Abstract
Valproic acid holds a prominent position as the primary therapeutic option for epilepsy cases. The utilization of antiepileptic agents like valproic acid has been linked to disturbances in sleep architecture. Although scant, there exists a subset of studies that allude to the potential manifestation of sleep apnea as an adverse effect of valproic acid administration. In this context, we present a case report of a 76-year-old man with a past medical history of arterial hypertension, atrial fibrillation, and obstructive sleep apnea syndrome treated with continuous positive airway pressure since 2020. Following the commencement of valproic acid treatment for vascular epilepsy, obstructive sleep apnea evolved toward to central sleep apnea. This case exemplifies the need for a heightened awareness of the multifaceted implications of valproic acid therapy on sleep patterns.
Introduction
Valproic acid (VA) stands as a preeminent therapeutic intervention extensively employed in the management of epilepsy across both the adult and pediatric populations. Robust evidence substantiates a functional interplay between epilepsy and sleep dynamics, as elucidated in prior research studies. 1
In light of this connection, it is noteworthy that antiepileptic agents, including VA, wield the potential to disrupt the intricate architecture of sleep patterns, as underscored by previous investigations. Nonetheless, the corpus of literature addressing the potential emergence of sleep apnea as a consequential offshoot of VA administration still needs to be explored.
The clinical implications inherent to sleep apnea syndrome are of paramount significance, encompassing an array of ramifications such as recurrent nocturnal arousals, inordinate daytime somnolence, and an escalated vulnerability to adverse cardiovascular sequelae ranging from cardiac failure and stroke to arrhythmias. 2 Anchoring our discourse within the framework of respiratory physiology, it is pertinent to acknowledge that gabapentinoids exert a modulatory influence upon γ-aminobutyric acid (GABA), thereby evoking a profile akin to that of baclofen in respiratory regulatory centers. Indeed, akin to opioids, baclofen has been well-documented for inducing a dampened respiratory drive, concurrently giving rise to central sleep apnea (CSA). 3 As we pivot to the realm of antiepileptic pharmacotherapy, the prominence of VA (VP) becomes apparent, encompassing a broad spectrum of utility extending beyond epilepsy management to encompass psychiatric disorders that exert an influence upon GABAergic signaling. Despite the conspicuous impact of antiepileptic compounds upon GABAergic tonicity, the intricate nexus between these pharmacological agents and the manifestation of CSA has regrettably remained an underexplored terrain. 4
Within this context, we herein report the case of a patient followed for obstructive sleep apnea, which evolved into CSA after the introduction of VA for vascular epilepsy. Case report: It is about a 76-year-old man with a past medical history of arterial hypertension, atrial fibrillation, and obstructive sleep apnea syndrome, treated successfully with continuous positive airway pressure (CPAP) since 2020. In November 2021, the patient had a hemorrhagic stroke complicated by a bilateral tonic-clonic epileptic seizure and has been treated with VA at 1000 mg/d from this date, with no further seizures. Five months later, the patient reported a worsening of his symptoms, such as apnea, startle awakening, which was not present before taking VA, and especially excessive daytime fatigue and sleepiness. The patient was referred to our department. Respiratory polygraphy (RP) was realized under his 9cmH2O CPAP. An expert sleep physician manually scored it and showed a residual apnea-hypopnea index (AHI) at 42 per hour with predominantly central events without Cheyne-Stokes breathing (Figure 1). Common causes of CSA, such as heart failure, neurological diseases, and opioid and alcohol consumption, have been eliminated. The other medications habitually taken by the patient are not known to belong to a pharmaceutical class that is associated with CSA. We did not detect hypercapnia in the blood gas assessment. After a concertation with his neurologist, VA has been switched to levetiracetam at 500 mg twice per day. The pressure of the CPAP was also decreased to 8cmH2O. At 3 months of checkups, the patient has noticed a significant clinical improvement: the cessation of startle awakenings and the improvement of daytime somnolence. The calculated Epworth score 5 was 3. RP has shown a residual AHI at 12/h with regression of central events (Figure 2).
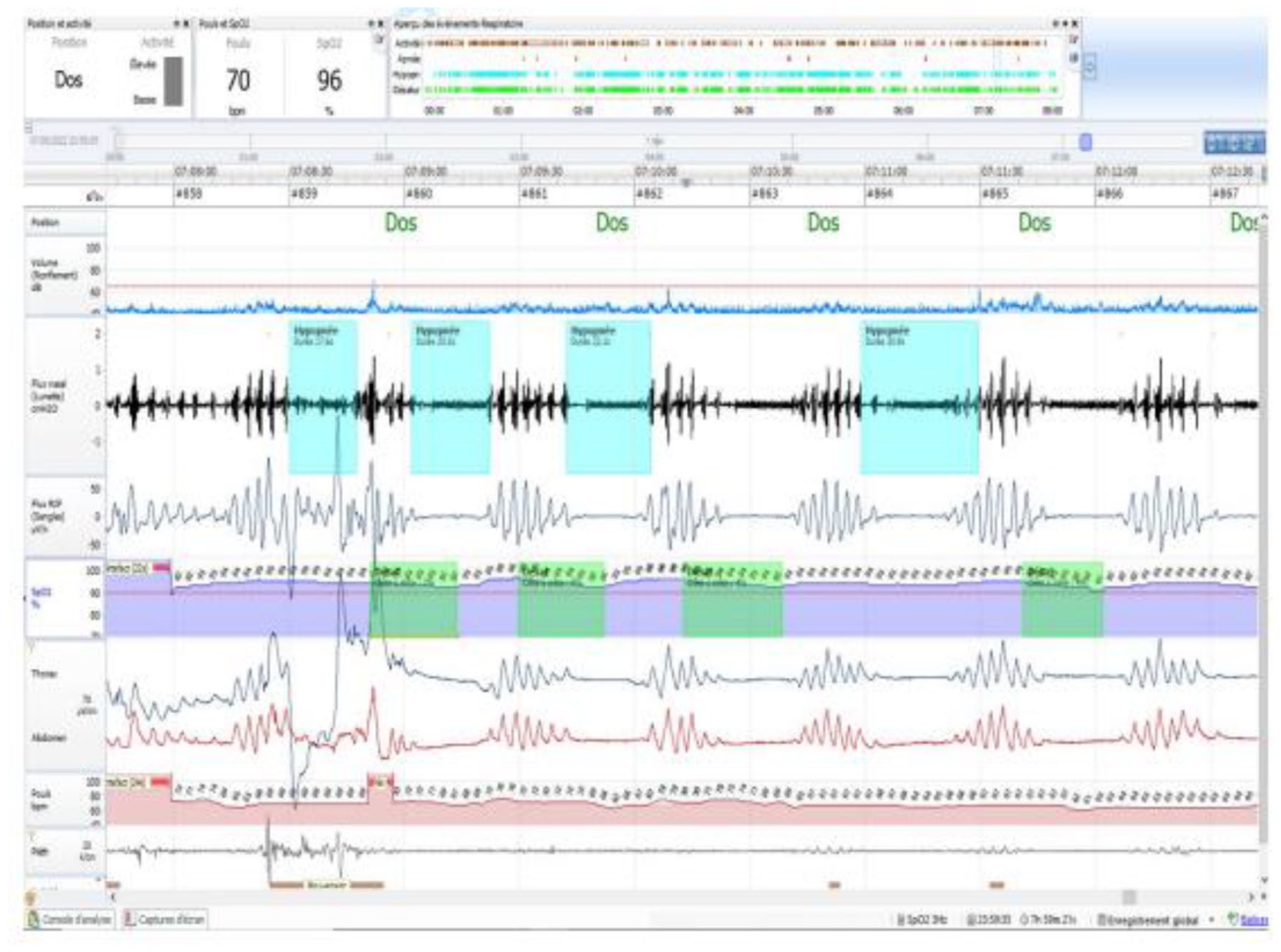
Respiratory polygraphy under valproic acid showing central events without Cheyne-Stokes breathing.

Respiratory polygraphy after valproic acid cessation showing regression of central events.
Discussion
CSA is characterized by repeated episodes of interruption or reduction of airflow as a consequence of a transitory pause or decrease in the central ventilatory drive during sleep. 6 CSA is less frequent in the general population than obstructive sleep apnea. It is generally associated with particular medical conditions. 7 Patients on opioid medication with congestive heart failure or stroke are the most likely to develop CAS. 6 It can also appear under CPAP treatment; this entity is called emergent CSA. 8 In our case, central respiratory events (CRE) appeared in RP lately after an improvement with CPAP treatment. We evoked stroke and iatrogenic causes in front of this clinical feature. Furthermore, in our case, CRE decreased after stopping VA, suggesting its causal role in inducing CSA. Two cases of CSA induced by VA were reported in the literature. 4 Another study was based on the World Health Organization’s global pharmacovigilance database. It has been shown that among the 72,821 adverse drug reactions reported by health professionals for VA, there were 56 cases of sleep apnea syndrome. 1 Two mechanisms are proposed to explain by which VA induces CSA. The first one is the depression of the central ventilatory drive. VA, similar to gabapentin, elevates GABA levels in the central nervous system, which induces a ‘baclofen-like’ effect on respiratory centers, leading to a lack of respiratory drive during sleep. 9 The second mechanism for CSA occurrence is that VA causes instability of ventilator control. 1 So, patients treated with VA should be screened for CSA. Given the importance of understanding the underlying mechanisms, sleep physicians and neurologists must collaborate closely when dealing with CSA. One priority should be exploring medications that do not disrupt GABA levels. It is worth mentioning that there are emerging agents, like lamotrigine and oxcarbazepine, that primarily act on voltage sodium channels. In addition, certain antiepileptics target receptors since seizure generation involves mediated excitation. These include compounds like felbamate and topiramate, which also inhibit sodium current. Zonisamide, which shares this mechanism, also helps reduce concentration. It is important to note that these new antiepileptic medications are associated with tolerability among epilepsy patients and generally have pharmacokinetic drug interactions. 10
Conclusion
In our situation, we carefully considered the effect of VA treatment on CRE based on existing research, the timing of events, and how they can be reversed after stopping treatment. By examining this case, we aim to emphasize the importance of being more alert to the possible connection between VA administration and the shift from obstructive sleep apnea to CSA. This will contribute to a better understanding of these intricate clinical dynamics.
Footnotes
Acknowledgements
Not Applicable.
Author contributions
C.M. revised the manuscript critically for important intellectual content N. M. wrote the manuscript H.R. analyzed the data and revised the manuscript critically for important intellectual content A.K. revised the manuscript critically for important intellectual content I.K. analyzed the data L.B. revised the manuscript critically for important intellectual content I.Z. validation S.M. conceived the project, validation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Written informed consent was obtained from the patient for the publication of the case report.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
