Abstract
We report the anesthetic management of a case of paraganglioma, which was challenging because of intraoperative circulatory changes and difficult ventilatory management. A 52-year-old man with a history of chronic obstructive pulmonary disease was scheduled for paraganglioma resection under general anesthesia combined with epidural anesthesia. Since a marked increase in blood pressure was observed immediately after administration of rocuronium, antihypertensive agents were administered as necessary. The ventilatory settings were initially adjusted to deliver a tidal volume of 7 mL/kg, and the drive pressure was maintained at 13 cm H2O or less. However, despite increasing the minute volume, PETCO2 increased to 60 mmHg and PaCO2 to 76 mmHg before tumor removal. Blood pressure decreased immediately after tumor removal, and PETCO2 and PaCO2 gradually returned to normal. We speculated that the increases in PETCO2 and PaCO2 might have been due to both an increase in endogenous catecholamine secretion as well as chronic obstructive pulmonary disease. It is important to preoperatively evaluate the functionality of the tumor and to anticipate perioperative cardiorespiratory instability in the management of paragangliomas.
Introduction
Adrenal medullary cell-like tumors originating in the chromophilic tissue of the sympathetic ganglia are called paragangliomas, and approximately 85% of them are pheochromocytomas derived from the adrenal glands. 1 Paragangliomas can be found from the cranium to the pelvic cavity but are only rarely observed in the vicinity of the pancreatic head. 2 Generally, perioperative management of paragangliomas is identical to that of pheochromocytoma. During anesthetic management of pheochromocytoma, it is difficult to control circulatory dynamics. 3 We report a case of asymptomatic paraganglioma close to the pancreatic head, in which intraoperative respiratory and circulatory management was difficult due to catecholamine release resulting from surgical manipulation or anesthetic management. We obtained written informed consent from the patient to publish this case report.
Case presentation
The patient was a 52-year-old man, weighing 66 kg, with a height of 182 cm, a history of chronic obstructive pulmonary disease (COPD), and who had smoked 50 cigarettes daily for the last 40 years. He was referred to our hospital because of lumbar back pain and dye accumulation in the lymph nodes of the hepatic hilum on positron emission tomography–computed tomography (PET-CT) imaging in a medical examination. Since paraganglioma was suspected based on close preoperative investigation, we decided to perform surgical excision of the tumor. Abdominal CT scan revealed a tumor 55 mm in diameter with contrast effect, bordering the dorsal surface of the pancreatic head and extending to the anterior surface of the inferior vena cava (Figure 1). Multiple emphysematous bullae on chest X-ray and CT scan, the largest of which was 54 mm in diameter (Figure 2(a) and (b)). Laboratory findings on admission showed high blood noradrenaline and dopamine levels of 7856 and 85 pg/mL, respectively. Urinalysis showed urinary noradrenaline and blood dopamine levels of 1650 and 1537 μg/day, respectively. However, his blood pressure was normal, and he had no subjective symptoms. Paraganglioma is known as a familial disease; however, the paternal family had no known history of paraganglioma. So preoperative gene identification was not performed. The patient was scheduled to undergo laparoscopically. Therefore, preoperative antihypertensive therapy with intravascular volume correction was omitted. The combined epidural-general anesthesia technique was planned for facilitating hemodynamic stability before tumor isolation and reduces pain. We prepared for intravenous phentolamine, continuous infusion of nicardipine and landiolol in case of potential intraoperative crisis pertaining to catecholamine surge during surgical manipulation.

Abdominal CT. The tumor was located behind the pancreatic head and in contact with the anterior surface of the inferior vena cava (⇧). The tumor was 55 mm in diameter with contrast effect.
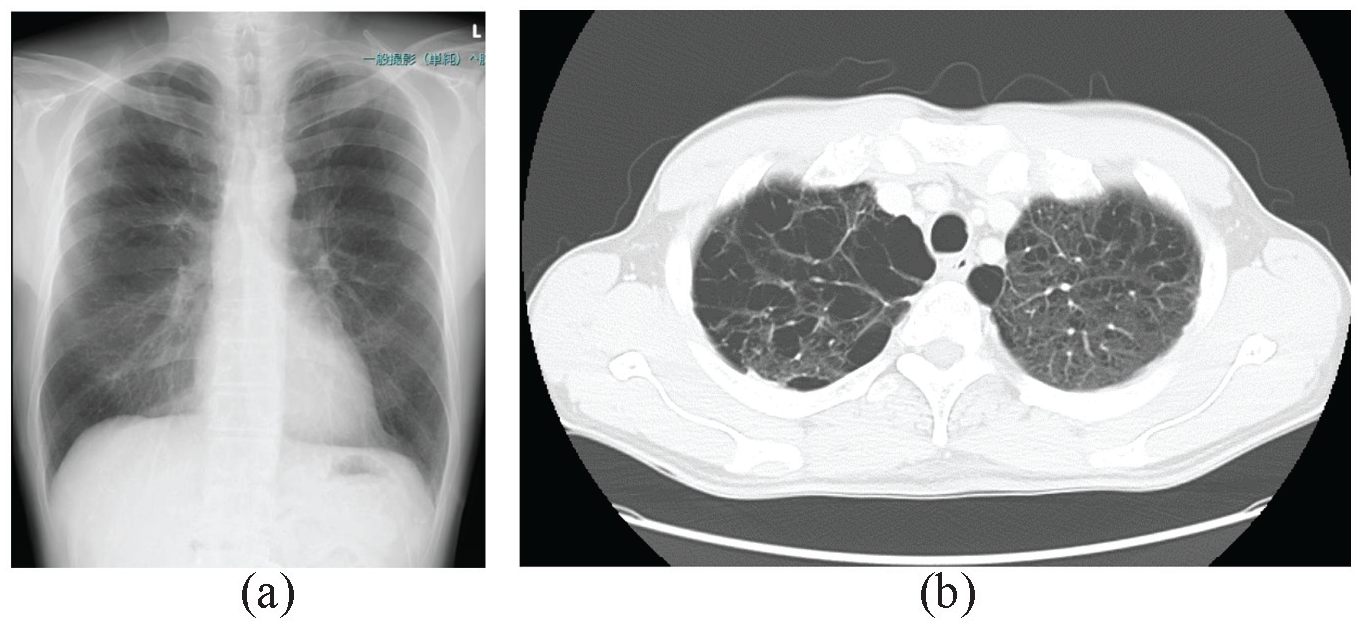
Chest X-ray and chest computed tomography (CT). (a) Chest X-ray showed slightly increased permeability in both lung fields. (b) Chest CT showed emphysema in the upper lung fields.
On the day of surgery, in the operating room, standard monitoring, including electrocardiography, pulse oximetry, capnography, noninvasive blood pressure measurement, and bladder temperature were performed, and anesthetic depth using patient state index (PSI) was monitored with Masimo SedLine® electroencephalogram (EEG) sensor (Masimo Co., Irvine, CA, USA). 4 Arterial catheter was inserted in the radial artery for monitoring intra-arterial blood pressure and measuring arterial blood gas, blood glucose or lactate. An epidural catheter was inserted via the Th 9-10 interspace, and 3 mL of 1% lidocaine with 1:100,000 adrenaline was injected. Next, the cold test was performed, and after confirming no reduction in cold sensation at the Th 9-10 level and no aberration in blood vessels and the subarachnoid space, general anesthesia was induced. Anesthesia was induced with 140 mg propofol, 6% desflurane, and 50 μg fentanyl, and 50 mg rocuronium was administered for muscle relaxation. However, his systolic blood pressure increased rapidly to 190 mmHg immediately after induction of anesthesia. Therefore, 1 mg nicardipine was injected. Tracheal intubation was performed after confirming the normalization of blood pressure. Anesthesia was maintained with desflurane inhalation so as to keep PSI levels between 25 and 50. In addition, remifentanil was administered for analgesia at a dose of 0.2–0.4 μg/kg/min, and rocuronium was administered as a continuous infusion of 20 mg/h. During surgery, abnormal increases in blood pressure or tachycardia were often induced by peritumoral manipulation or vascular processing, which were managed by the administration of nicardipine and phentolamine, and landiolol, respectively (Figure 3).

Anesthesia record.
After commencing laparoscopy, the surgeons found that laparoscopic procedure was complicated and decided that the tumor excision was converted via a laparotomy. Intraoperatively, since the tumor was adherent to the head of the pancreas and was difficult to dissect from the surrounding tissues, the operative procedure was changed to combined resection of the tumor and the pancreatic head.
Regarding the respiratory management, since the patient had COPD with several large emphysematous bullae, volume control ventilation with a tidal volume of 6 mL/kg and positive end-expiratory pressure of 4 cm H2O was commenced, and tidal volume and respiratory rate were adjusted so that the drive pressure did not exceed 13 cm H2O. Since an adequate minute ventilation was ensured, his initial PETCO2 and PaCO2 levels were 46 and 50 mmHg, respectively, with a respiratory rate of 8 breaths per minute and a tidal volume of 500 mL. However, the patient subsequently developed progressive hypercapnia despite increasing his minute volume. Before tumor removal, arterial blood gas analysis showed: pH: 7.18, PCO2: 76.5 mmHg, PO2: 111 mmHg, HCO3−: 28.5 mmol/L, and lactate: 2.34 mmol/L. His body temperature was 36.5°C just after induction of anesthesia, although it gradually increased and remained between 37.5°C and 37.8°C despite surface cooling. The cause of hypercapnia was considered to be hypermetabolism due to endogenous catecholamine production and obstructive ventilatory impairment.
Thereafter, since peritumoral vascular dissection followed by removal of the tumor resulted in a marked decrease in systolic blood pressure to around 60 mmHg, noradrenaline 0.1–0.2 μg/kg/min was administered to maintain blood pressure. Fluid infusion volume during the operation was approximately 3500 mL. Although the patient had restricted ventilatory settings, hypercapnia and acidemia gradually improved after tumor removal. He did not develop hypoglycemia after removal of the tumor. Lactate levels, which had increased to 2.4 mmol/L just before tumor removal, also returned to normal by the end of surgery. The total amount of fluid infusion was 6655 mL, total blood loss was 723 mL, and total urine volume was 2450 mL. Total operation time was 8 h 53 min, and total anesthesia time was 10 h 40 min. The pathological diagnosis of the retroperitoneal mass removed was paraganglioma.
Postoperatively, administration of noradrenaline 0.1 μg/kg/min was required on his admission to the intensive care unit (ICU). However, the patient was extubated immediately after surgery as his respiratory parameters had improved by this time. Oxygen 4 L/min was administered via a face mask, and arterial blood gas analysis indicated: pH: 7.358, PaCO2: 48 mmHg, and PaO2: 148 mmHg. Postoperative analgesia was provided by continuous infusion of 0.185% levobupivacaine at the rate of 5 mL/h through the epidural catheter. Blood glucose levels were maintained within normal limits. On the second postoperative day, catecholamine administration was discontinued, and the patient was transferred out of the ICU. There were no respiratory or circulatory complications thereafter.
Discussion
Paraganglioma is classified as functional and non-functional based on the secretory activity of catecholamines. 2 The disease only rarely remains clinically asymptomatic. In this case, the patient was considered to have non-functional paraganglioma because of the lack of clinical symptoms. However, since his blood pressure increased after induction of anesthesia, strict antihypertensive management was required intraoperatively. According to previous reports, both tumor size and degree of catecholamine production correlate strongly with intraoperative hemodynamic instability. 5 It has also been shown that the risk of systolic blood pressure exceeding 200 mmHg is higher in open than in laparoscopic surgeries. 6 In our case, given the large tumor size of 55 mm and the fact that he underwent open surgery, he was considered to be a high-risk candidate for abnormal hypertension, as reported in the previous literature. In non-functional pheochromocytomas or paragangliomas, preoperative antihypertensive treatment using α-blockers reportedly does not contribute to intraoperative blood pressure control, 7 although another report states that it is necessary to control preoperative blood pressure with α-blockers, even in patients with normal blood pressure. 8 We believe that it is important to administer blood pressure medication preoperatively in order to prevent unexpected blood pressure elevations during surgery. The Clinical Practice Guidelines of the Endocrine Society in the United States of America recommend that all patients with hormonally functional pheochromocytoma receive preoperative α-adrenergic receptor blockers for 7–14 days. 2 It appeared that all patients should receive preoperative α-blockers if they had elevated blood and urinary catecholamine levels. In addition, the failure to administer α-blockers, even in asymptomatic patients, may have affected the circulatory variables. In pheochromocytoma, although the stimuli of intubation or tumor handling are the typical triggers of blood pressure elevation, in our case, the patient’s blood pressure was elevated following administration of sedative drugs during anesthesia induction and after the administration of muscle relaxants. There are several previous reports of blood pressure elevation after rocuronium administration in the anesthetic management of pheochromocytoma, 9 and the mechanism of this phenomenon has been suggested to be the enhancement of noradrenaline release from the sympathetic nervous system by rocuronium.10,11
Paraganglioma also leads to abnormal glucose homeostasis caused by catecholamine release. Increased catecholamine levels lead to preoperative hyperglycemia due to increased glycogenolysis in the liver, inhibition of insulin secretion from pancreatic cells, and increased insulin resistance in skeletal muscles. 12 In addition, decrease in catecholamine levels after tumor excision can cause postoperative hypoglycemia. 13 In this case, hyperglycemia was not recognized preoperatively, and hypoglycemia did not occur after tumor extraction.
Since this case was complicated by COPD with large bullae, the tidal volume and the drive pressure were limited to 7 mL/kg and 13 cm H2O, respectively, although this resulted in severe acidemia due to hypercapnia. The cause of hypercapnia was thought to be elevated catecholamine levels or alveolar hypoventilation. However, the minute volume was maintained at a sufficient amount. Therefore, catecholamine levels should have been measured in this case. Albeit in surgeries requiring creation of a pneumoperitoneum, hypercapnia is known to be able to stimulate the release of adrenaline and noradrenaline. 14 Our experience suggests that preoperative hypercapnia should be avoided as much as possible even in cases of open abdominal surgery. Although there are no previous reports on the anesthetic management of open abdominal surgery for pheochromocytoma in patients with concomitant COPD with hypercapnia, the present case suggests that the respiratory management of such patients is formidable. After tumor removal, the hypercapnia in this patient rapidly improved, suggesting that increased carbon dioxide production due to hypermetabolism played a major role in the respiratory acidosis.
Conclusion
Anesthesia management is extremely important during the surgical treatment of paraganglioma, and the general management should be based on the preoperative endocrine assessment as well as the intraoperative respiratory and circulatory changes. In particular, the perioperative care of patients complicated with COPD should be carefully managed.
Footnotes
Acknowledgements
The authors thank Forte Science Communications, Tokyo, Japan, for English language editing.
Author contributions
S.S. and K.N. contributed to the anesthesia management of the patient, conceptualization of the case report, and writing of the original draft. Y.I. and K.N. edited the manuscript. H.U. was the overall supervisor of this case. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
