Abstract
The great saphenous vein is the optimal conduit for infrapopliteal bypass surgery in chronic limb-threatening ischemia. A prosthetic bypass graft is an acceptable option when the autologous vein is unavailable and the anatomy is not favorable for endovascular therapy. However, the low patency rate of prosthetic grafts calls for adjunct strategies to improve the outcomes. Tapered reinforced grafts and vein cuffs are proposed measures. We herein present a case of a 78-year-old man with chronic limb-threatening ischemia and an extensive necrotic lesion on the dorsum of the left foot. He was successfully treated with a prosthetic bypass using two adjunct strategies to help improve patency: a tapered reinforced graft and a vein cuff in the distal anastomosis. After debridement and partial skin grafting, the wound healed completely in 2 months. The bypass graft was patent for 1 year.
Keywords
Introduction
The management of chronic limb-threatening ischemia (CLTI) includes accurately staging the disease severity and providing timely revascularization. 1 In cases of complex anatomy, open bypasses have demonstrated favorable results. 2 A good-quality great saphenous vein (GSV) is the optimal conduit of choice for infrapopliteal bypass surgery. 1 When the autologous vein is unavailable, and the anatomy is not favorable for endovascular therapy, a prosthetic bypass graft is an acceptable option in patients with good runoff. 1
Prosthetic grafts in infrapopliteal bypass have poor outcomes. 3 Dacron and expanded polytetrafluoroethylene (PTFE) grafts have low patency rates in below-the-knee bypasses. 4 Adjunct strategies have been proposed to improve these results. 5 Reinforced PTFE grafts may avoid conformation and path length changes during normal knee flexion. 6 Tapered PTFE grafts may influence fluid hemodynamics and ultimately reduce shear stress-induced intimal hyperplasia. 7 Vein cuffs may increase graft patency when tibial arteries are the targets. 8 The improved patency rates have been attributed to a shift in the site of intimal hyperplasia from the anastomosis to the vein cuff. The cuff’s wider cross-sectional area is thought to accommodate the redistributed site of intimal hyperplasia more readily. 9
We herein present a case of a patient with CLTI who was successfully treated with a prosthetic bypass using two adjunct strategies: a tapered reinforced graft and a vein cuff in the distal anastomosis. Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Case report
A 78-year-old man presented with an extensive necrotic lesion on the dorsum of the left foot after an episode of bullous erysipelas 2 months earlier (Figure 1). He had no fever nor signs of systemic infection. His medical history included poorly controlled systemic hypertension, hypothyroidism, and dyslipidemia. He had no symptoms of coronary artery disease and was not an active smoker. On physical examination, femoral and popliteal pulses were symmetrically palpable, but pedal pulses were absent in both limbs. The handheld Doppler detected only biphasic flow in the posterior tibial artery and no flow in the dorsalis pedis artery in both feet. The ankle-brachial indexes (ABI) were 0.48 on the right and 0.57 on the left. The left foot was markedly pale, and there was no hyperemia or pus drainage from the necrotic wound. The white blood cell count was normal. According to the WIfI classification, 1 his foot was W3 I2 fI0 (clinical stage 4), which gave him a high risk of amputation.
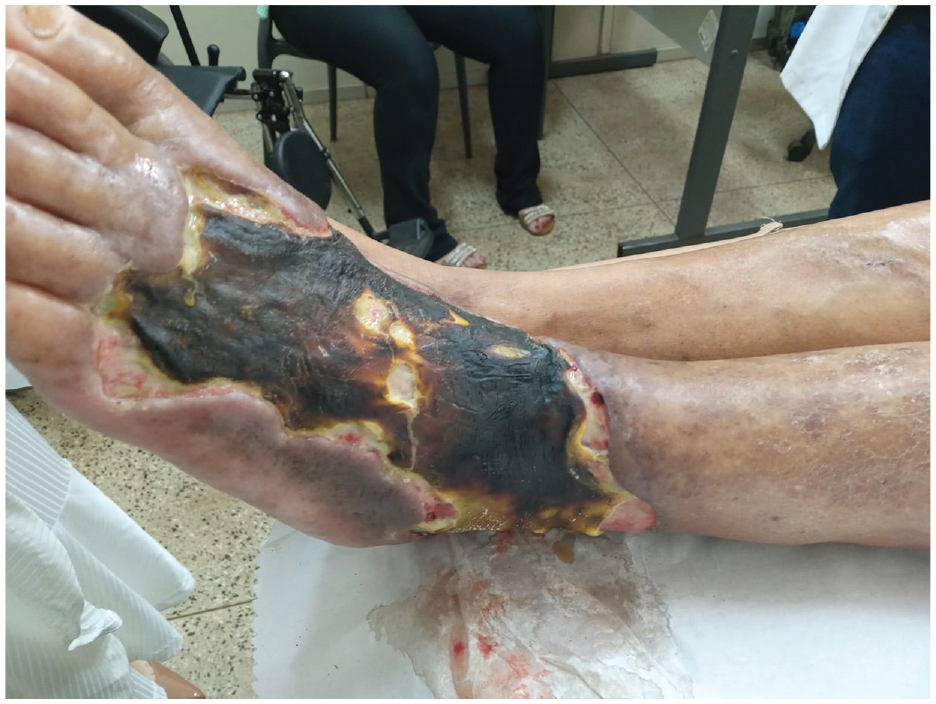
Digital image showing an extensive necrotic lesion on the dorsum of the left foot after a previous episode of bullous erysipelas.
To evaluate the possibility of revascularization, he underwent digital subtraction angiography. The aorta-iliac segment did not have significant disease. The left common and superficial femoral arteries exhibited diffuse atherosclerotic disease without significant stenosis. The left popliteal artery presented an aneurysm in its upper third. This artery occluded below the knee joint, just next to the emergence of the tibio-peroneal trunk. Both anterior tibial and posterior tibial arteries were not contrasted. Enlarged genicular arteries were observed as collaterals and filled the peroneal artery in the mid-leg, the single runoff vessel that reconstituted the pedal arch (Figure 2). The right limb was not studied. B-mode ultrasound confirmed the popliteal artery aneurysm, which had a diameter of 2.1 cm. Considering the peroneal artery as the target arterial path, the Global Limb Anatomic Staging System (GLASS) 1 stage was III (femoropopliteal grade 4 and infrapopliteal grade 4).

Digital subtraction angiogram. (a) The left common and superficial femoral arteries exhibited diffuse atherosclerotic disease without significant stenosis. (b) The left popliteal artery presented an aneurysm in its upper third (black arrow). (c) This artery occluded below the knee joint, just next to the tibio-peroneal trunk emergence (white arrow). Both anterior tibial and posterior tibial arteries were not contrasted. Enlarged genicular arteries were observed as collaterals and filled the peroneal artery in the mid-leg (black arrowhead). (d) The peroneal artery (black arrowhead) was the single runoff vessel to the ankle. (e) In the ankle, the dorsalis pedis (white asterisk) and the posterior tibial (white arrowhead) arteries were contrasted and formed an incomplete pedal arch.
An open bypass was recommended for limb salvage, as the patient has an acceptable cardiac risk and a complex anatomy, which was not favorable for endovascular therapy. However, ultrasound mapping revealed that both GSV presented wall thickening and chronic thrombi (previous thrombophlebitis) in the lower two-thirds. Both small saphenous veins and cephalic and basilic veins in both upper limbs had small diameters and were inappropriate for use as conduits. The ultrasound also evaluated the contralateral arteries, and there was no aneurysm.
Thus, we opted to do a femoro-peroneal bypass using a PTFE graft. To help improve the bypass patency, two adjunct strategies were employed: a tapered reinforced graft (Advanta VXT 7-4mm, Getinge AB, Göteborg, Sweden) and a vein cuff in the distal anastomosis. Through medial thigh access, the proximal anastomosis was made in the distal superficial femoral artery, proximal to the aneurysm, which was treated with a proximal and distal ligation. The graft was tunneled in the subcutaneous tissue, and the ringed section was positioned beside the knee joint. Through medial leg access, the distal anastomosis was done in the peroneal artery with a vein cuff, using the recently described “Brighton sock” technique. 10 Briefly, a short segment of the proximal ipsilateral GSV was harvested and submitted to saline distension. Macroscopic inspection allowed us to choose a healthy venous piece of adequate diameter. Portions of the vein that were non-distensible and presented wall thickening and chronic thrombus were removed. The vein was spatulated at one end and was anastomosed to the peroneal artery in a standard fashion. The “free end” of the vein was longitudinally incised and sewn to the distal end of the PTFE graft (Figure 3). Figure 4 summarizes what was done.

Intraoperative digital images. (a) Medial thigh access view—the proximal anastomosis (black arrow) was made in the distal superficial femoral artery (white arrow). (b) Medial leg access view—the distal anastomosis was done in the peroneal artery (white arrowhead) with a vein cuff (black arrowhead) using the “Brighton sock” technique.

Schematic diagram of the procedure. A femoro-peroneal bypass was done using a tapered reinforced expanded polytetrafluoroethylene (PTFE) graft and a vein cuff in the distal anastomosis. The proximal anastomosis was made in the distal superficial femoral artery, just proximal to the aneurysm, which was treated with a proximal and distal ligation. The distal anastomosis was done in the peroneal artery with a vein cuff, using the “Brighton sock” technique. A short segment of the proximal ipsilateral great saphenous vein was harvested and submitted to saline distension. Macroscopic inspection allowed us to choose a healthy venous piece of adequate diameter. The vein was spatulated at one end and was anastomosed to the peroneal artery in a standard fashion. The “free end” of the vein was longitudinally incised and sewn to the distal end of the PTFE graft.
The completion angiogram showed that the proximal anastomosis provided a good inflow, and the popliteal aneurysm was excluded. The distal anastomosis had an excellent outflow to the peroneal artery with a fast flow to the pedal vessels (Figure 5). The post-operatory ABI raised to 0.81. During the procedure, intravenous heparin (5000 UI) was administered before the arterial clamping. While the patient was in the hospital, subcutaneous enoxaparin (40 mg) was daily given for thromboembolism prophylaxis. Intravenous cefuroxime (750 mg every 8 h for 14 days) was the antibiotic regimen. The patient kept taking oral acetylsalicylic acid (100 mg) for life.

Completion angiogram. (a) The proximal anastomosis (black arrow) provided a good inflow, and the popliteal aneurysm was excluded. (b) The distal anastomosis (white arrow) had an excellent outflow to the peroneal artery.
The necrotic tissue was resected in a second procedure 3 days after revascularization. The wound bed presented active bleeding and was covered with silver sulfadiazine cream and petrolatum gauze, which was changed daily. Two weeks after debridement, a partial skin graft harvested from the ipsilateral thigh was used to cover the wound, which showed complete healing in 2 months (Figure 6). No secondary procedures in the bypass or in the wound were further required. The bypass graft was patent in the 1-year follow-up consultation, and the wound was still completely closed. The Doppler scan showed no hemodynamically significant peak systolic velocity (PSV) elevations at either anastomosis or within the graft, with PSV ranging between 23.3 and 39.6 cm/s and triphasic waveforms throughout.
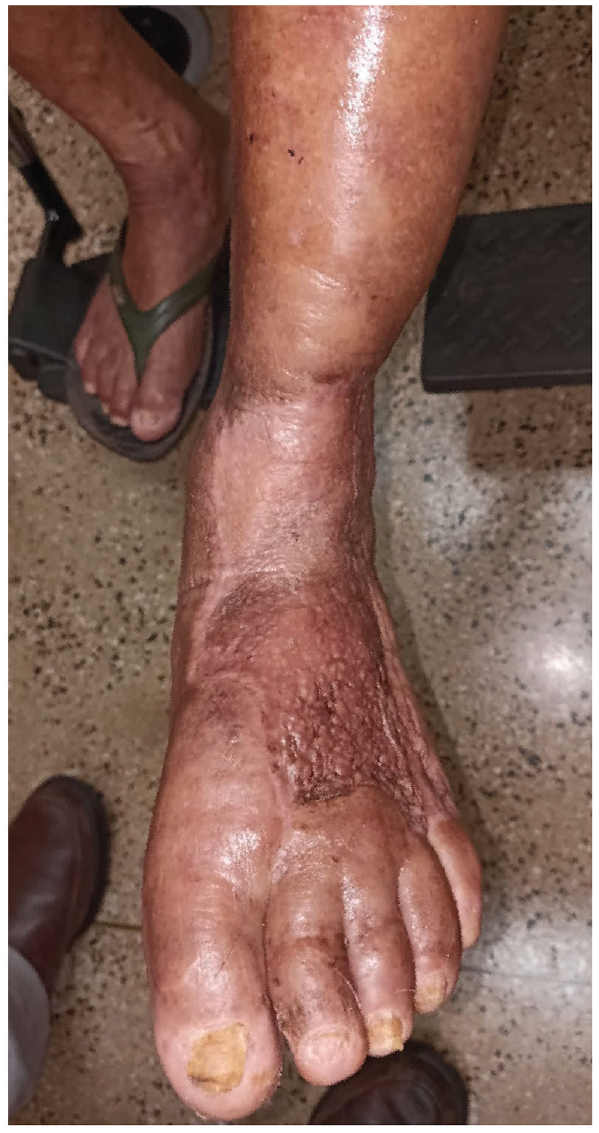
Follow-up digital images. A partial skin graft harvested from the ipsilateral thigh was used to cover the wound, which presented complete healing in 2 months.
Discussion
The present case is an example of how the global vascular guidelines can help decision-making in the management of CLTI. The WIfI classification allows accurate staging of the limb threat, while the GLASS can estimate the complexity of intervention. 1 Our patient had a WIfI stage 4, representing a high risk of amputation. With an ischemia grade of 2, a revascularization procedure would greatly benefit him. Conversely, he had a complex anatomy (GLASS III) with a popliteal/tibio-peroneal trunk chronic total occlusion. Although possible, an endovascular approach would have fewer chances of success due to the unfavorable anatomy. The treatment of the popliteal aneurysm would require a covered stent. Thus, an open bypass was considered the most appropriate therapeutical option.
The best conduit for the revascularization procedure would be the GSV. A recent randomized trial demonstrated that a good single segment of GSV is superior to endovascular therapy. 2 However, ultrasound mapping showed that both GSVs had thrombophlebitis in the lower two-thirds. Also, both small saphenous veins and upper limb veins were small and inappropriate.
In a patient lacking a venous conduit and unfavorable for endovascular therapy, choosing the optimal approach remains a significant challenge. 1 Prosthetic conduits are the next rational choice, although their patency rates in infrapopliteal bypasses are low. 3 Infection is also a major concern in limbs with open wounds, as in the present case. Adjunct strategies have been proposed to improve the results of prosthetic bypass to tibial targets. 5 In our patient, two of these measures were employed: a tapered reinforced graft and a vein cuff in the distal anastomosis.
Tapered PTFE grafts bring a more compatible size match when a bypass between vessels of different diameters is required. This geometrical conformation has the potential benefit of promoting less turbulent flow, thus reducing wall shear stress and intimal hyperplasia. 11 These grafts have been widely used for hemodialysis fistulae when a significant diameter mismatch between an artery and a vein is expected. 7 An additional advantage is the reinforced section, which may avoid conformation and path length changes during normal knee flexion. 6 This could prevent graft kinking and thrombosis. 6
Vein cuffs in distal anastomosis are not a new concept. 12 They may improve graft patency by promoting a shift in the site of intimal hyperplasia from the critical areas of the anastomotic heel, toe, and floor in a PTFE-artery anastomosis to the graft–vein interface. 8 The cuff’s wider cross-sectional area is likely to accommodate the redistributed site of intimal hyperplasia more readily. 9 Another possible mechanism is that the vein cuff causes an alteration in blood flow dynamics at the artery–cuff interface, thus forming a cohesive vortex in the conduit. This would alter the wall shear stress and reduce the stimulus for intimal hyperplasia.10,13,14 Vein cuffs also may bring the following technical advantages: allow an easier anastomosis, particularly to small vessels 15 ; help preserve the outflow in the event of graft thrombosis 16 ; and provide a better compliance match between the vein and the recipient artery. 17 The most employed vein cuffs are the Miller cuff 18 and the St. Mary’s boot. 19 Recently described, the “Brighton sock” adaptation is reproducible and easy to perform. 10 This technique is proper when there is a short and healthy vein segment of adequate diameter, as in our case. The vein is not longitudinally open. It is only spatulated on both ends. Thus, the vein is used in its whole diameter, which resembles a composite graft with a small venous part. Figure 7 illustrates the most used techniques.

Most employed techniques for vein cuffs.
There is some evidence that a vein cuff at the distal anastomosis site improves primary graft patency rates for infrapopliteal PTFE bypasses. 20 However, data are limited in scope and quality. 8 Nguyen et al., in an extensive review, recommended that if prosthetic conduits must be used, they should be performed with a distal venous adjuvant. 3 McPhee et al. demonstrated that vein cuffs may improve the results of prosthetic bypass surgery and should be considered when feasible. 21
A composite graft, part PTFE, and part vein would be also a possibility in our case. These procedures have a high early failure rate, not better than pure prosthetic conduits, and are discouraged by Nguyen et al. 3 As stated above, these authors recommend using a vein cuff if prosthetic conduits are used. Curiously, a vein cuff using the “Brighton sock” technique resembles a composite graft with a small venous part. Compared with composite grafts, vein cuffs require shorter vein segments, avoid possible stenosis due to diameter mismatch between the anastomosed structures, and allow technically more accessible performance of the distal anastomosis. 17
Although our patient had CLTI, the etiology of the arterial occlusion is a point that should be addressed. The patient was 78 years old with poorly controlled hypertension and dyslipidemia. Atherosclerosis could certainly be the cause of the occlusion. Embolism from the aneurysm was also considered. In both situations, the occlusion was chronic, and neither fibrinolysis nor thrombectomy was cogitated. The bypass was considered the most appropriate therapeutical option.
Our patient had a favorable evolution with the adopted approach. The wound healed completely, and the graft was patent for 1 year. It is essential to point out that the clinical outcomes of prosthetic grafting in CLTI are susceptible to runoff. 1 As the runoff was favorable, we opted to proceed with a nonautologous conduit and employed adjunct strategies to improve graft patency.
Conclusion
In this case, the global vascular guidelines helped decision-making in the management of CLTI. The patient had a favorable runoff but was unfavorable for endovascular therapy. Without an adequate GSV, he was successfully treated with an infrapopliteal prosthetic bypass graft with two adjunct strategies: a tapered reinforced graft and a vein cuff in the distal anastomosis.
Footnotes
Acknowledgements
None.
Author contributions
Conception and design: MBD, KBG, MBJ, and EEJ. Analysis and interpretation: MBD, KBG, MBJ, and EEJ. Data collection: MBD, KBG, MBJ, and EEJ. Writing the article: MBD, KBG, and MBJ. Critical revision of the article: MBD, KBG, MBJ, and EEJ. Final approval of the article: MBD, KBG, MBJ, and EEJ. Statistical analysis: n/a. Overall responsibility: MBD.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series. Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Guarantor
The guarantor: Marcelo Bellini Dalio accepts official responsibility for the overall integrity of the manuscript, including ethics, data handling, reporting of results, and study conduct.
