Abstract
Low back pain affects over 20% of individuals during their lifetime, and in some patients, it may be associated with scar tissue formation after surgery. Small-fiber neuropathy and scar tissue dysfunction can lead to localized pain by affecting signals to the thalamus. Transcutaneous neuromodulation using Tape with Magnetic Particles shows promise in relieving perceived pain, modulating vascularization and the autonomic nervous system, and reducing dermal temperature. In the present case, a 24-year-old woman with L5-S1 disk herniation experienced low back pain and leg pressure. The surgical intervention provided temporary relief, but scar restrictions caused pain recurrence. Tape with Magnetic Particles application initially induced scar hypothermia and pressure tolerance during posteroanterior tests on lumbar spinous processes increased, reducing pain perception for at least 12 h. Transcutaneous neuromodulation with Tape with Magnetic Particles modulated dermal temperature immediately and for 12 h, reducing perceived pain and sustaining improvement thereafter. This highlights the potential of Tape with Magnetic Particles in managing chronic low back pain associated with scar tissue.
Introduction
One of the most common and difficult problems seen in clinical practice is low back pain (LBP). 1 The likelihood of experiencing acute LBP at some point in one’s life is approximately 70%–85%, and out of those affected, 90% are expected to endure more than one episode. 2 The prevalence of chronic pain affects about 20%–25% of the population.3,4 Several authors have demonstrated a correlation between scarring and LBP.5–7 According to the study by Leal et al., 8 more than 70% of the women in the study with LBP had painful surgical scars. They do not define whether the pain comes from the epidermal or ectodermal scar, from the dermal and connective tissue, or the mesodermal scar, as they originate from different embryological sacs. This differentiation may be relevant to locate the origin of the pain, thus knowing which one to work on first.
During the scarring process, there is a modification within the tissue where there is a settlement of a new vascular system and a change in elasticity. 9 The literature suggests that scar tissue should be different from the original tissue 9 and that there is a difference in thermal behavior between the scar and the peri-scar areas. 10 During tissue repair, there is a change in the amount and nature of collagen production, capillaries, hair follicles, and glands, resulting in a scar with a different color from the original tissue, as well as a loss of elasticity.9–11 Manual therapy increases vascularization of the scar and peri-scar area, suggesting a modification of connective tissue function after treatment. 10
It is possible to document skin changes caused by vascular phenomena due to neurovegetative reflexes in the same area of the body where the patient complains of pain. 12 Infrared (IR) imaging shows the superficial circulation of the skin, thus reflecting the relationship between the sympathetic nervous system (SNS) and cutaneous blood flow. 12 In this way, IR can classify and categorize the mechanisms involved that reflect the performance of the SNS and help in the study of pain. As is well established today, vasomotor control of the skin is performed entirely by the SNS. IR measures the indirect reflex of the sympathetic control of cutaneous microcirculation,13,14 and the cutaneous microcirculation, including the intercellular and interstitial fluids of the lymphatic system, is the heat transport medium of the circulation.15–18 The control of the SNS is performed by medullary fibers; sympathetic afferent fibers from the posterior root go to the spinal ganglion and the intervertebral, then join with sympathetic efferent fibers of the white branch which converge to the paravertebral sympathetic ganglion, then return to the paravertebral sympathetic ganglion and peripheral nerve through the gray branch.15–18 Skin sympathetic nerve activity is connected with the dorsolateral prefrontal cortex, which is connected with emotional arousal or anxiety and the modulation of skin blood flow and sweat release. 19 It has been shown that epidermal cells are innervated by the Autonomic Nervous System (ANS). 20
Comprehending cutaneous sympathetic innervation is crucial for IR pain analysis, as it detects hyperthermic regions and vasoconstrictor responses in spinal nerves that influence pain and thermal energy due to vascular flow, even in cases of external nerve injury without visible anatomical damage. 15 Emerging theories on the relevant protective feedback mechanisms of trigger points include modulation of the presynaptic and postsynaptic SNS. 21 Science suggests that the sensitive nervous system plays a role in localized inflammation by releasing algogenic and vasoactive neuropeptides, including substance P, vasoactive intestinal peptide, neuropeptide Y, and calcitonin gene-related peptide. 15 The sensory neuron, primarily located in the dorsal root ganglion and extending to various body tissues, is likely central to inflammation production. 15 Small-fiber neuropathy (SFN) has traditionally been considered purely a disorder of the peripheral nervous system, characterized by neuropathic pain and degeneration of small-diameter nerve fibers in the skin. 22 Patients with SFN exhibited reduced thalamic connectivity with sensory-motor areas and cutaneous nerve degeneration, correlated with diminished thalamic connectivity to pain-related regions across neuropathic pain phenotypes. 22
During embryonic development, epiblast cells proliferate and migrate to replace the hypoblast, forming the definitive endoderm. The subsequent migration of these cells between the epiblastic and endodermal layers gives rise to a third layer, the mesoderm, with structures vital for life such as the skin and central nervous system (CNS) being primarily derived from the ectoderm. Dysfunctions in these can cause pain or pathologies throughout the body. 23 If there are dysfunctions in the skin, such as scars, it will alter vascularization, 19 and therefore influence afferent stimuli to the thalamus, 22 producing local pain by affecting mesodermal elements of the somatic system (bone-musculo-dermis), or areas that share innervation. The axial skeleton, voluntary muscle, and the dermis of the neck and trunk are derived from somites from the mesoderm innervated by the peripheral nervous system – the first somite subdivision that appears is the sclerotomes. 23 For this reason, anteroposterior pressure is applied to the spinous processes of the lumbar levels.
Kinesiology tape (KT) has been applied to try to influence LBP improvement and rehabilitation in clinical settings, 24 as well as vascularization using a pedaling motion. 25 It is applied for several days26–29 to evaluate its effects but has shown mixed results. A meta-analysis from Li et al. 30 concludes that KT cannot provide significantly greater pain relief to patients with chronic LBP compared to a sham/placebo control. Tape tension is a key factor in the taping method; however, there is limited science regarding the reinforced tension effects of KT on functional performance and pain in healthy individuals. Lee et al. 24 found no significant changes when applying KT from insertion to origin, with 75% elongation after exercise. According to Selva et al., 31 different KT tapes will produce different levels of tension even if the same elongation is used, making it impossible to compare the findings of the studies, and presenting new challenges for research in this field.
Ultraviolet B rays produce electromagnetic energy. 32 Epidermal cells are modulated by UV electromagnetic waves, and although each one has a function, they share a communication network.10,13 This modulates cutaneous sensory transduction, 33 influencing both inflammatory responses and the immune and endocrine systems as a whole.20,34,35 The tape with magnetic particles (TMP) is an elastic adhesive bandage with particles with magnetic properties. When TMP comes in contact with electromagnetic fields, such as those generated by living beings, it creates a magnetic flux, that, in turn, produces a magnetic induction proportional to the variation of the magnetic flux. It has a local or metameric effect, and a systemic or central action in improving perceived pain36,37 by modulating lower limb vascularization, 36 modulating the ANS, 37 and increasing muscle activity and strength. 38 The results of these studies led us to hypothesize that transcutaneous neuromodulation of epidermal cells by TMP could modulate back dermal body temperature and reduce perceived scar pain.36–38
Case
The patient is a 24-year-old white woman with LBP, with pressure radiating from the anterior aspect of the thigh to the knee, and through the external calf to the external ankle. Following a computed axial tomography (Figure 1) of the lumbosacral area on June 13, 1996, prescribed by Dr. Jaime Broseta, professor of neurosurgery, the report concluded that the L3-L4 and L4-L5 spaces showed no signs of disk disease or radicular involvement, and he reported a central disc herniation of L5-S1.
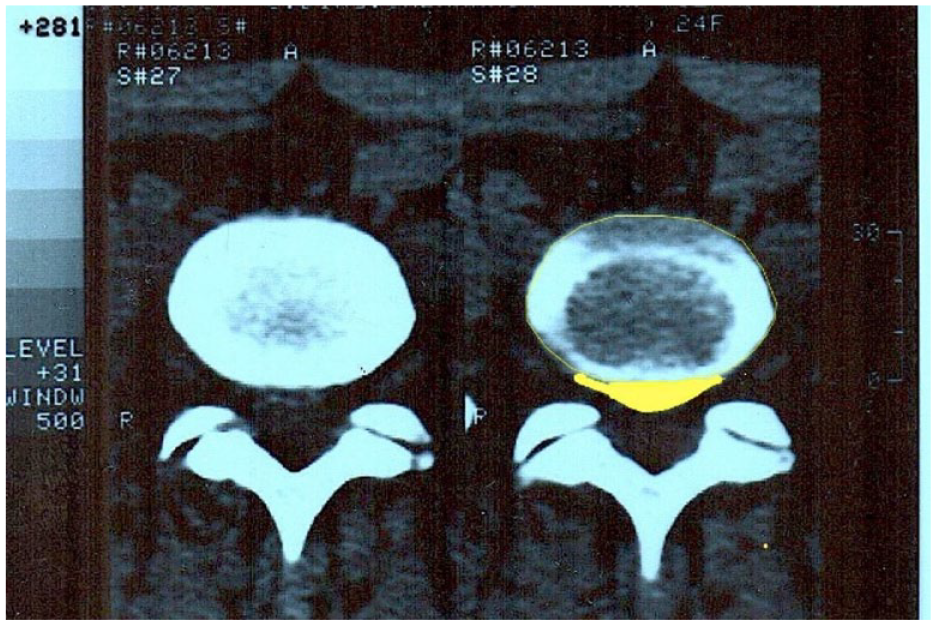
Lumbosacral computed axial tomography.
After drug treatment with oral and injectable anti-inflammatory non-steroidal anti-inflammatory drugs by the traumatologist without effect, she underwent surgery on 25 June 1996, with discectomy at this level using bilateral flavectomy. The operation was uneventful.
In the postoperative period, there were no problems and she was recovering the mobility to be able to perform daily life activities using exercises at home (development of mobility and progressive increase in lumbopelvic and lower limb range of motion with lumbopelvic core stabilization exercises) and in the swimming pool (swimming crawl with a flotation device between the legs, exercises to go up and down stairs in the water and with a flotation device between the legs to do the gesture of running sitting down). She did not have symptoms again until a year and a half later (December 1997), when the restrictions and adhesions of the scar were treated since the patient was moving-blocked and the first LBP appeared 2 days after the treatment. From that LBP onwards, there were successive episodes when she moved more than she was accustomed to in her daily activities without being able to maintain a static standing position during the acute phases. During the following years, she developed chronic pain with acute episodes of pain, in which ibuprofen at 600 mg/8 h was taken. She did not perform continuous physiotherapy, only when she was in acute phases, and did not exercise regularly. Twenty-three years after mobilization of the scar and with pain persisting, TMP was applied to the scar without creating tension in an attempt to modulate the perceived pain. The patient maintained the improvement until 2 months after the application of TMP.
This clinical case aimed to show how the patient’s chronic pain was addressed. The measurements taken of the change in temperature of the healed area were compared with the rest of the back. By not creating tension with TMP and acting on epidermal cells (ectoderm), it was hypothesized to influence small fiber neuropathy of the scarred area and maladaptive brain plasticity. This document was written following the CARE (CAse REport) guidelines 39 for case reports. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the University of Valencia (protocol code 1118645, September 5, 2019). Written informed consent was obtained from the patient involved in the study.
Assessed variables and measuring instrument
The materials used were an unmarked KT used as a control, and a TMP (Magnetic Tape®, S.L., Valencia, Spain) as the experimental treatment. Both tapes were black and could not be identified by their appearance. In addition, dermal temperature was measured with IR thermography (IRT) of images recorded with an OPTRIS PI 450 IRT camera coupled to the Optris PI Connect software (Optris, Berlin, Germany). The IRT camera has a noise-equivalent temperature difference <40 mK with 38° × 29° FOV, a wide range of temperature from 20°C to +100°C, a spectrum range of 7.5–13 μm, a focal plane array sensor size of 382 × 288 pixels, an emissivity set at 0.98, and a measurement uncertainty of ±2% of the overall temperature reading. The size of the capture frame was 55.4 × 40.63 cm (1.5 mm/px). In addition, a Lexitron T2N high-emissivity blackbody (Sensotec Instruments, Barcelona, Spain) was used for the real-time calibration of the IR camera, a PCE-THB 38 weather meter (PCE Instruments, Albacete, Spain) for the control of the room environment, and cold lasers YCHG-650 (Apinex, Montreal, Canada) for the positioning of the participant.
Pressure pain threshold (PPT) measurements were collected using a Wagner Force Dial FDK 20 algometer (Wagner Instruments, Greenwich, CT, USA). The numerical pain rating scale (NPRS) was used to assess perceived pain, quantifying pain intensity. 40 The highest level of pain experienced during rest was evaluated using a 10-point scale (NPRS) (0: absence of pain, 10: extreme pain). Finally, adhesive markers, a laptop computer, and a stretcher were also used (Figure 2).

Equipment and workplace.
The participant was acclimatized in an isolated room (4.87 × 8.53 m2) without temperature sources, and at a mean temperature of (22.1 ± 0.7°C) and a relative humidity of (47 ± 5%) for 15 min.41–43 The participant lay prone on a hydraulic stretcher, and the camera was placed perpendicular to the subject for a more accurate reading. 43
Thermography is an imaging technique that records IR radiation from the skin, with the potential to detect asymmetries in body temperature and relate them to pathologies or risk of injury. However, the location of the reference points and the region of interest (ROI) can influence the recording process. The protocol described by Molina 23 was used due to its very good intra- and inter-examiner reliability and reproducibility since the influence on skin temperature is negligible. A methodology of the measurements and therapeutic approach was followed in 25 steps, as detailed in Appendix A. In addition, the patient followed the recommendations prior to thermography (Appendix B), and the application on the patient’s body is seen in Figure 3.

Location of the region of interests and landmarks.
Results on dermal body temperature
The study analyzed the cutaneous neurovascular response of the TMP versus a control tape to a long-standing fibrous scar, by placing the tape on the skin without creating tension. For this purpose, two ROIs were generated on both sides of the tape, with another one farther away at the dorsal level. The thermal differences between the dorsal reference ROI and the ROIs close to the tape were calculated. The order of application of the tape was established in a double-blind fashion (neither the patient nor the operator knows which is the experimental tape). First, the control tape was applied, and then later the TMP was. Two conditions were applied in a crossover design for each condition with a washout period of 1 day between the trials. The images of the intervention are seen in Figures 4 and 5.

(a) Positioning of the thermographic capture system. (b) lumbar scar and landmarks.

Landmarks and tape application over the lumbar scar.
The thermal differences pre-intervention, at 1, 15, 30 min post-intervention, and 12 h post-intervention between the control tape and the TMP were analyzed. An interactive video of the procedure is found in the Supplemental material (https://youtu.be/tBiD_qmg75E). Regarding the neurovascular response of the ROIs near the tape with respect to the dorsal reference ROI: The ordinate axis (Y) shows the temperature difference between the ROI being detailed (left or right) and the reference ROI, which is separated cranially. The 0 indicated that the temperature was equal in both; if it appeared negative, it was that the ROI (left or right) was colder than the reference ROI (Figure 6).

(a) Neurovascular response of the left region of interest (ROI) near the tape with respect to the dorsal reference ROI. (b) Neurovascular response of the right ROI near the tape with respect to the dorsal reference ROI.
When the TMP was first applied, it was observed that the areas lateral to the scar were more hypothermic than the reference ROI (see Rpre.ΔT and Lpre.ΔT). When the TMP was placed, the temperature difference began to increase, decreasing the temperature on the sides of the tape with reference to the reference ROI (Supplemental Material Video: https://youtu.be/tBiD_qmg75E). It is worth noting that there is an abrupt starting point for the temperature decrease once the TMP is placed. With the control tape, it was observed that the differences that occurred were more typical of the body’s thermal oscillation. It was observed that there was a significant temperature variation with the use of the TMP compared to the control tape. At 12 h, the temperature change was still evident. With the control tape, and without, considering the uptake at 12 h as mentioned above, the temperature variation was minimal (Figure 7).

(a) Neurovascular response of the left region of interest (ROI) near the tape with respect to the dorsal reference ROI. (b) Neurovascular response of the right ROI near the tape with respect to the dorsal reference ROI.
Results on pain
Results for NPRS
The results of the posteroanterior pressure with an algometer on the lumbar spinous processes at 3 kg of pressure before applying the tapes, provided 9 out of 10 pain at L2, 8 out of 10 at L4, and 10 out of 10 at L5 in the three repetitions performed at each level. The same method was used as previously described in another published study. 37 After applying the placebo tape (Table 1, tape 2), the values for pain did not change. On the other hand, when applying the experimental tape (Table 1, tape 1), the values for pain decreased to 7, 3, and 5, respectively. After 12 h of treatment, the NPRS values increased when the posteroanterior pressures were performed again with an algometer, but without reaching the initial values, obtaining now values of 7, 6, and 9, respectively.
Results of NPRS exerting posteroanterior pressures with an algometer.

NPRS: Numerical Pain Rating Scale.
Results for PPT
The results of the posteroanterior pressures with an algometer on the lumbar spinous processes, applying maximum pressure until the patient could no longer tolerate the pain before applying the tapes, provided 2.1 kg of pressure on average on L2, 1.2 kg on L4, and 1.2 kg on average on L5. After applying the placebo tape (Table 2, tape 2), the values for posteroanterior pressures were not significantly modified. On the other hand, when applying the experimental tape (Table 2, tape 2), the values for posteroanterior pressures increased, allowing pressures with more kg before reaching maximum pain, and reaching 2.9 kg on average at L2, 4.5 kg at L4, and 3 kg at L5. After 12 h of treatment, the kg supported at L2 increased to 4.3 and remained very similar at L4 with 4.2 kg, and at L5 with 2.9 kg of maximum pressure.
Results of PPT exerting posteroanterior pressures with an algometer.

PPT: Pressure Pain Threshold (in kilograms of pressure).
Discussion
The skin temperature of the trunk is higher and less prone to fluctuations due to changes in ambient temperature. 28 This further influences the reliability of the results of this case, in addition to the fact that the ambient temperature was controlled. Even so, the changes in temperature were significant before the first minute of the 30 min of thermographic recording.
It is possible that the structure on which the TMP acts is the epidermis since no tension is created during the application, and no action is taken on fasciae, muscles, or any other element that could be affected by the tension created from the surface. Epidermal cells, Merkel cells, keratinocytes, Langerhans cells, and melanocytes connect the skin with the mind through a complex communication network and are closely related to the neuroendocrine and immune systems. In addition, epidermal cells act on the nervous system locally and centrally. 37 Epidermal keratinocytes are in contact with and stimulate the rest of the epidermal cells, 35 acting as nociceptive modulators and noxious sensors both in healthy and pathological conditions. They may act as primary nociceptive transducers as a complement to sensory neurons. 44
The main findings of the present study were (1) the application of transcutaneous neuromodulation by TMP on the lumbar scar without creating tension and (2) that 30 min on the skin immediately modulated the temperature of the back, helping to reduce the perceived pain, and this was maintained 12 h later. These are important findings since after 23 years with LBP (chronic LBP) and functional impotence, the patient was able to carry out the activities of daily living with less perceived pain.
These improvements can be explained by the fact that transcutaneous neuromodulation by TMP can influence the epidermal cells, and this influences the cutaneous nerves. 35 Through the cutaneous nerves, the SNS is influenced, which carries out the vasomotor control of the skin and acts on the cutaneous blood flow, therefore acting on the cutaneous microcirculation, including the intercellular and interstitial fluids of the lymphatic system, coinciding with the area where the patient complains of pain.15,20 Transcutaneous neuromodulation by TMP regulates lower limb vascularization 3 and modulates the ANS, 37 thus influencing the SNS through modulation of epidermal cells. The epidermis comes embryologically from the ectoderm-like brain; therefore, it acts directly on the CNS and any influence on the epidermis is a priority for the CNS. As Chao has already described, there is a strong connection of cutaneous nerves with the thalamus. 22
In Slomka’s study, vascularization increases after KT application without creating tension, after 4 days of lumbar application with movement involved, and without knowing the mechanism of action. 19 In Liu K’s study, there is only improvement in vascularization on fan application for several days. 26 Windisch et al. thermographically determined the temperature of the operated knee joint of patients for 7 days, and only the temperature of the lateral side of the wound was higher in the KT bandage group than in the control group. Thus, an increase in blood circulation can be assumed here. 29 In these studies, local vascularization increased, possibly due to the movement performed, and the tension created at these times. This increase in temperature in the lumbar area is not compared with another point distal to the one evaluated, so it is not possible to define whether there is an improvement in symptoms or a modulation of temperature. By contrast, Ptak et al. 27 found no significant differences in the application of KT on the abdominal musculature. Konieczny et al. analyzed changes in skin surface temperature after short-term application and removal of KT versus rigid tape without obtaining statistically significant changes. 45
KT tension is a key factor in the bandaging method; however, there is limited science regarding the effects of reinforced KT tension on functional performance and pain in healthy individuals. Lee et al. found no significant changes when applying KT from insertion to origin, with 75% lengthening after exercise. 24 Selva et al. concluded that the studies performed so far are not reproducible; therefore, their results are not reliable in that one reports that KT produces effects while the other reports that it does not. 31 According to Selva et al.’s study, 31 depending on the characteristics (body dimensions) and properties (skin elongation) of each subject in the sample, bandages with different elongations must be applied to achieve the same deformation in all tapes, therefore producing the same effect. The absence of these data at this time limits the reliability of previous clinical studies, makes it impossible to compare their findings, and presents new challenges for research in this field.
In the present study, to standardize the application and make it reproducible, transcutaneous neuromodulation by TMP was applied without creating tension, the patient was not asked to move, and the patient remained in the prone position during the entire session with stable and controlled room temperature. This was performed using a temperature measurement protocol. The temperature variations correspond to changes in the circulation of the dermal layer, which is controlled by the sympathetic neurovegetative system 8 that is activated by the epidermal cells.20,35,37 Li et al. concluded in a meta-analysis that KT cannot provide significantly greater pain relief to patients with chronic LBP compared to a sham/placebo control. 30 On the other hand, the application of TMP can be imposed as an easy application to reduce pain since the algometry went from 10 of NPRS in L5 withstanding 1.2 kg of pressure, which was the most painful level, to 5 of NPRS withstanding 3 kg of pressure after the application of TMP. After 23 years from the surgical intervention, the different temperatures in the back and the average temperature of the area of the scar were stabilized with an area distal to the scar in less than a minute and were maintained for 12 h. These rapid changes can be explained by the modulation of central pathways.
With respect to scar tissue, local vascularization is altered during the healing process, with the formation of new vessels. In fact, the newly developing granulation tissue undergoes angiogenesis. 10 The phases of the healing process are influenced by mechanical forces, and these forces have an important impact on the quality and speed of tissue repair. 10 Recent studies have shown that the application of external and oriented mechanical force could reduce collagen deposition during the healing phase. This process can be used to enhance tissue repair and recovery from injury. 10 Although there is a correlation between scar and LBP,5–7,46 it remains to be defined whether the pain comes from the epidermal scar, the fascial or muscular scar, or both. The embryologic origin of both is different and therefore, different pathways will be used. Our case confirms that the epidermal scar influences pain and temperature, so the epidermal scar should not be forgotten in the patient’s treatment.
Manual therapy increases the vascularization of the scar and the peri-scar area. This suggests a modification of connective tissue function after treatment 10 ; thus, manual therapy can modify fascial tensions by improving movement. However, in this case, it was counterproductive to improve movement without having sufficiently strong musculature to control such movement. According to our results, where only the epidermis has been influenced, we recommend performing epidermal scar modulation due to its ectodermal origin and, if necessary, manually or instrumentally treating the adhesions of the connective tissue embryologically originating from the mesoderm later. This is because the ectodermal epidermis was created first, followed by the mesodermal musculoskeletal structures. Therefore, transcutaneous neuromodulation by TMP could help to reduce neurogenic inflammation by helping to reduce perceived pain, and possibly acting on the other structures innervated by peripheral sensory neuron endings that are more likely to be involved in the production of inflammation, such as the skin, viscera, muscles, tendons, and joints. 15
Limitations and future directions
This study has several limitations that should be considered. First, as this is a case report, our preliminary results in a single patient should be interpreted with caution and cannot be generalized to all patients with scarring. In this regard, it would be interesting to perform a randomized controlled trial with a long-term intervention, although this type of study may be very difficult to perform because of the difficulty of finding patients with the combination of the surgical intervention performed and the pain latency time. Second, the metameric components of the lumbar scar area should be evaluated, such as hip muscle strength, joint mobility, and periarticular vascularization. Finally, the lack of opportunity to reevaluate the patient later than 2 months is recognized as a limitation of the present report.
Lessons learned
Transcutaneous neuromodulation of epidermal cells by TMP is a paradigm shift in the current treatment of pain and activation of the ANS. Epidermal scars should be considered to resolve or mitigate chronic pain by their direct central modulation.
Conclusions
Transcutaneous neuromodulation by TMP modulated the dermal body temperature of the back during the first minute of application and was maintained for 12 h. It also decreased the perceived pain after 30 min of application and maintained the improvement after 12 h. In addition, the kilogram of pressure supported by the patient was increased. It is postulated that transcutaneous neuromodulation by TMP helps to reduce neurogenic inflammation, helping to reduce perceived pain. Possibly, it acts on the rest of the local metameric structures such as the dermis, viscera, muscles, tendons, and joints by modulating the peripheral endings of the sensory neurons that are more likely to be involved in the production of pain. In addition, it is modulated at a systemic level by acting on epidermal cells innervated by the ANS. It is advisable to modulate the epidermal scar due to its ectodermal origin in patients with chronic pain, and subsequently treat musculoskeletal dysfunctions since its mesodermal embryological origin is posterior to the ectodermal structures.
Footnotes
Appendix A
Appendix B
Note: Failure to comply with any of these recommendations conditions the performance of the thermography, as it may be influenced by these factors.
Acknowledgements
We thank the study patient for making this possible.
Author contributions
F.S.S., Y.N.I., and J.M.P. contributed to conceptualization; F.S.S. and J.M.P. contributed to methodology; J.M.P. contributed to software; J.M.P. and E.A.S.R. contributed to validation; J.M.P. and E.A.S.R. contributed to formal analysis; Y.N.I. and J.M.P. contributed to the investigation; F.S.S. and J.M.P. contributed to resources; J.M.P. contributed to data curation; F.S.S., S.F.C., J.M.P., and E.A.S.R. contributed to writing—original draft preparation; F.S.S., S.F.C., J.M.P., and E.A.S.R. contributed to writing—review and editing; J.M.P. contributed to visualization; F.S.S., J.M.P., and E.A.S.R. contributed to supervision; F.S.S. contributed to project administration; and F.S.S. contributed to funding acquisition. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The data presented in this study are available on request from the corresponding authors. The data are not publicly available due to ethical restrictions.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors report a potential conflict of interest in the fact that FSS is the developer of the magnetic tape. The rest of the authors do not present any relationship of any kind with the brand.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the University of Valencia (protocol code 1118645, September 5, 2019).
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental Material
Video of the procedure: ![]() . The video shows the dermal body temperature variations induced by different tapes applied to a scar. It was observed that with TMP there was an initial reduction in temperature in the tissues adjacent to the scar, followed by a gradual increase. This thermal change was statistically significant and maintained its magnitude at 12 h post-application. By contrast, the application of the control tape induced hardly any alteration in skin temperature during the observation period.
. The video shows the dermal body temperature variations induced by different tapes applied to a scar. It was observed that with TMP there was an initial reduction in temperature in the tissues adjacent to the scar, followed by a gradual increase. This thermal change was statistically significant and maintained its magnitude at 12 h post-application. By contrast, the application of the control tape induced hardly any alteration in skin temperature during the observation period.
