injection showed prominent left vertebral and basilar arteries and filling of both internal carotid arteries through posterior communicating arteries. A faint blush of contrast was noticed at the parasellar region coinciding with meningioma. The patient received three treatment sessions of gamma knife radiosurgery as follow: 20 cc of the tumor was treated with 12 Gy (15 August 2015), 1.7 cc was treated with 10 Gy (31 January 2016), and 2.5 cc was treated with 11 Gy (13 August 2016) which resulted in complete clinical recovery and tumor size reduction. Compensation from the posterior communicating and external carotid arteries might explain the complete clinical recovery after tumor size reduction with gamma knife radiosurgery.
Keywords
Introduction
The parasellar region is defined as the area immediately surrounding the pituitary (sella turcica). It is anatomically complex as it has no specific landmark boundaries. Its arbitrary boundaries include the cavernous sinus (laterally), nasopharynx, and the medial parts of the temporal lobes. The boundaries may also involve embryonic remnants. 1 The cavernous sinus is bounded inferiorly by the basisphenoid and sphenoid sinus and superiorly by diaphragma sella, suprasellar subarachnoid spaces containing the optic nerves and chiasm, hypothalamus, tuber cinereum, and anterior third ventricle. 2 Parasellar lesions include pathologies which arise from the structures surrounding the sella turcica (neoplastic, inflammatory/infectious, developmental, and vascular lesions). 2 These structures include the meninges, visual pathways, other cranial nerves, major blood vessels, hypothalamus, pituitary gland, brain parenchyma, and skull base. The manifestations of parasellar pathologies are diverse depending on the location, size, and growth potentials of each lesion and its compression on or damage to the adjacent specific structures. 3 It has been observed that 9% of parasellar lesions are due to extension of primary pituitary adenoma (a sellar mass) to the parasellar region. 3 These manifestations may include headache, increase intracranial tension, visual loss, cranial nerve palsies, diencephalic syndrome, diabetes insipidus, hypopituitarism, and hyperprolactinaemia (if there is compression on the pituitary stalk). 3
Meningioma is the second commonest (~10%–15%) parasellar lesion after pituitary adenoma. It is the commonest tumor of the anterior and central skull base.4,5 It is the commonest primary, extra-axial, non-glial intracranial space-occupying tumor, comprises 15% of primary brain tumors, and has an incidence rate of 3–13/100,000. It appears at any age with a peak incidence in adult women in their 40s. 6 Meningioma commonly locates in the parasagittal region and frequently originates in the suprasellar (diaphragma sella and tuberculum sella), frontobasal, temporobasal, sphenoid wing (medial lesser wing of sphenoid), anterior clinoid or petroclival regions, cavernous sinus wall, and optic nerve sheath.3,7 Meningioma usually manifests by slow and progressive signs. The usual clinical presentations are visual disturbance and occasionally endocrine dysfunction with mild increase in blood prolactin concentrations. It has been observed that the malignant behavior of meningioma may occur with any grade.3,7 Meningioma rarely occupies the sella mimicking a pituitary adenoma. 8 Cerebrovascular stroke is a rare presentation of parasellar space occupying lesion particularly meningioma of skull base due to compression on or encroaching the cavernous portion of internal carotid artery (ICA) or its branches.4,9–11
Description of the case
A 30-year-old right-handed man presented with acute weakness of the right upper limb (24 April 2015) which increased in severity and progressed within 2 days to involve the right lower limb. The patient denied presence of headache or seizures. Neurological examination showed right upper motor neuron facial muscles’ weakness and weakness of the right hand of 2/5 Medical Research Counsil (MRC) scale, shoulder, toes, and ankle (3/5) and knee and hip muscles (4/5); diminished sensation on the right side of the body (hemihypoalgesia); diminished right tendon jerks (biceps, brachioradialis, triceps, quadriceps, and ankle); and right Babinski sign. He had normal other cranial nerves and fundus. His National Institute of Health Stroke scores (NIHSS) 12 was 12 and his modified Rankin Scale (mRS) grade was 4 (i.e. moderately severe disability: unable to do bodily needs or walk unassisted). 13 Computed tomography (CT) of the brain (24 April 2015) showed left fronto-temporo-parietal hypodense area consistent with ischemic infarction in the territory of the middle cerebral artery (MCA) (Figure 1). Magnetic resonance imaging (MRI) (1.5 T) (25 April 2015) also showed a right parasellar solid lesion extended caudally into the right sphenoid sinus and greater wing of sphenoid bone and basisphenoid and also expanded the cavernous sinus and attenuated the right ICA. It also had a smaller left parasellar extension. There was no suprasellar extension. The pituitary stalk was kept central. The optic chiasm was normal. This lesion enhanced uniformly and strongly following gadolinium injection. The lesion measured about 4.1X4X3.5 cm in its maximum diameter (Figure 2). Treatment with dexamethasone as dehydrating agent for 2 weeks (8 mg intravenously twice daily) resulted in partial recovery of motor symptoms (mRS grade = 3, i.e. moderate disability or able to do bodily needs with some help and walk unassisted). Magnetic resonance angiography (MRA) showed absence of both ICAs. The anterior (ACA) and middle (MCA) cerebral arteries had normal courses and calibers and mostly fed via the posterior communicating arteries (PCAs). The vertebral and basilar arteries had normal flow void signal, courses, calibers, and enhancements (25 April 2015). Upon catheterization of both common carotid arteries (CCAs), occlusion of both ICAs was noted almost at their origins (13 July 2015). ACAs and MCAs had normal courses and calibers and mostly fed via the PCAs. There were faint visualization of the left terminal ICA and its bifurcation via faint collaterals (ophthalmic artery). The right ICA and its branches were not visualized. Left vertebral injection showed prominent left vertebral and basilar arteries as well as filling of both ICAs through patent both PCAs. Both ACAs and MCAs were opacified but there was slight and slower filling on the left side which might be due to partial filling from external carotid artery (ECA) or a relatively smaller left PCA. A faint blush of contrast was noticed in the parasellar region coinciding with meningioma in concern which apparently got its blood supply from the right internal maxillary artery. External carotid (ECAs), common carotid (CCAs), and subclavian arteries had normal calibers. The venous circulation was unremarkable (Figure 3). Hormonal profiles for thyroid (thyroid-stimulating hormone, T3 and T4), reproductive (follicle-stimulating hormone and luteinizing hormone), growth, cortisol, and prolactin hormones were unremarkable. Automated perimetry (central 24, 2, Sita-standard strategy) (30 May 2015) showed mild generalized depression of retinal sensitivity in both eyes and bilateral significant field defects. Transoesophogeal echocardiography and patient’s coagulation profile were unremarkable. The patient underwent focused gamma knife radiosurgery (GKRS) using Leksell Model C or Perfexion (Elekta Inc.) (first session) (15 August 2015). The lower part of the tumor (20 cc) (which showed tumor’s extension around the visual pathway) was treated with 12 Gy to the 50% isodose with 98% cover. Complete clinical recovery was observed within 3 weeks after the first GKRS session. In the second session, 1.7 cc was treated with 10 Gy to the 50% isodose with 93% cover (31 January 2016). In the third session, 2.5 cc was treated with 11 Gy to the 50% isodose with 94% cover (13 August 2016). On each session, the maximum dose to the visual pathway was 8 Gy which was considered safe. Radiological follow-ups of the patient were done every 6 months for 2 years after the third session (29 August 2018) and showed reduction of the tumor size (3.9×3×3 cm) (Figure 4) which coincided with his unremarkable neurological examination (grade 0 on mRS or no symptoms) (15 June 2019). This study was conducted according to the principles established in Helsinki. Informed written consent was obtained from the patient to publish the details of his clinical history, laboratory, and neuroimaging data.

Axial and sagittal CT brain (A and B) scans showed left fronto-parieto-temporal hypodense lesion (ischemic infarction) (Date: 04/24/2015). MRI-brain showed left white matter lesion in the left fronto-parieto-temporal region (wedge shape) which was hypointense in axial T1W1 (C) with no detected enhancement in post contrast scans, hyperintense in axial FLAIR (D), axial and coronal T2WI (E and F) and DWI (G) scans (i.e. subacute ischemic infarction) (Date: 04/25/2015).
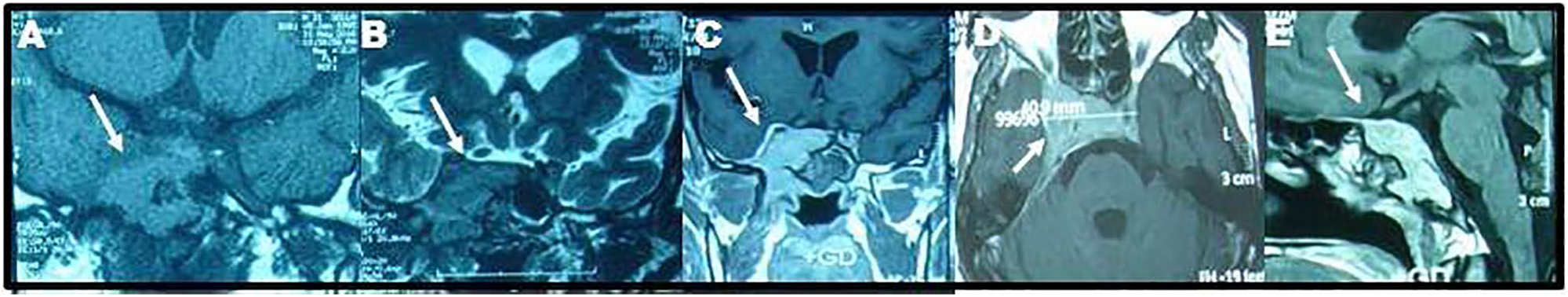
MRI-brain scans [coronal T1W1 (A) and T2WI (B) and coronal (C), axial (D) and sagittal (E) T1W1 with gadolinium enhancement (Gd)] showed a solid lesion which extended caudally into the right sphenoid sinus and greater wing of sphenoid bone and basisphenoid. The lesion also expanded the right cavernous sinus and attenuated the right ICA. It had a smaller left parasellar extension. It had no suprasellar extension and the pituitary stalk was kept central. The pituitary gland and optic chiasm were normal. Following Gd injection, it enhanced uniformally and strongly. The lesion measured about 4.1X4X3.5 cm in its maximum diameter. (Date: 04/25/2015).

MRA showed absence of both ICAs (A and B). ACA and MCAs had normal courses and calibers and mostly fed via PCAs (C). The vertebral and basilar arteries had normal flow void signals, courses, calibers and enhancements (A, B and C) (Date: 04/25/2015). Digital subtraction angiography using selective catheterization of both CCAs and left vertebral arteries showed occlusion of both ICAs. There was faint visualization of the left terminal ICA and its bifurcation via faint collaterals from ophthalmic artery. The right ICA or its branches were not visualized (D-F). Left vertebral injection showed prominent left vertebral and basilar arteries (F-H) and filling of both ICAs through patent PCAs. Both ACA and MCA were opacified but there was slight and slower filling on the left side from ECA. ECAs, CCAs and subclavian arteries had normal caliber (I and J). A faint blush of contrast was noticed at the parasellar region coinciding with meningioma (H-K). Venous circulation was normal (L) (Date: 07/13/2015).

Follow-up MRI-brain scans showed significant reduction of the tumor size after GKRS (3.9X3X3 cm) [Dates: 08/13/2016 (B), 01/31/2017 (c), 05/15/2017 (D) and 01/29/2018 (E)] compared to his initial scan (4.1X4X3.5 cm) [Date: 08/15/2015 (A)].
Discussion
The differential diagnosis of parasellar tumors remains challenging even with the current advances in neuroimaging. A multidisciplinary approach (neurological, endocrinological, ophthalmological, neuroimaging, and histopathological) is necessary for proper diagnosis. Because of the location of the parasellar lesion in our patient, the diagnosis of meningioma was based on the neuroimaging findings, being isointense to the gray matter in T1WI and T2WI MRI-brain scans, and its homogeneous vivid enhancement. 2 Meningioma differs from pituitary adenoma by its differential uniform enhancing pattern and normal hormonal profile and from other parasellar tumors by its potential to encase the ICA, narrow its lumen, and compromise the cerebral blood blow. The occurrence of stroke due to meningioma has been rarely reported. Previous reports observed that skull base meningioma may compress the carotid artery and produce transient neurological symptoms including loss of consciousness, hemiparesis, paresthesias, and global amnesia.14–19 Few case reports documented cerebral infarction due to compression of ICA by skull base meningioma (summarized in Table 1).
Case reports of patients presented with cerebrovascular strokes due to skull base meningioma.

MRI-brain: magnetic resonance imaging of the brain; CT: computed tomography; ICA: internal carotid artery; ECA: external carotid artery; ACA: anterior cerebral artery; MCA: middle cerebral artery; N/A: not applicable.
The rate of development of cerebral ischemia as a complication of meningioma is unknown. In an attempt to estimate the incidence of cerebral ischemia due to compression of the ICA by meningioma, Komotar et al. 9 retrospectively reviewed the medical records of 1617 patients with meningioma from their institution and identified three with meningiomal involvement of ICAs. The authors estimated that the incidence of meningioma-related cerebral ischemia by carotid artery compression was 0.19% (3/1617). Lévêque et al. 5 observed that 32.7% of midline meningioma caused infiltration to ACAs.
The mechanisms of cerebrovascular stroke associated with meningioma are either direct compression or vascular infiltration by the tumor, or indirectly by coagulation disorders or a combination of hypoperfusion and thromboembolization.9,10,14,20,21 We suggest that the complete recovery of our patient was due to reconstitution of blood flow through the PCAs and efficient collaterals from ECAs. Some authors suggested that although meningioma commonly involves the ICA or even completely encases the ICA and its bifurcation into MCA and ACA, its presentation by cerebrovascular stroke is rare because (1) meningioma is a slowly growing and non-invasive tumor. It does not exert sufficient external force to significantly compress the high pressure arterial vasculature compared with venous vessels which can be easily compressed or injured; (2) The slow growth rate of meningioma allows the development of substantial collateral drainage; 22 (3) the thick muscular media segment of the ICA usually offers substantial resistance to vascular compromise. 23
The optimal treatment of meningioma and other benign parasellar tumors is complete surgical removal by a craniotomic route if they are symptomatic or growing lesions. Endovascular intervention is sometimes used before surgery to reduce the size of bulky meningioma. 21 Transsphenoidally (TSS) debulking approaches are currently used to remove lesions involving the tuberculum sella, suprasellar region, cavernous sinus or clivus with minimal lesion to adjacent vital structures. 24 However, parasellar tumors usually have irregular margins and adhere to vital neurovascular structures and do not allow complete resection without the danger of affecting critical brain areas; 25 therefore, surgical debulking and radiotherapy are preferred for such cases and for those with known or suspected residual disease or recurrence after previous surgery. 24 Stereotactic fractionated radiosurgery, which delivers a single fraction of high-dose ionizing radiation on mapped targets, keeping the exposure of adjuvant tissues to a minimum, is preferred to treat unresectable and recurrent sellar and parasellar meningiomas after resection. 3 The mechanism of fractionated radiosurgery is to alter DNA of the tumor (i.e. arrest cell division) resulting in stable tumor size or its shrinkage due to reduction of angiogenesis. The selected dose of radiotherapy is based on an empirical algorithm that evaluated tumor volume, proximity to critical structures (such as the optic apparatus and pituitary gland), preexisting neurological deficits, and previous history of treatment with fractionated radiation therapy. Among published series of 100 or more patients, the rates of significant long-term complications of fractionated radiotherapy ranged from 0% to 16% in centers utilizing median doses of 12–15 Gy and 8 Gy or less has been found to be safe for the optic apparatus. 7 Some reported susceptibility to visual decline despite the 8 Gy dogma.26,27
For the presented patient, treatment with GKRS looks optimal, because resection of the tumor was considered excessively risky and the patient was unwilling to accept the risks of resection. It has been estimated that the risks of injury of adjacent vital structures and persistent neurological deficits after surgery of skull base meningioma is about 60%. 28 Although, the patient had complete clinical recovery for 3 years after the third GKRS session (15 June 2019) and reduction of the tumor size (Figure 4) and absence of visual side effect using the same doses as recommended in the literature, were noted,26,27 we cannot guess the future prognosis as it needs follow-up for at least 5–10 years.
Many authors observed good outcomes with GKRS treatment for parasellar or sellar meningioma.27–31Lee et al. 27 followed-up 159 patients with cavernous sinus meningioma and treated with radiosurgery for a mean of 35 months (range 2–138 months). The authors observed improvement of neurological status in 29%, stabilization of manifestations in 62%, and worsening in 9%. Kondziolka et al. 29 followed-up 972 patients with meningioma in diverse locations and treated with GKRS (a median tumor margin dose of 13 Gy) at the University of Pittsburg. The authors reported tumor control rates in 93% at 5 years and 87% at both 10 and 15 years. These results confirmed an earlier study of 159 patients from the same institution in which tumor volumes decreased in 3%, remained stable in 60%, and increased in 6% of patients. Minniti et al. 30 in their review of 18 studies with 2919 skull base meningioma and treated with GKRS, observed that the 5-year tumor control rate was 91%. Seven of the studies (1626 skull base meningioma) reported an average of 10-year control rate of 87.6%. Sheehan et al. 31 in their multicenter study on 763 patients with sellar and/or parasellar meningioma and treated with GKRS (some of their patients did at least one resection before GKRS or more than one GKRS session), the authors did follow-up for patients for median of 66.7 months (range 6–216 months) and reported stabilization or reduction of tumor volumes in 90.2% and the progression-free survival rates were 3, 5, 8, and 10 years were 98%, 95%, 88%, and 82%, respectively. The authors observed that tumor margin dose of less than 13 Gy significantly increased the likelihood of tumor progression after GKRS. They also noted that in the last clinical follow-up, no change or improvement in neurological conditions in 86.2% of patients, symptom progression in 13.8%, and new or worsening cranial nerve deficits in 9.6%. The long-term worsening has been attributed to the re-growth of the tumor and/or edema and necrosis as adverse effects from radiation. However, some authors reported a poor correlation between clinical and radiographic changes. For example, Bindal et al. 32 reported that 54.5% of patients with neurological progression did not have changes in neuroimaging, and 27.2% had simultaneous neurological and radiographic progression.
Conclusion
We describe an adult male with cerebral infarction as a result of ICA compression by a skull base meningioma encased the cavernous sinus. Angiography showed occlusion of both ICAs at their origins. Compensation from PCAs and partial filling from ECA may explain the complete recovery of neurological manifestations after GKRS. To our knowledge, massive parasellar meningioma with occlusion of both ICAs has not been reported before in the literature.
Footnotes
Acknowledgements
We thank the patient for his approval to publish his data and neuroimagings.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
