Abstract
Central airway obstruction often presents with respiratory symptoms identical to those of common respiratory diseases. Diagnosis of central airway obstruction is challenging in clinical practice, especially misdiagnosed as asthma or chronic obstructive pulmonary disease in case of “normal” chest X-ray. Here, we reported five central airway obstruction cases: the first and fourth cases misdiagnosed as asthma, the second masquerading as chronic obstructive pulmonary disease exacerbation, the third diagnosed incorrectly with non-resolving pneumonia, and the fifth misdiagnosed as chronic obstructive pulmonary disease. We then analyzed diagnostic clues potentially useful to differentiate central airway obstruction from asthma/chronic obstructive pulmonary disease. A multidisciplinary approach to manage central airway obstruction is essential, particularly selecting judiciously the method of respiratory support due to the high risk of completed airway collapse or obstruction during interventional period.
Introduction
Central airway obstruction (CAO) is defined as the occluding status occurring in the trachea, main bronchus, bronchus intermedius, or lobar bronchus presented with dyspnea, cough, or wheezing which requires medical attention. Its etiologies are diverse and could be classified as malignant (primary or metastatic cancer) and non-malignant (e.g., post-intubation, post-tracheostomy, post-tuberculosis, tracheomalacia, bronchomalacia, subglottic stenosis). 1 Its clinical presentation also varies and is affected by the reduction of cross-sectional area (CSA) at the narrowing position. In detail, patients with reduction of CSA ⩽50% could be asymptomatic, whereas those with reduction >71% present symptoms at rest or with mild exertion. 2 However, the reduction in CSA may not necessarily correspond to the clinical symptoms. This can be explained by inflammatory reactions and increased respiratory secretions that may be encountered in various clinical scenarios, such as infections and post-bronchoscopy procedures, which can further exacerbate pre-existing respiratory symptoms in patients with mild-to-moderate CAO.
Chest X-ray (CXR) and spirometry were reported not sensitive enough to detect CAO.1,3 In cases where patients cannot cooperate reasonably to perform forced expiratory efforts, the flow–volume (F–V) loop may appear abnormal and fail to detect CAO. 4 Additionally, depending on the patient’s position (upright or supine), the F–V loop may show abnormalities when there is external compression of the trachea due to goiters or anterior mediastinal masses. 4 Furthermore, the F–V loop may appear normal when the airway diameter is still above 8 mm. 5 Misdiagnosis of CAO as asthma/chronic obstructive pulmonary disease (COPD) results in challenges in clinical practice.6,7 Moreover, the proper management strategy requires personalized therapy ranging from conservation to medical or surgical interventions. 8 Herein, we described five CAO cases, two with non-malignant etiology misdiagnosed as asthma, one with malignant etiology masquerading as COPD exacerbation, one with malignant etiology misdiagnosed as non-resolving pneumonia, and one with malignant etiology misdiagnosed as COPD.
Case series
Case 1
A 54-year-old female patient presented to our outpatient clinic with a 15-month history of progressing dyspnea, wheezing, and occasional cough. At the initiation, her dyspnea only appeared with strenuous exertion. Because of the normal CXR (Figure 1(a)) and the result of spirometry with forced expiratory volume in 1 s (FEV1)/forced vital capacity (FVC) 0.76, she was diagnosed and treated as an asthmatic at another hospital during 1 year before seeking to our hospital. Her spirometry revealed FEV1 (1.61L), FVC (2.11L), and the negative reversibility. However, her dyspnea, predominantly inspiratory phase, worsened gradually even at mild activities. On the physical examination, wheeze was documented over both lung fields and loudest at the upper third of trachea. Re-evaluation of her spirometry revealed the blunting of both limbs of the F–V curve (Figure 1(b)). Suspecting tracheal obstruction, the cervical computed tomography (CT) with contrast material undertaken showed the subglottic stenosis (Figure 1(c) and (e)). Flexible bronchoscopy revealed the narrowness of trachea right below the glottis 1 cm (Figure 1(d)) and resulted in the episode of dyspnea immediately post-bronchoscopy. The otolaryngologist was consulted, and tracheostomy was conducted. Laboratory tests, including antinuclear antibodies, anti-double stranded DNA, and antineutrophil cytoplasmic antibodies, were negative. The laryngotracheal reconstructive surgery was conducted with the insertion of a Montgomery laryngeal stent. The histopathology of hypertrophic tissue causing tracheal stenosis showed chronic inflammation with cytokeratin (−), chromogranin (−), synaptophysin (−), Ki67 (+), and vimentin (+). The final diagnosis was idiopathic subglottic stenosis (iSGS). The patient had the Montgomery laryngeal stent removed after 3 months, and there were no signs of recurrence after 6 months of follow-up.

Central airway obstruction due to idiopathic subglottic stenosis. (a) Chest X-ray is normal. (b) Flow–volume curve shows the blunting pattern resembling a rectangle. (c) Cervical computed tomography (CT) in the sagittal plane reveals the narrow position of trachea (red arrow). (d) Bronchoscopic images with mucous edema and obstruction right below the glottis (red arrow). (e) Cervical CT in the axial plane reveals the narrow position of trachea (red arrow).
Case 2
A 51-year-old male patient with 30 pack-year history of smoking hospitalized because of dyspnea. He initiated dry cough and mild dyspnea on exertion 3 months ago. His dyspnea attacks occurred occasionally, and he was diagnosed and treated with COPD exacerbation at a provincial hospital. His CXR showed normal lung fields accompanied by the mediastinal bulging (Figure 2(a)). However, his episodic breathlessness worsened with cyanosis, and he was transferred to our hospital. Here, he continued being diagnosed with COPD exacerbation. Without the attack of dyspnea, he showed alert, breathing slightly, and bilateral breath sounds clearly on auscultation. During the attack of dyspnea, he showed agitation, cyanosis, severe respiratory muscle contraction, and an almost silent chest. During the period of hospital stay, he developed hemoptysis, which triggered the severe attack of dyspnea with cardiac arrest. The chest CT undertaken revealed the mediastinal tumor invasive into the lower third of trachea right above the carina (Figure 2(b)). Re-evaluation of CXR identified respectively the narrowness of trachea which had been missed at the initiated evaluation. Diagnosis of CAO with mediastinal tumor was established, and the thoracic surgeon was consulted to resolve it with possible support of extracorporeal membrane oxygenation (ECMO). However, his family refused further intervention and desired his discharge. He deceased after that.
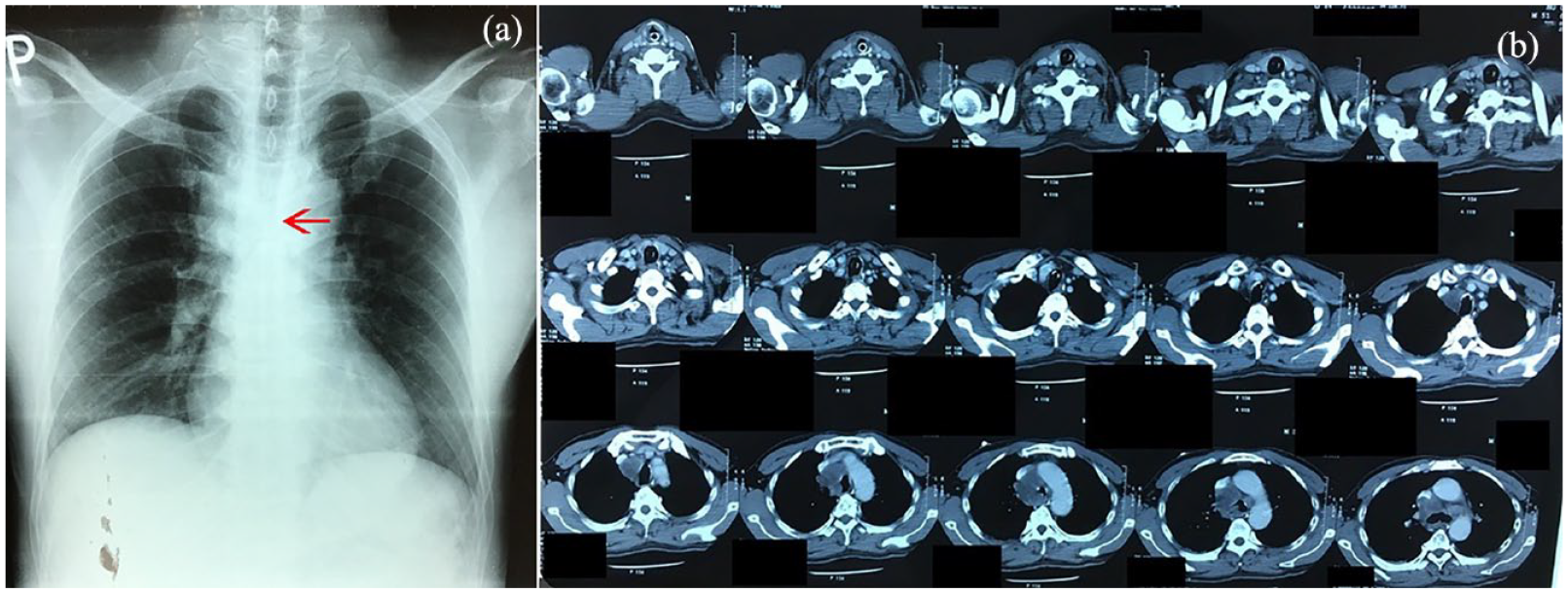
Central airway obstruction at the inferior limit of trachea. (a) Chest X-ray revealed normal lung fields but suspecting the loss of patency at the lower limit of trachea (red arrow). (b) Chest computed tomography reveals the mediastinal tumor invasive into the trachea.
Case 3
A 30-year-old non-smoker male patient with a 2-year history of gout, was hospitalized due to productive cough, dyspnea, and infectious ulceration of tophi nodule on his right foot. He experienced productive cough and episodic breathlessness over the past 2 months before admission. He was treated for 4 days at a provincial hospital but did not improve, and he was transferred to our hospital. On the physical examination at our department, wheezing was documented in both lung fields. His CXR revealed bilateral alveolar infiltrates predominant in the upper lobes (Figure 3(a)). The initial diagnosis was a combination of non-resolving pneumonia, asthma, and infectious ulceration of tophi. Treatment with cefoperazone/sulbactam, teicoplanin, and levofloxacin was initiated, but his respiratory failure worsened after 5 days of treatment leading to him being intubated. Meropenem and colistin were changed when the multidrug-resistant Acinetobacter baumannii was detected via sputum and pus cultures. During mechanical ventilation, the airway pressure was still high despite changing the endotracheal tube and suctioning secretions. Arterial blood gas analysis revealed severe respiratory acidosis (pH 7.1 and PaCO2 65 mmHg). Suspecting an airway obstruction, the bedside flexible bronchoscopy undertaken revealed tumorous lesions at the lower third of trachea and in both main bronchi (Figure 3(b)) (histopathological result was lung adenocarcinoma). Six hours later, the patient developed severe hypoxemia, refractory respiratory acidosis, and hypotension, eventually dying from a combination of these factors. The patient’s condition rapidly deteriorated within 3 days of admission to the intensive care unit.
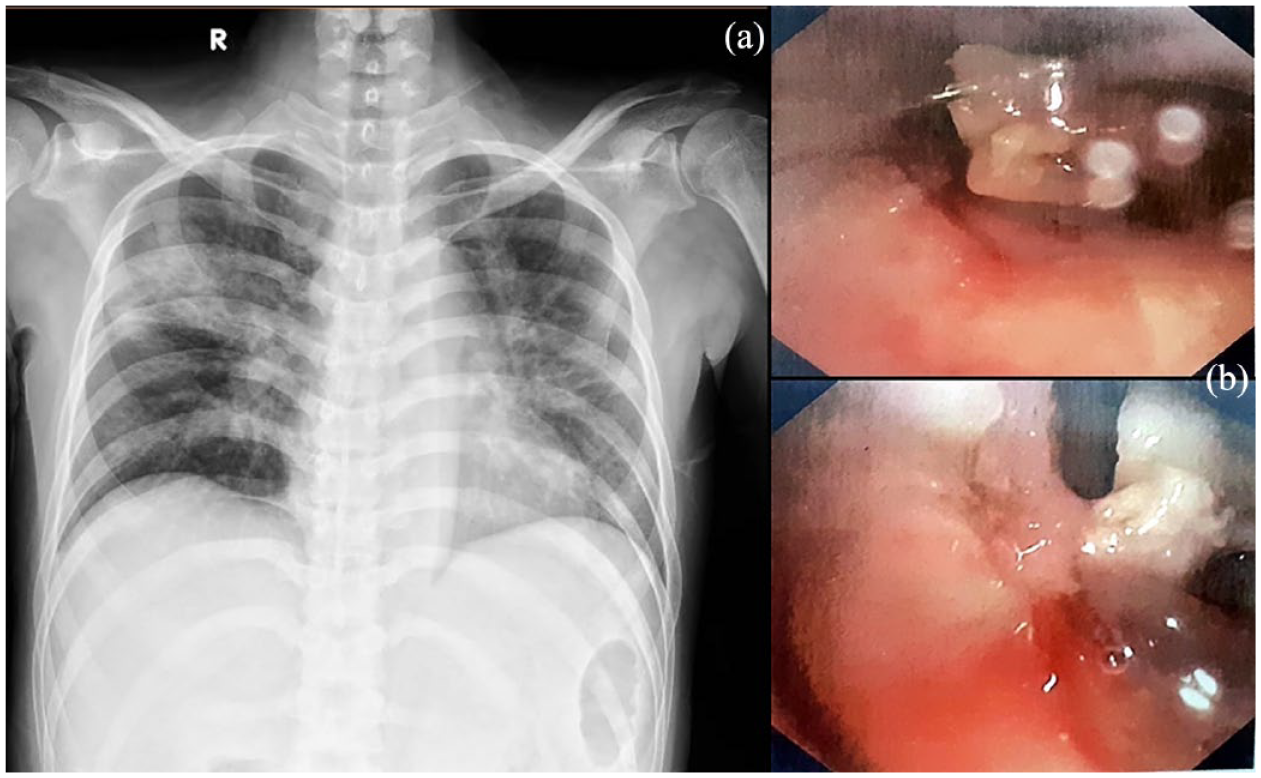
Non-resolving pneumonia relating to central airway obstruction. (a) Chest X-ray revealed bilateral alveolar infiltrates. (b) Bronchoscopic images showed tumorous lesions causing airway obstruction at the lower part of trachea.
Case 4
A 40-year-old non-smoker male patient presented at our respiratory outpatient clinic with a 1-month history of wheezing, exertional dyspnea, and cough. He was diagnosed and treated with asthma but did not improve. Two months ago, he had pesticide self-poisoning but survived after 10 days of mechanical ventilation. His past medical history was unremarkable. On physical examination, his pulse 70 beats/min, blood pressure 130/80 mmHg, respiratory rate 18/min, peripheral capillary oxygen saturation (SpO2) 99% with ambient air, and he breathed normally exception to wheezing loudest at cervical area radiating to two lung fields on auscultation. His CXR was normal, and the result of spirometry showed the pattern of fixed upper airway obstruction in F–V loop (Figure 4(a)). His chest CT scan showed tracheal stenosis (smallest inner diameter of 4 mm) at the level of cervical (C7) and thoracic (T1) vertebra (Figure 4(b) and 4(c)). After the diagnosis of post-intubation tracheal stenosis, the otolaryngologist was consulted, and the patient was indicated to undergo rigid bronchoscopic dilation. His condition improved after the procedure, and there was no recurrence after 5 months of follow-up.

Central airway obstruction due to post-intubation tracheal stenosis. (a) Flow–volume loop showed the pattern of fixed upper airway obstruction. (b) and (c) The images of chest computed tomography in axial and sagittal planes reveal the tracheal stenosis (red arrows).
Case 5
A 60-year-old male patient with a 40 pack-year history of smoking presented at the respiratory outpatient clinic because of exertional dyspnea and hoarseness for 1 month. He was diagnosed with COPD, but his condition did not improve with bronchodilator. He did not complain about difficulty swallowing even when asked about it. His CXR seemed normal at the initial evaluation and rigid laryngoscopy revealed reducing the mobility of right vocal cord. However, the narrowing of the trachea on CXR could be identified with the caution of evaluation (Figure 5(a)). The result of spirometry showed the F–V loop with the pattern of variable intrathoracic obstruction (Figure 5(b)). Suspecting mediastinal tumor, chest CT scan with contrast material undertaken showed the mass (43 × 42 mm) compressing and pushing the trachea (Figure 5(c)). Upper gastrointestinal endoscopy with biopsy was done to define the final diagnosis of esophageal squamous cell carcinoma. The oncologist and the thoracic surgeon were consulted, identifying the high risk of airway collapse when general anesthesia. He has been treated with chemotherapy and if tumor did not respond, would proceed to surgery under ECMO if required. Three months of follow-up, he showed improvement with chemotherapy.

Esophageal squamous cell carcinoma causing central airway obstruction. (a) Chest X-ray with the narrowing of trachea (red arrow). (b) Flow–volume loop with the pattern of variable intrathoracic obstruction. (c) The esophageal tumor compressing and pushing the trachea.
Discussion
Dyspnea and wheezing are common in many respiratory diseases including CAO. Clinical manifestation of CAO varies by the narrowing severity and the airflow velocity 2 and includes symptoms mimicking breathlessness of asthmatics, exertional progressive dyspnea comparable to that of COPD patients, or dyspnea attacks like asthma/COPD exacerbations. Thus, CAO could be potentially misdiagnosed as asthma or COPD. The first and fourth cases were misdiagnosed with asthma because of exertional dyspnea and normal CXR. Likewise, the second case was misdiagnosed with COPD exacerbation because of the attack of dyspnea and CXR without parenchymal abnormality. On the other hand, the asthma was misdiagnosed in the third case because of young age (30 years old), episodic dyspnea, and wheezing on auscultation. The last case was misdiagnosed as COPD because of a 40 pack-year history of smoking, exertional dyspnea, and normal CXR. However, several clinical features including inspiratory breathlessness, wheezing predominant in cervical or sternum area, or accompanied hemoptysis could be important clues to distinguish with asthma/COPD. A high index of suspicion combined with holistic evaluation is required to avoid the omission of CAO.
Previous published studies noticed findings suspecting CAO on the F–V curve including the flattening pattern in patients with tracheal stenosis and the biphasic pattern on the expiratory limb in those with unilateral main bronchial obstruction.7,9 The first and fourth cases revealed the blunting pattern of F–V curve resembling a rectangle which was missed at the initial evaluation. The last case showed the pattern of variable intrathoracic obstruction. Increased recognition for the obstructing patterns of the F–V curve is essential in detecting CAO. In contrast to the useful role of spirometry in diagnosing CAO, the procedure of spirometry could result in severe respiratory failure which should be taken precaution in clinical practice. 1
CXR was insensitive to detect CAO, especially in cases misdiagnosed with asthma/COPD. 1 However, evaluating the patency of both trachea and main bronchi should be considered to avoid the missed finding on CXR similar with the second and fifth cases. Chest CT has been more effective than CXR and given more information relating to obstruction such as location, extension, severity, and morphology.1,2 Despite the noninvasive advantages of radiologic diagnosis, comparing to bronchoscopic evaluation Kassem Harris et al. 10 revealed 31% of undetected CAO on chest CT images. Bronchoscopy has still been the mainstay in both diagnosis and therapeutic intervention. On the other hand, the risk of bronchoscopy causing respiratory failure due to airway obstruction among CAO patients should be evaluated carefully. 1 Our first case fell into severe respiratory failure requiring post-bronchoscopy tracheostomy immediately. Therefore, a trained and experienced physician has been recommended to conduct this intervention according to international guideline. 8
Lung cancer may manifest with insidious symptoms of CAO leading to initial incorrect diagnoses in our second and third cases (COPD/asthma), and the malignant CAO itself could result in complicated progression in disease, such as accompanied hemoptysis or non-resolving pneumonia. 8 The study by Cyrus Daneshvar et al. 11 revealed that approximately one-third of CAO among newly diagnosed lung cancer cases failed to detect during the initial chest CT scan result, which could contribute misdiagnosis in clinical practice. Besides, whether CAO contributing to lung cancer mortality is unknown, published studies showed mixed results. It was uncontroversial that the role of CAO to worsen pneumonia was important in our third case. Management of CAO becomes more complex because of firstly dealing with its complications.
Management of CAO including bronchoscopic intervention or surgery aims mainly to restore the airway patency. 2 However, the location of airway obstruction occurred at the two limits of the trachea (superior relating to cricoid cartilage and inferior relating to tracheal bifurcation) which constituted challenges for therapeutic intervention. For instance, it is very difficult to ensure ventilation when conducting any invasive procedure in the second, third, and fifth cases because of high risk of completed airway collapse or occlusion leading to respiratory failure. The potential role of ECMO, which plays as a safe bridge to interventional therapy, was reported in similar case. 12
Although CAO patients are often encountered by pulmonologists, a multidisciplinary approach to treat them is necessary. The otolaryngologist showed the contribution mainly in management of our first and fourth cases. The insertion of Montgomery laryngeal stent was indicated for the first case after tracheotomy to resolve dyspnea. The iSGS is a rarely non-malignant etiology causing CAO. 13 To our best knowledge, this is the first case report in Vietnam. Long-term follow-up is required for patients with iSGS because of high recurrence rates. 13
Conclusion
CAO should be considered as a differential diagnosis of asthma or COPD (stable and exacerbation) because of the similarity of common respiratory symptoms. Both the holistic evaluation of clinical scenario and the high index of suspicion are required to avoid the omission of diagnostic clues. Its therapeutic strategy could vary between individuals and require a multidisciplinary team to plan from respiratory support to suitable interventions.
Footnotes
Acknowledgements
We thank the patients and their families for giving permission to publish this case series.
Author contributions
L.N-H., K.N-D., B.L-K., N.N.-T., N.D-M., T-H.T.-N., and N.V-H contributed to conceptualization; B.L-K., K.N-D., N.N-T., and N. D-M. contributed to formal analysis; L.N-H, K.N.-D., B.L-K, T-H.T-N., and N.V-H. contributed to investigation; L.N-H, K.N.-D., B.L-K, and N.N-T. contributed to methodology; L.N-H, K.N.-D., B.L-K, N.N-T, N.D-M, T-H.T-N., and N.V-H. contributed to writing—original draft.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the legally authorized representatives of the subjects in case 2 and case 3 and from the subjects themselves in case 1, case 4, and case 5 for the publication of the case series.
