Abstract
Primary cardiac tumours are rare and most of them are benign. Myxomas, fibroelastomas and lipomas are common in adults. Primary valvular cardiac tumours are even more rare and affect all four valves in a similar proportion. Valvular lipomas are very rare. In the pulmonary valve there is only one described. Lipomas can be spindle-cell varieties. But of these, there is only one described in a valve, and it is placed in the aortic valve. Pulmonary valve lipomas can produce obstruction to the right ventricular outflow tract as well as pulmonary valve regurgitation, or pulmonary embolism. Symptoms may be dyspnoea, angina, arrhythmias, or syncope. We aim to illustrate with this case report how we came into this very rare pathology, so we present a 54-year-old woman with a giant spindle-cell lipoma located in the anterior pulmonary leaflet and severe dyspnoea. Total resection of the tumour was performed and restoration of valve function was obtained by means of bicuspidization of the remaining pulmonary leaflets. She had a good recovery after surgery and no complication during the postoperative evolution, being discharged from hospital after 7 days from surgery, with echocardiographic control showing good biventricular function, absence of tumour or obstruction, and minimal pulmonary valve regurgitation.
Introduction
Cardiac tumours are less than 1% of all tumours.1,2 Most of them are metastatic. 2 We must exclude this adverse possibility in any case of cardiac tumour diagnosis. 2 Primary cardiac tumours are rare and most of them are benign. 1 Primary cardiac valve tumours affect all four valves and they are found in less than 4% of all primary cardiac tumours. 3 The pulmonary valve is the least affected by primary cardiac tumours. 4 Myxomas are the most common benign cardiac tumours in adults, but its valvular location is rare. 1 Cardiac papillary fibroelastoma (CPF) is the most common tumour of the heart valves, being aortic valve leaflet location about half of them and the pulmonary valve location the less frequent.1,4 However, CPF are the most common tumours of the pulmonary valve.4,5 Primary tumours of the right ventricular outflow tract (RVOT) are very rare, 6 most of them are rhabdomyomas in babies and myxomas in adults.7,8 Among lipomas, cardiac lipomas are rare. 9 The frequency of cardiac lipomas is between 0.3% and 8.4%.1,10 Cardiac lipomas located on cardiac valves are rare.11,12 They are usually intramural or epicardial in location.13,14 Pulmonary valve tumours can cause either mechanical obstruction or valve regurgitation. 10 Both can cause severe right ventricular failure or syncope. 15 Spindle-cell lipomas are a relatively infrequent variant of benign lipomatous soft tissue tumour, especially infrequent in the heart.16,17 In many cases, symptoms are usually due to embolization of a part of the tumour or cloths that can grow over the tumour.18,19 They can also be due to failure of valve closure, valve obstruction, compression or displacement of the coronary arteries, alterations over the cardiac electrical system, or occupation of one or more of the cardiac chambers. From this we can expect symptoms like dyspnoea, angina, arrhythmias or syncope from congestive heart failure, pulmonary embolization and heart rhythm disturbances.18,19 Sometimes stemming from constitutional symptoms such as fever, weight loss or fatigue, and immune manifestations of myalgia, weakness and arthralgia may accompany the above symptoms and the outcomes may change after tumour resection.18,19 Infection occurs less frequently. A pathologically benign tumour can have a clinical malignant behaviour if it causes serious complications or death.15,20 The Gold Standard for diagnosis of cardiac masses is echocardiography, mainly transesophageal. 4 CT and MRI are useful to determinate the possible aetiology, diagnose pulmonary pathology and are helpful when establishing a strategy for intervention. 21 To have the final diagnosis of the type of tumour, it is mandatory to conduct an anatomopathology exam. We must not forget to exclude the tumour malignancy or metastatic. The possibility of recurrence is very low in benign primitive cardiac tumours.3,19
Case report
To illustrate a very uncommon situation but with a very frequent symptom, we present a 54-year-old woman with a history of type-2 diabetes mellitus and high blood pressure with left ventricular hypertrophy. A week before going to the cardiologist, she presented severe dyspnoea without paroxysmal nocturnal dyspnoea, orthopnea, chest pain or syncope. She recognized palpitations in the last year. She had no fever or cough or any other respiratory symptoms.
At the physical examination, she had moderate obesity and large breasts, no palpable precordial thrill, no organomegaly and a large nodule in the right thyroid lobe.
Cardiac auscultation showed a grade IV/VI holosystolic murmur in the aortic focus, not irradiated to the neck or axillae, followed by a slight proto-mesodiastolic murmur. Lung and neurological system exam were normal. Standard laboratory analysis was normal. Electrocardiogram: sinus rhythm, ventricular hypertrophy and incomplete right bundle branch block. RT-PCR for SARS-CoV-2 (COVID-19) was negative. Three hemocultures of peripheral blood were negative.
The transthoracic echocardiogram showed an echo-dense image in the RVOT, initially interpreted as a thrombus, but transesophageal echocardiography was recommended and showed an echo-dense mass of 2.5 cm, rounded and with clear limits, with an anchoring in the anterior face of the RVOT and that protruded in each systole to the pulmonary artery trunk, regressing in the diastole to the RVOT, with negligible valve regurgitation (Figure 1(a)). With this finding the diagnosis changed to a possible myxoma in RVOT.

(a) Transesophageal echocardiography showing the right ventricular outflow tract and the trunk of the pulmonary artery with a tumour pedunculated on the pulmonary valve. (b) Cardiac MRI T1 with contrast. (c) Cardiac MRI T1 with contrast. (d) Cardiac MRI T1 with contrast. (e) Cardiac MRI T1 without contrast. (f) Cardiac MRI T2.
CT-angiography showed a hypodense mass in RVOT, adhered to its wall and protruding through the pulmonary valve, with normal trunk, pulmonary arterial branches and parenchyma, and no debris suggesting pulmonary embolisms. This suggested that myxoma was the most probable diagnosis.
Cardiac MRI showed a 20-mm pedunculated mass in RVOT, a slight increase in the signal in T1 and a slight decrease in T2, with moderate progressive uptake of with gadolinium contrast, for which the radiologist gave us these three diagnoses in decreasing order of suspicion: CPF, slow flow hemangioma and myxoma (Figure 1(b)–(f), and Supplemental Video 1).
The protocolized oncological blood analysis, ultrasound and CT of the abdomen and pelvis, allowed us to rule out a metastasis as diagnosis as there were no other tumours.
The treatment was surgical resection. It was performed by median sternotomy, cardiopulmonary bypass with two venous cannulae and heart arrest with antegrade cold cardioplegia. Within 91 min of cardiopulmonary bypass time and 64 min of aortic cross-clamping, a small fashioned transverse right ventricular infundibulotomy was performed near the plane of the pulmonary valve. We were able to observe the pedicle of the tumour implanted on the anterior pulmonary valve leaflet, and the main tumour mass that was trilobed. The main tumour mass was loosely adhered to part of the other pulmonary valve leaflets, which could easily be separated with a blunt dissection. It was a yellowish rubbery mass, soft, smooth, flexible, with well-defined edges, without invasion of neighbouring structures and about 2 cm in diameter (Figure 2). Total resection of the tumour was performed, and restoration of valve function was obtained by valve reconstruction by means of the remaining leaflets bicuspidization of the pulmonary valve, folding the annulus of the anterior pulmonary valve, but without wrinkling it.
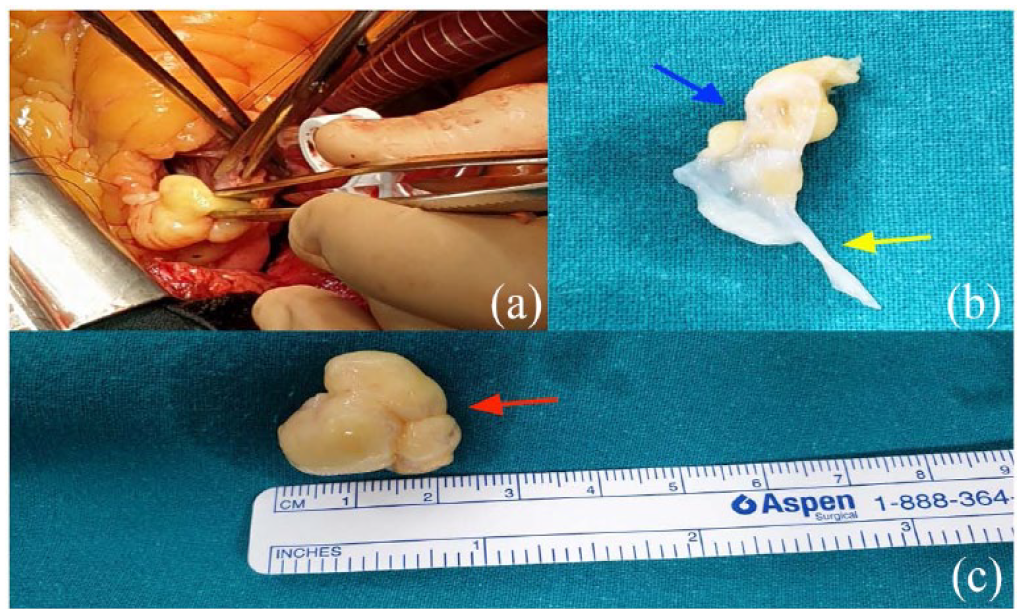
(a) Intraoperative photo showing the tumour through a little right ventricular infundibulotomy. (b) Blue arrow pointing to the pedicle of the lipoma. Yellow arrow pointing to pulmonary valve leaflet. (c) Main tumour mass, trilobed (red arrow). It is a yellowish, soft, smooth mass, with well-defined edges, without invasion of neighbouring structures, flexible and about 2 cm in diameter.
The intraoperative transesophageal echocardiogram showed that there were no tumour remains, there was no obstruction or pulmonary valve regurgitation.
The pathologic study described a well-defined lipomatous formation that stretches its endocardial covering. Tumour area of mature monovacuolar adipocytes with good capsular delimitation of the endocardial strip. Area of spindle cells and adipocytes, without atypia, and a weft of coarse, rope-like bands of collagen fibres. The immunohistochemical study shows immunoreactivity, especially in areas for CD-34, and positivity for CD-34. Negative for vascular and endocardial endothelial material CD-31 (Figure 3). A spindle-cell lipomatous benign tumour of the pulmonary valve was the diagnosis.

(a) Microphotograph of a well-defined lipomatous formation that stretches its endocardial covering (H-E, ×50). (b) Tumour area of mature monovacuolar adipocytes with good capsular delimitation of the endocardial strip (H-E, ×100). (c) Area of spindle cells and adipocytes, without atypia, and a weft of coarse, rope-like bands of collagen fibres (H-E, ×400). (d) The immunohistochemical study shows immunoreactivity especially in spindle-cell areas for CD-34 (×50). (e) Positivity for CD-34 (×400). (f) Negative for vascular and endocardial endothelial material CD-31 (×100).
She had a good recovery after surgery and no complication during the postoperative evolution, being discharged from hospital after 7 days from surgery, with echocardiographic control showing good biventricular function, absence of tumour or obstruction and minimal pulmonary valve regurgitation (Figure 4(a) and (b), and Supplemental Video 2).
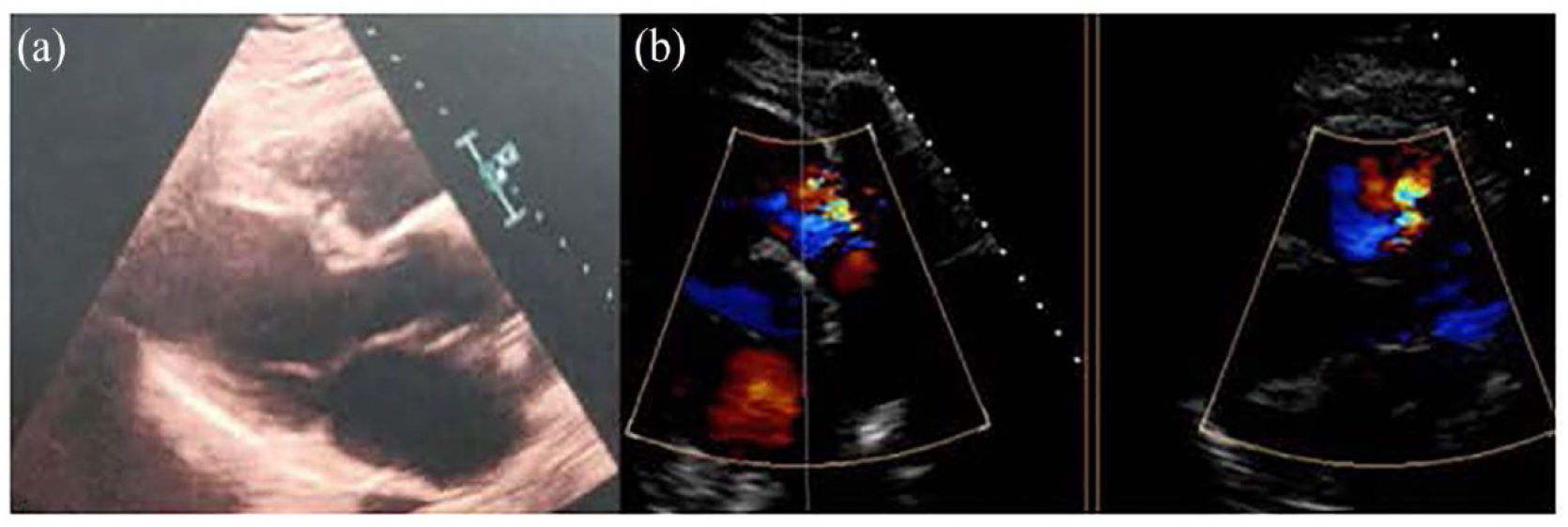
(a) Postoperative transthoracic ultrasound showing absence of tumour or obstruction in the pulmonary artery. (b) Postoperative transesophageal echocardiography showing minimal pulmonary valve insufficiency.
After 1-year follow-up, no evidence of the tumour by echocardiographic control.
Discussion
Usually, when a patient presents sudden dyspnoea with normal pulmonary exploration and an intense murmur in foci of the cardiac base, not irradiated to the neck or axilla, one must rule out aortic valve pathology, mainly because it is more frequent. This patient illustrates how any presumably atypical cardiological symptom with an unusual evolution, accompanied by an unexpected examination or a change in posture, should make us think about a cardiac tumour.
Although the tumour in the case presented is rare, this case exposes how to proceed in other more common valvular tumours.3,22 Early diagnosis and intervention are the key to preserve the normal function of the involved valve and to prevent potential critical events.3,22 Valve reconstruction in our case avoids complications related to prosthetic valves, such as thrombosis, endocarditis, paravalvular leak or the need for anticoagulation. A macroscopically very similar tumour located in the pulmonary valve leaflets that turned out to be a CPF was resolved, preserving the anatomical structure, by valve-sparing surgery 23 and it was a reference to us when planning the surgical approach and resolution.
Conclusion
Our case of a cardiac tumour and specifically primary pulmonary valve tumours that obstructs the RVOT with atypical symptoms and examination is a challenging case on how to suspect and confirm the diagnosis and how to manage it. The indication for surgery comes from cardiac mass diagnosis. The diagnosis of spindle-cell lipoma in the pulmonary valve was not confirmed until the tumour was observed in the intervention and the subsequent pathological analysis, despite the availability of echocardiography, CT and MRI.
Footnotes
Acknowledgements
We would like to express our sincere gratitude to all the individuals who have contributed to the publication of this case report, especially to Professor Rafael Martinez-Sanz, for his support and guidance.
We are also grateful to the Hospital Universitario de Canarias for allowing us and providing us with the resources and support we needed for this publication
Finally, we would like to thank the patient for her generosity and time
Author contributions
G.S.-F. is the corresponding author; A.B.-A. is the patient’s cardiologist and is in charge of the pre- and postoperative echocardiographic studies; H. Á.-A. is the specimen pathologist and is in charge of the anatomopathology images and information; J.M.-L. is in charge of the figures and videos; J.J.J.-R. is the bibliography reviewer; R.M.-S. is the main surgeon in this case and the text and figures supervisor.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received financial support for the publication of this article from Cardiolink S.L.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
