Abstract
Breast surgery is performed to achieve local control in patients with breast cancer. Visualization of the anatomy with a virtual reality software platform reconstructed from magnetic resonance imaging data improves surgical planning with regards to volume and localization of the tumor, lymph nodes, blood vessels, and surrounding tissue to perform oncoplastic tissue rearrangement. We report the use and advantages of virtual reality added to the magnetic resonance imaging assessment in a 36-year-old woman with breast cancer who underwent nipple sparing mastectomy with tissue expander reconstruction.
Keywords
Introduction
Over the last few decades new imaging techniques have been developed in breast cancer imaging to improve the medical treatment and surgical planification, such as four-dimensional (4D) mammograms, breast magnetic resonance imaging (MRI), computed tomography (CT) scan, and three-dimensional (3D) printing. However, the understanding of a 3D biologic structure is complex and requires visualization of the interplay of all the structures surrounding the tumor.
Recently, 3D virtual reality (3D-VR) has shown promising results in several fields due to the reconstruction of a 3D structure with minor details that can be explored in a 3D fashion, which are challenging to assess on a two-dimensional (2D) screen. 1 Several studies have shown that 3D-VR improves the speed and accuracy of surgeon analysis, specifically via the accuracy of preoperative MRI scans prior to breast surgical interventions.2–4 Moreover, it has shown a better understanding of the anatomy among different surgical fields such as craniofacial trauma, complex congenital disease surgery, urology, abdominal pediatric surgery, surgical oncology, plastic surgery, among others.5–10 We present the use of 3D-VR in a breast cancer patient who underwent nipple sparing mastectomy (NSM) with reconstruction as an additional tool to improve surgical planning.
Case report
A 36-year-old female was referred to the breast surgery clinic due to a palpable mass in the left breast. Past medical history was relatively unremarkable. She was thin with a body mass index (BMI) of 22 and was a former smoker. She had a significant family history of breast cancer however genetic testing was negative. On physical examination two firm masses were palpated in the lower pole of the left breast, with neither appearing to be fixed to the skin or chest wall. The first was estimated at 1.5 cm in the lower inner quadrant just superior to the inframammary fold and the second was found to be retro areolar and was estimated at 2 cm.
Diagnostic imaging and ultrasound guided core biopsy were performed and both masses were found to be consistent with invasive ductal carcinoma, estrogen receptor (ER) and progesterone receptor (PR) positive, and HER2 negative. MRI did not show additional areas of concern and no axillary or internal mammary adenopathy was noted.
Prior to the surgery, MRI images were reconstructed with the AVATAR MEDICAL software, which showed the dimensions of the tumors and distance from the tumor to the skin, with the trajectory and anatomy the breast perforator arteries identified (Figure 1). The location of the perforators was assessed both with the standard 2D MRI images as well as the 3D reconstruction. In addition, standard review of mammograms and MRI (Figure 2) was performed prior to the day of surgery.
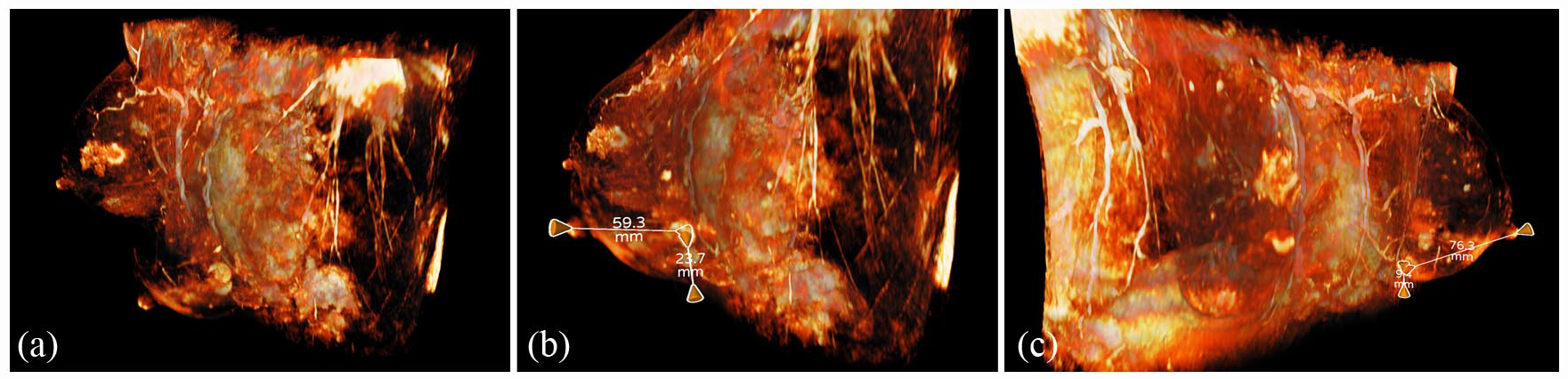
Visualization of the avatars based T1-weighted contrast-enhanced MRI using the AVATAR MEDICAL software. Different three-dimensional views assessing the location of the two tumors (a–c). Three-dimensional view of the breast perforator arteries from the intercostal arteries (left breast) and internal thoracic artery (left and right breast) (a). Demonstration of the measurement of the distances from the superior tumor to nipple-areolar complex (59.3 mm) and to skin (23.7 mm) (b). Different view showing the distances from the inferior tumor to nipple-areolar complex (76.3 mm) and to skin (9.4 mm) (c).

MRI of the patient with two tumors in the left breast (a, b).
The patient was taken to the operating room for an NSM of the left breast. During the mastectomy, an inframammary fold incision was performed; the breast was completely freed from the chest wall and separated from the subcutaneous fat. The perforators that had been visually identified preoperatively were preserved. After removal of the specimen, three left axillary sentinel lymph nodes were identified and removed. The patient then underwent reconstruction of breast with a tissue expander.
Post-operatively, the patient experienced no complications and progressed without incident. She was discharged home the following day. Pathology confirmed negative margins for both tumors, the tumor measurements were 2.0 and 1.6 cm, nuclear grade 2, 1/3 nodes with isolated tumor cells, and AJCC 8th ed. pT1c N0(i +)(sn). 11
On follow-up, surgical flaps were noted to be well-perfused, and incisions healed well. The nipple of the left breast had mild ischemic changes which resolved by the next follow up visit. Moreover, the patient was seen by plastic surgery as an outpatient to undergo implant-based reconstruction during the upcoming months.
Discussion
We presented a case that showed the impact of 3D VR in surgical planning compared to the current standard of care in a patient with breast cancer who underwent NSM and Sentinel Lymph Node Biopsy (SLNB) with reconstruction.
The 3D VR is created based on MRI data that allows the visualization of volumetric organs in an intuitive fashion, and allows the surgeon to interact with the future specimen. 1 With the aid of this new technology, the preoperative understanding of the volumetrics from an anatomic structure may enhance the performance and confidence during surgery, especially in memory storage of complex visualized structures. 12
Important topics to be considered at the time of NSM include the blood supply to the flap, the proximity of the tumor to the skin, and the incision performed. 13 Hence, prior to this procedure, visualization of flap perforators is useful in guiding the flap dissection with the hope of sparing the vessels to avoid flap ischemia. In fact, the 3D-VR is helpful in visualizing the anatomical distribution of the perforators, which are vital to the flap at the time of the reconstruction. The use of 3D-VR allowed us to rotate the MRI image and see it in three dimensions, which gave us an improved preoperative mental picture of the perforators and the tumors. We suspect that 3D virtual reality may improve surgical outcomes with NSM and possibly in any flap due to the high resolution of the blood supply in any tissue.
In addition, 3D-VR allowed a better visualization of the tumor distance from the skin from every angle seen in the 3D reconstruction software. Preoperative MRI has demonstrated improvement on surgical outcomes, and specifically in free margin rate, 14 however a previous visualization in 3D-VR can provide even better analysis of the MRI images prior to the surgery. As mentioned above, virtual reality plays an important role in surgical planning and medical learning in terms of scene and direction among different scenarios. Intraoperatively, previous analysis of the location from the tumor, blood supply, and lymph nodes gave a targeted dissection and use of different maneuvers to improve the precision of surgical technique. For instance, when approaching the tumor which was close to the skin, Mayo scissors were preferred over the electrocautery to avoid contact with the tumor. 12 Moreover, the advantage of this technology over intraoperative ultrasound (IOUS) with or without color Doppler is the planning of case which can result is a faster approach of the tumor and a tentative faster learning curve;15,16 however, both together could lead to a higher accuracy in margins and identification of perforators especially in complex cases,17–19 further studies are recommended that compare surgical and oncologic outcomes with both technologies.
While 3D reconstruction was subjectively helpful in understanding the anatomy, the most obvious limitation to consider is the prone position of the patient when the MRI is performed, which elongates the breast and limits the ability to see the exact position of the masses during the surgery. Therefore, 3D-VR could be greatly enhanced by pairing it with data obtained from supine MRI. Supine MRI would likely provide a more accurate roadmap in which the surgeon could visualize the structures as they will appear in the OR, and this seems to be the appropriate next step in evolution. The fact that the perforator anatomy is less variable with positional change given the proximity to the chest wall musculature may make the benefit of supine MRI in this application less meaningful. Furthermore, this tool in conjunction with 3D printing can improve surgical planning and achieve better outcomes as suggested in previous literature and both technologies have shown promising positive results in the surgical field.20,21 In addition, as of now, there is no way to use the technology to aid with sentinel node biopsy, although with the use of magnetic node localization, this is certainly an area of interest in the future. With magnetic node localization the MRI data and subsequent 3D reconstruction would provide a detailed roadmap prior to surgery and could theoretically lead to less dissection and reduced rates of lymphedema.
Conclusion
In conclusion, the use of 3D virtual reality may enhance surgical planning and execution in the treatment of breast cancer and its reconstruction. Certainly, prospective randomized controlled trials using this technology would be needed to confirm benefit. Potentially with a better understanding and assessment of pre-surgical imaging, the speed and accuracy of excision and reconstruction could be achieved, especially in high-risk patients and patients with complex location of tumors in breast surgical oncology.
Footnotes
Acknowledgements
We thank the AVATAR MEDICAL team: Maxime Guillerm and JJ Ross for all the help and support to facilitate the AVATAR MEDICAL software and the support to do this project.
Author contributions
G.D.K. contributed to conceptualization—ideas; data curation; methodology; validation; visualization; writing—original draft; writing—review & editing. M.M. contributed to conceptualization—ideas; data curation; methodology; validation; visualization; writing—original draft; writing—review & editing. J.S.T.R. contributed to validation; writing—review & editing. D.L. contributed to conceptualization—ideas; data curation; methodology; validation; visualization; writing—original draft; writing—review & editing.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
