Abstract
Vanishing bile duct syndrome is a rare clinical manifestation, and many clinicians tend to classify vanishing bile duct syndrome as a surgical disease and perform emergency surgery, leading to poor prognosis for patients. In this report, we present a case of a patient initially diagnosed with probable vanishing bile duct syndrome. However, through a meticulous step-by-step investigation, we ultimately determined that the patient was suffering from Brucella infection-induced hemophagocytic syndrome, which contributed to the development of the probable vanishing bile duct syndrome. Once a definitive diagnosis was established, the patient underwent treatment following anti-Brucella and Hemophagocytic lymphohistiocytosis-2004 protocols, leading to an improvement in the patient’s condition. We conducted a literature review on brucellosis, and it demonstrated the lack of specificity in diagnosing Brucella infections and the diverse range of clinical manifestations. Failure to arrive at a definitive diagnosis may result in clinical misdiagnosis and delayed treatment, thereby leading to grave consequences.
Introduction
Hemophagocytic syndrome (HPS) is a rare and life-threatening immunological syndrome characterized by the uncontrolled activation of cytotoxic lymphocytes and macrophages. This leads to cytokine-mediated tissue injury and multiorgan dysfunction, and is typically associated with various underlying conditions, including infections, immunodeficiency, and other diseases. Clinical presentation includes high fever, hepatosplenomegaly, rapid deterioration of liver function, and central nervous system dysfunction. 1 The etiological factors contributing to HPS include: (1) viral infections (such as herpes viruses, Epstein–Barr virus (EBV), cytomegalovirus, and hepatitis viruses, etc.), (2) bacterial infections (such as Salmonella, Escherichia coli, Staphylococcus aureus, etc.), (3) parasitic infections and fungal infections, (4) associations with malignancies (lymphomas, acute leukemias, etc.), and (5) associations with autoimmune diseases (such as systemic lupus erythematosus 2 ). According to the updated HPS-2004 criteria, diagnosis of HPS requires the presence of at least five out of the following eight features: (1) Fever (38.5°C or more); (2) Splenomegaly; (3) Cytopenias⩾2 of 3 cell lineages (hemoglobin <9 g/dL or <10 g/dL in infants younger than 4 weeks; platelets (PLT) <100 × 103/mcL; and neutrophils <1 × 103/mcL); (4) Hypertriglyceridemia and/or hypofibrinogenemia (fasting triglyceride⩾3 mmol/L; fibrinogen (⩽1.5 g/L); (5) Hemophagocytosis; (6) Hyperferritinemia (⩾500 mcg/L); (7) High sIL-2R levels (⩾2400 U/mL); (8) Low/absent natural killer (NK) cell activity. Histopathological findings in HPS include: Haemophagocytosis in bone marrow and Haemophagocytosis in reticuloendothelial organs. 3
Vanishing bile duct syndrome (VBDS) is a pathological condition characterized by the progressive disappearance of the bile duct system following various types of damage, including immunological, infectious, and chemical factors. It manifests with hepatosplenomegaly, jaundice, and other associated symptoms. The extent of bile duct loss directly correlates with the severity of the patient’s condition and the prognosis, with a broader loss of bile ducts indicating a more critical state and a poorer prognosis. The clinical diagnosis of VBDS is based on the evaluation of clinical presentations, laboratory tests, and imaging data. Elevated alkaline phosphatase (ALP) and jaundice index can serve as auxiliary evidence. However, a definitive diagnosis of VBDS typically requires a liver biopsy performed through needle aspiration. 4 This biopsy provides crucial diagnostic information and confirms the presence of VBDS.
Brucellosis is a prevalent zoonotic infectious disease in Asia. Human brucellosis includes Brucella melitensis, Brucella suis, and Brucella abortus. The disease is primarily transmitted to humans through contact with infected animals such as cattle, sheep, pigs, rabbits, or their excrement, as well as the ingestion of infected meat or unpasteurized dairy products. The transmission routes include direct contact with the skin and mucous membranes, gastrointestinal tract infection, 5 and respiratory system transmission; 6 mother-to-child transmission of Brucella is extremely rare. In China, there were 326 cases of brucellosis reported in 1993, with an incidence rate of 0.028/100,000, the lowest in the history. However, since 1995, the incidence has steadily increased, and brucellosis has spread to all provinces. In 2019, there were 44,036 cases reported, with an incidence rate of 3.2513/100,000. 7 The clinical presentations of brucellosis is diverse, but the lack of specificity makes early diagnosis challenging. 8 Brucellosis can affect any organ system, 9 including the genitourinary system (orchitis and epididymitis), blood system (anemia, leukopenia, thrombocytopenia, disseminated intravascular coagulation), 10 central nervous system (meningitis), 11 bone and joint system (joint pain can involve the sacroiliac joint), 12 respiratory system (bronchitis, lobar pneumonia, pleural effusion), 13 and liver and gastrointestinal system (liver or splenic abscess, pancreatitis, colitis). 14 The proportion of liver involvement in brucellosis can be as high as 10%, 15 and it typically presents as hepatitis, hepatosplenomegaly, jaundice, and elevated transaminases. Although jaundice is a rare sign in the course of brucellosis, it can be the primary clinical manifestation. The definitive diagnosis of brucellosis requires the presence of the following three conditions: (1) Epidemiological history or clinical manifestations suggestive of brucellosis; (2) Positive tiger red plate agglutination test; and (3) Tube agglutination titer of 1:100 or higher.
Here, we report a case of Brucella infection-induced HPS with subsequent development of the probable VBDS.
Case presentation
Patient information
A 37-year-old male with no significant past medical history presented to Shanxi Bethune Hospital with a 10-day history of dizziness, fatigue, nausea, vomiting, and joint pain, accompanied by a fever for the past 5 days, with a body temperature peaking at 40°C.
Physical examination on admission
Body temperature was 38.6°C, a respiratory rate of 26 breaths/min, a pulse rate of 88 beats/min, and jaundice of the skin and conjunctival. Tenderness was noted in the right upper abdomen without rebound tenderness or muscle tension. The liver and spleen were not palpable, Murphy’s sign was positive, and there was percussion pain over the liver. Bowel sounds were heard at a rate of 3 times/min.
Laboratory test results
Aspartate transaminase 778.7 IU/L (15–40 IU/L), alanine transaminase 371.6 IU/L (9–50 IU/L), total bilirubin 96.7 μmol/L (⩽26 μmol/L), direct bilirubin 61.2 μmol/L (⩽4 μmol/L; Figure 1(a) and (b)), gamma-glutamyl transferase 191.7 IU/L (10–60 IU/L), and ALP 351.8 IU/L (45–125 IU/L). Blood work revealed white blood cell (WBC) count 2.7 × 109/L (3.50–9.50 × 109/L), red blood cell count 4.3 × 1012/L (4.50–5.80 × 1012/L), and PLT count 45 × 109/L (125–350 × 109/L; Figure 1(c–e)). Trends in the laboratory test indicators during the patient’s hospitalization can be observed in Figure 1.

(a) Trend graph of ALT and AST changes during the patient’s hospitalization. (b) Trend graph of TB and DB changes during the patient’s hospitalization. (c) Trend graph of WBC changes during the patient’s hospitalization. (d) Trend graph of RBC changes during the patient’s hospitalization. (e) Trend graph of PLT changes during the patient’s hospitalization. Horizontal coordinates indicate date, the vertical coordinate indicates the value.
Timeline and therapeutic interventions
The patient received symptomatic treatment, including fasting, fluid infusion, acid suppression, anti-inflammatory medication, liver protection measures, and albumin infusion. Magnetic resonance cholangiopancreatography (MRCP) revealed edema of the gallbladder wall and disappearance of intrahepatic bile ducts (Figure 2). Based on the patient’s clinical presentation of abdominal pain and jaundice, along with elevated transaminases and ALP levels, a preliminary diagnosis of probable VBDS was made. However, the patient’s blood routine examination showed a decrease in WBC count, red blood cell count, and PLT count, which is unusual for VBDS. Therefore, we began considering the possibility of other underlying diseases in the patient.
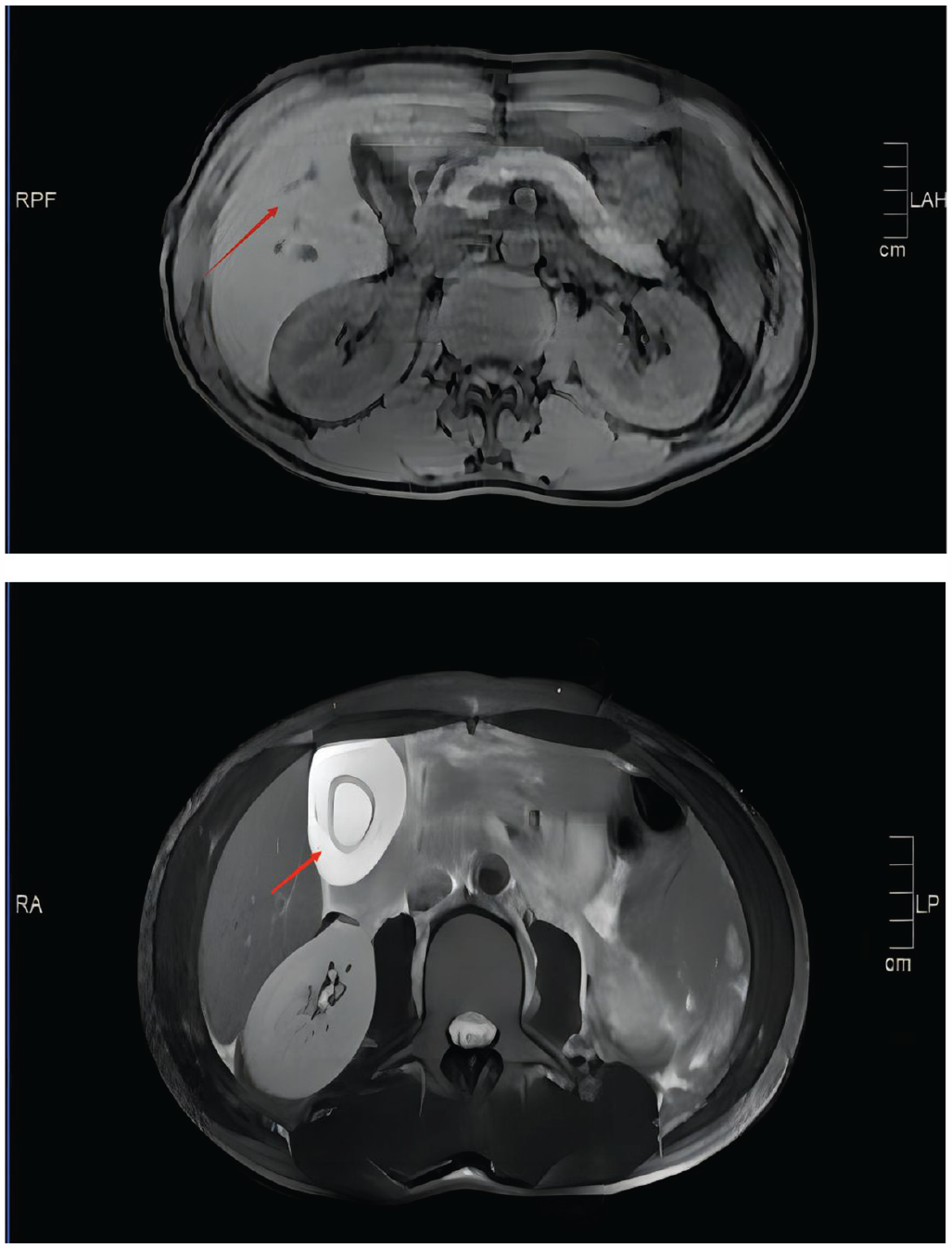
MRCP + plain scan of the liver, bile, and spleen was performed: Severe edema of the gallbladder wall and disappearance of intrahepatic bile ducts (red arrow).
The patient underwent an extensive diagnostic workup, including blood culture (to exclude severe infection), the antinuclear antibody spectrum (to exclude autoimmune disease), all of which showed normal results. Fine-needle aspiration of cervical lymph nodes, along with immunohistochemical analysis, suggests reactive lymphoid hyperplasia (Table 1). Additional investigations for infectious etiologies such as EBV, hepatitis A, B, C, and E, HIV, and syphilis all returned negative results (Table 2).
Aerobic and anaerobic blood culture, spectrum of antinuclear antibodies, and lymph node puncture results.

No abnormalities detected in aerobic and anaerobic blood culture. No abnormalities observed in the spectrum of antinuclear antibodies. Axillary lymph node puncture suggests reactive lymphocyte hyperplasia.
Infectious disease indicator results.
Hepatitis A,B,C, and E; HIV; and syphilis were negative.
Further examination revealed abnormal levels of fibrinogen 1.26 g/L (2.38–4.98 g/L), triglycerides 2.87 mmol/L (⩽1.7 mmol/L), soluble IL-2 receptor alpha chain (sCD25) 938.93 U/ml (92–402 U/ml), NK cell activity 1.33% (47.6%–76.8%), and ferritin>15000 ng/ml (23.9–336.2 ng/ml). On July 18, a bone marrow biopsy revealed the presence of haemophilus cells (Figure 3), meeting criteria (1), (3), (4), (5), (6), and (8)of the updated HPS-2004 criteria. Confirming the diagnosis of HPS as the underlying cause of the probable VBDS. The patient was subsequently treated with the Hemophagocytic lymphohistiocytosis (HLH)-2004 protocol, consisting of Dexamethasone (17 mg/day) and Etoposide (250 mg/day), 16 The treatment effect was satisfactory, with improvements in fever, nausea, vomiting, and joint pain. By July 24, the patient’s condition gradually stabilized. After taking the medication for 2 months, the patient reported no abnormalities and discontinued treatment. To determine the etiology of HPS, Further testing for infectious indicators such as kala-azar and brucellosis was conducted. The brucellosis antibody panel test, tube agglutination test, and tiger red plate agglutination test were performed on July 22, 2022, at Jin Yu Clinical Laboratory in Taiyuan city. The results showed positive Brucella immunoglobulin G (IgG ) antibodies, positive Brucella agglutination test (1:200 ++), and positive Brucella tiger red plate agglutination test (+) (Table 3). Combined with the patient’s clinical symptoms, this confirms the diagnosis of Brucella infection. Symptomatic treatment medications include rifampicin capsules, doxycycline hydrochloride capsules, compound sulfamethoxazole tablets, bicyclol tablets, and prednisone acetate tablets. The patient achieved recovery after 8 weeks. Through further inquiry into the medical history, it was discovered that the patient had a history of contact with sheep. Therefore, the truth finally surfaced that the patient was eventually diagnosed with Brucella infection-induced HPS, which subsequently led to the development of probable VBDS.
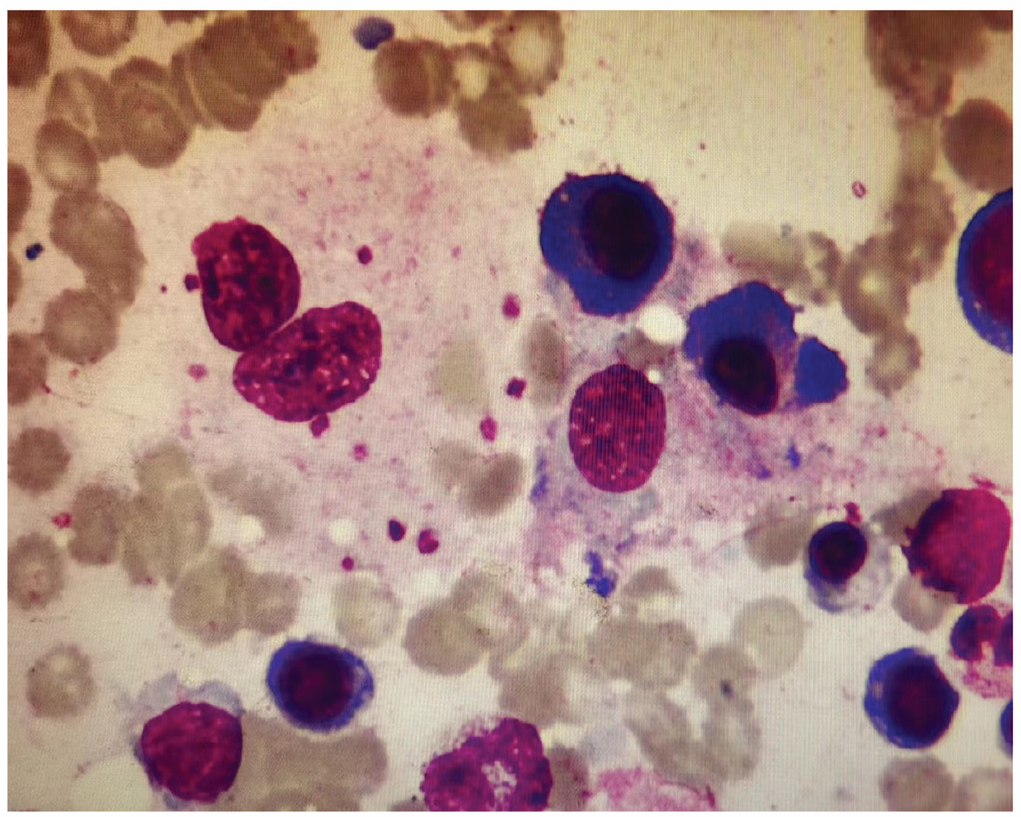
Bone marrow cell morphology report: granulocytes occupy 40.5% of nucleated cells, erythrocytes occupy 33.5% of nucleated cells, lymphocytes occupy 17.5% of nucleated cells, phagocytes occupy 3.5% of nucleated cells, with phagocytosed material, including mature red blood cells and platelets.
Brucella test results.

Jin Yu Clinical Laboratory in Taiyuan city showed positive test result for Brucella IgG antibodies, positive results for Brucella agglutination test (1:200 ++) and Brucella tiger red plate agglutination test (+).
Follow-up and outcome
He followed up as an outpatient, which normalized after completing anti-brucellosis and HLH-2004 therapy.
Discuss
This case was attributed to Brucella infection, which resulted in HPS, subsequently leading to probable VBDS due to sustained damage to the biliary system. In the early stages of the disease, the clinical symptoms such as high fever, chills, abnormal liver function, and jaundice are similar to clinical symptoms of VBDS. Magnetic resonance imaging (MRI) revealed severe edema of the gallbladder wall and disappearance of the intrahepatic bile ducts, which are typical features of VBDS (Figure 2). Although liver biopsy is the gold standard for diagnosing VBDS, the underlying cause of the patient’s condition has not been identified at present. Considering the patient’s compromised liver and coagulation function, performing a liver biopsy to confirm VBDS could easily lead to massive bleeding and endanger the patient’s life. Therefore, we can only highly consider it as VBDS and diagnose it as probable VBDS. Historically, our understanding of this disease has been limited, and invasive procedures have often resulted in uncontrolled bleeding and ultimately patient mortality. For example, Gao 17 reported a case of a patient admitted with the initial diagnosis of acute cholecystitis characterized by “gallbladder edema with significant wall thickening and intrahepatic bile duct disappearance,” without fully considering HPS or VBDS. An emergency laparoscopic cholecystectomy was performed, but the patient’s condition did not improve after surgery and even worsened. The postoperative pathological diagnosis confirmed gallbladder non-Hodgkin’s lymphoma, and the patient eventually died. The MRCP imaging data presented in Gao’s report the gallbladder edema observed in this case showed remarkable similarities, both demonstrating abnormal gallbladder edema and bile duct disappearance. Therefore, when we encountering patients with similar imaging presentations, it is crucial to remain vigilant regarding the possibility of HPS. Any invasive procedure at this stage could potentially result in patient death. Thus, it is essential to actively investigate the underlying cause and develop an appropriate treatment plan. In this case, as the disease progressed, the patient experienced a gradual decline in red blood cells, WBCs, and PLTs, along with increased ferritin levels, decreased NK cell activity, and elevated triglycerides. These findings met the criteria (1), (3), (4), (5), (6), and (8) of the updated HPS-2004 criteria. The bone marrow biopsy indicated the presence of haemophilus cells. At this point, confirming the diagnosis of HPS. Further investigations were conducted to determine the cause of HPS, including kala-azar, Brucella, ultimately leading to a diagnosis of brucellosis. In this case, the clinical manifestations of brucellosis, HPS, acute cholangitis, systemic inflammatory response syndrome, and VBDS overlap, posing challenges in terms of diagnosis and differential diagnosis for clinicians. If we make a single diagnosis to any these diseases, it may delay the condition, affect the prognosis, or even lead to death.
Therefore, early identification of the disease and the formulation of an appropriate treatment plan are crucial for ensuring a favorable prognosis. In this case, the absence of specific initial symptoms related to Brucella infection posed challenges for clinicians in establishing a connection between VBDS and brucellosis. Consequently, achieving an accurate diagnosis after admission assumes paramount importance.
We collected a series of case reports associated with Brucella infections and summarized them in (Table 4).18–27 These reports highlight that Human brucellosis remains the most prevalent zoonotic disease worldwide, affecting numerous countries and a wide range of age groups, including newborns, young adults, and elderly patients. The primary mode of transmission is through the consumption of unpasteurized milk from infected animals. 28 Transmission through placenta, breast milk, and sexual contact is extremely rare. 23 In our case report, upon further inquiry into the patient’s medical history, a history of contact with sheep was identified. This literature review encompasses 10 case reports, where 10% of brucellosis cases presented with HPS as the initial symptom and 20% presented with liver failure as the initial symptom. Other initial symptoms included undulating fever, liver abscess, and cirrhosis, with varying manifestations. Consequently, the early and accurate diagnosis of brucellosis assumes significant importance. In clinical practice, when a patient presents with admission symptoms such as jaundice, dizziness, fatigue, nausea, and vomiting, along with abdominal pain and cholecystitis upon physical examination, and an MRI reveals notable gallbladder edema and bile duct disappearance, it is crucial to consider various factors, including the possibility of HPS. Simultaneously, it is imperative to differentiate HPS from surgical conditions like gallstones and acute cholangitis, and refrain from premature surgical intervention until a confirmed diagnosis is established.
Articles related to the diagnosis of Brucella infection.

Ref. No.: References number; Y: Years; M: Male; F: Female.
Conclusion
The occurrence of Brucella infection-induced HPS followed by the subsequent development of the probable VBDS is extremely rare. In clinical practice, when imaging studies reveal findings such as hepatosplenomegaly, significant gallbladder wall thickening with edema, the disappearance of the bile duct, and an accompanying decrease in whole cell count, a comprehensive examination is necessary. Consider the possibility of HPS, actively search for underlying primary diseases, refrain from blind liver biopsy, and promptly initiate treatment based on the identified etiology to reduce the mortality rate.
Footnotes
Acknowledgements
The authors would like to thank the patient’s for permission to share the medical history for educational purposes and publication.
Authors’ contributions
M.X. and S. M. wrote the article and contributed equally to this work; X.F. provided clinical information; M.X. provided pictures. All authors reviewed the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
