Abstract
R-CHOP chemotherapy has been established as the first-line standard treatment for elderly patients diagnosed with diffuse large B-cell lymphoma. However, an increased risk of Pneumocystis jirovecii pneumonia in diffuse large B-cell lymphoma patients after the rituximab-based chemotherapy has been reported. We describe a case who developed intermittent cough, fever and shortness of breath after five cycles of R-CHOP treatment for diffuse large B-cell lymphoma. A rapid deterioration in patient’s respiratory condition prompted us to adopt an aggressive anti- Pneumocystis jirovecii pneumonia strategy that combined the conventional trimethoprim/sulfamethoxazole and another two antimicrobials, caspofungin and clindamycin. This is the first report mentioning the successful treatment of severe Pneumocystis jirovecii pneumonia with a triple-drug regimen in a HIV-uninfected patient. The aim of our report is also to emphasize that early and correct diagnosis of Pneumocystis jirovecii pneumonia in immunocompromised HIV-uninfected patients is very important. Relevant oncologists should be alert to the risk of Pneumocystis jirovecii pneumonia in patients receiving R-CHOP chemotherapy.
Introduction
Pneumocystis jirovecii is well-known as an opportunistic fungal pathogen, which causes life-threatening pneumocystis pneumonia in human immunodeficiency virus (HIV)-infected patients.1,2 However, with the increasingly wide use of chemotherapeutic agents and immunosuppressants, Pneumocystis jirovecii pneumonia (PJP) is nowadays commonly found in HIV-uninfected patients diagnosed with hematological malignancy, solid cancer, and rheumatic diseases, 3 as well as in patients receiving immunosuppressive therapy after organ transplantations. 2 Diffuse large B cell lymphoma (DLBCL) is the most common type and aggressive form of B-cell non-Hodgkin’s lymphoma (NHL). In elderly patients with DLBCL, the current standard first-line treatment is R-CHOP (rituximab, cyclophosphamide, doxorubicin, vincristine, prednisolone) chemotherapy. 4 Rituximab is a chimeric monoclonal antibody targeting the CD20 antigen, expressed on both malignant and normal B lymphocytes. 5 The addition of rituximab to CHOP chemotherapy has been demonstrated to increase event-free and overall survival. 6 However, R-CHOP-related opportunistic PJP has been reported. Indeed, PJP prophylaxis should be recommended to high-risk DLBCL patients, and potential P. jirovecii infection warrants significant attention during or after the chemotherapy.7–9 Herein, we present a case of severe PJP in a DLBCL patient after the R-CHOP chemotherapy.
Case presentation
A 59-year-old male was diagnosed with DLBCL (stage IA) after presenting the left leg pain and swelling for 6 months and magnetic resonance imaging (MRI) scan demonstrated abnormalities of the bone marrow cavity of the left tibia suggestive of malignancy. His past medical history included coronary artery disease treated with percutaneous coronary angioplasty and stenting and atorvastatin, clopidogrel, and beta-blocker therapy. The patient was thus started on R-CHOP chemotherapy, which consisted of rituximab 375 mg/m2 (0.5 g on day 1), cyclophosphamide 750 mg/m2 (1.2 g d1), vincristine 1.4 mg/m2 (2 mg d1), doxorubicin liposome (6 mg d1), and prednisone 100 mg (d1-5 q21d).
Overall, each round of R-CHOP treatment went well except for an episode of severe bone marrow suppression after the second chemotherapy cycle, accompanied by bacterial pneumonia, from which the patient recovered with antibiotic therapy. Two weeks after completing the fifth chemotherapy cycle, the patient weighing 64 kg was readmitted to assess the efficacy of the chemotherapy regimen by positron emission tomography-computed tomography (PET-CT) scan. On admission, he complained of intermittent mild cough with a small amount of white sputum. The vital signs were normal. There were no palpable lymphadenopathy and auscultation of the lung fields bilaterally demonstrated normal vesicular breath sounds without adventitious sounds. Laboratory results revealed leukocytosis, lymphocytopenia, moderate anemia, elevated C-reaction protein level, normal procalcitonin, and normal serum 1-3-beta-D-glucan level (Table 1). The hepatic and renal profiles were within normal limits. Sputum cultures did not isolate any respiratory pathogens and the serum cytomegalo virus (CMV) viral load was undetectable on polymerase chain reaction (PCR) testing. The HIV test was nonreactive during each course. The chest X-ray on admission exhibited patchy shadows in both lung fields (Figure 1).
Patient’s laboratory findings during the hospital stay.
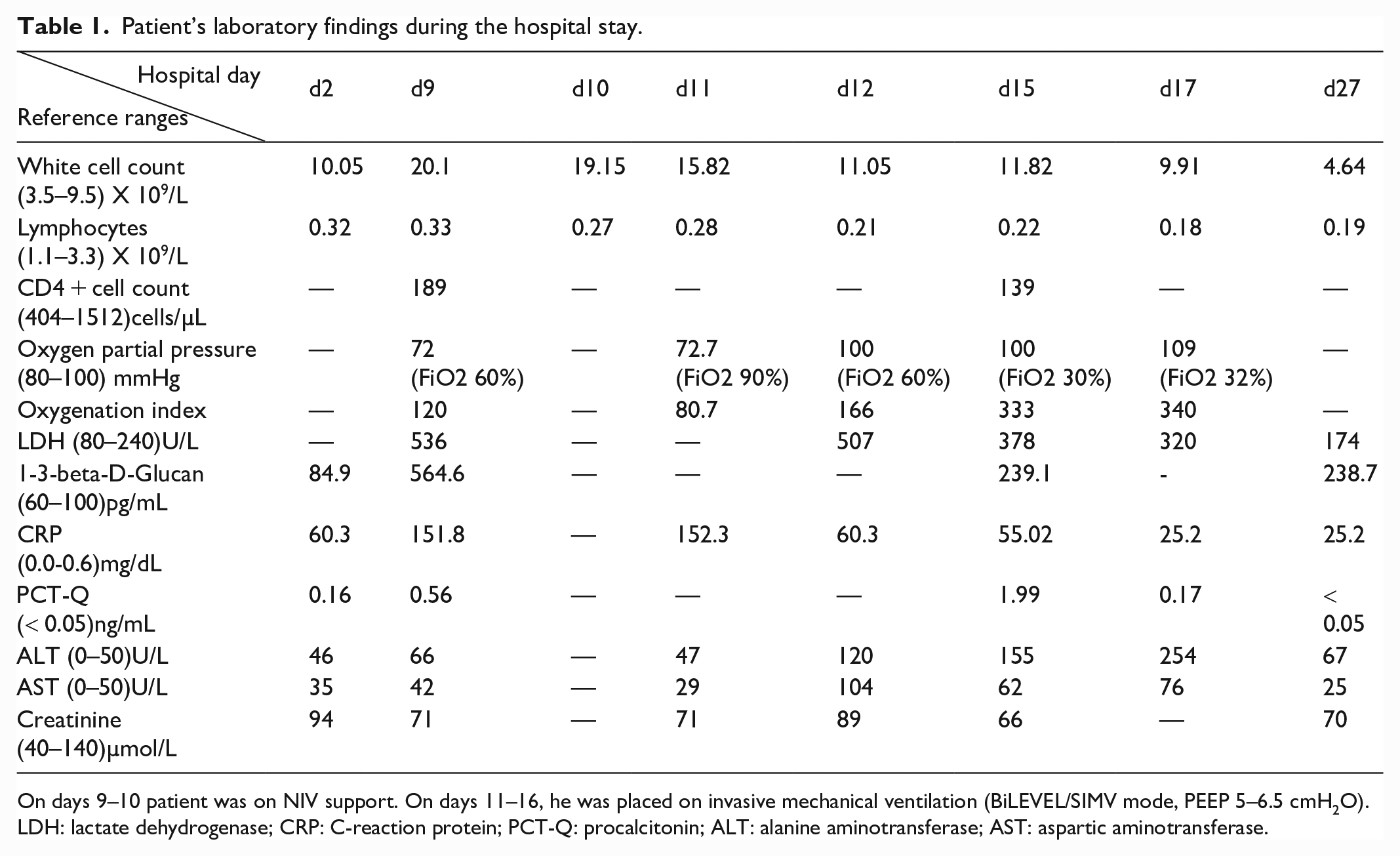
On days 9–10 patient was on NIV support. On days 11–16, he was placed on invasive mechanical ventilation (BiLEVEL/SIMV mode, PEEP 5–6.5 cmH2O).
LDH: lactate dehydrogenase; CRP: C-reaction protein; PCT-Q: procalcitonin; ALT: alanine aminotransferase; AST: aspartic aminotransferase.

On hospital day 1, a chest X-ray revealed bilateral patchy shadows.
The patient’s PET-CT scan showed that the tumor focus in the lower left tibia had decreased in size and glucose metabolism, suggesting that his lymphoma was in remission. However, multiple patchy and cord-like opacities with increased glucose metabolism were observed in the lungs, indicating pneumonia. Meanwhile, the patient experienced daily fever up to 38.9°C, and had a mild cough with minimal white sputum. He was thus empirically treated with cefoperazone and levofloxacin (Figure 2).
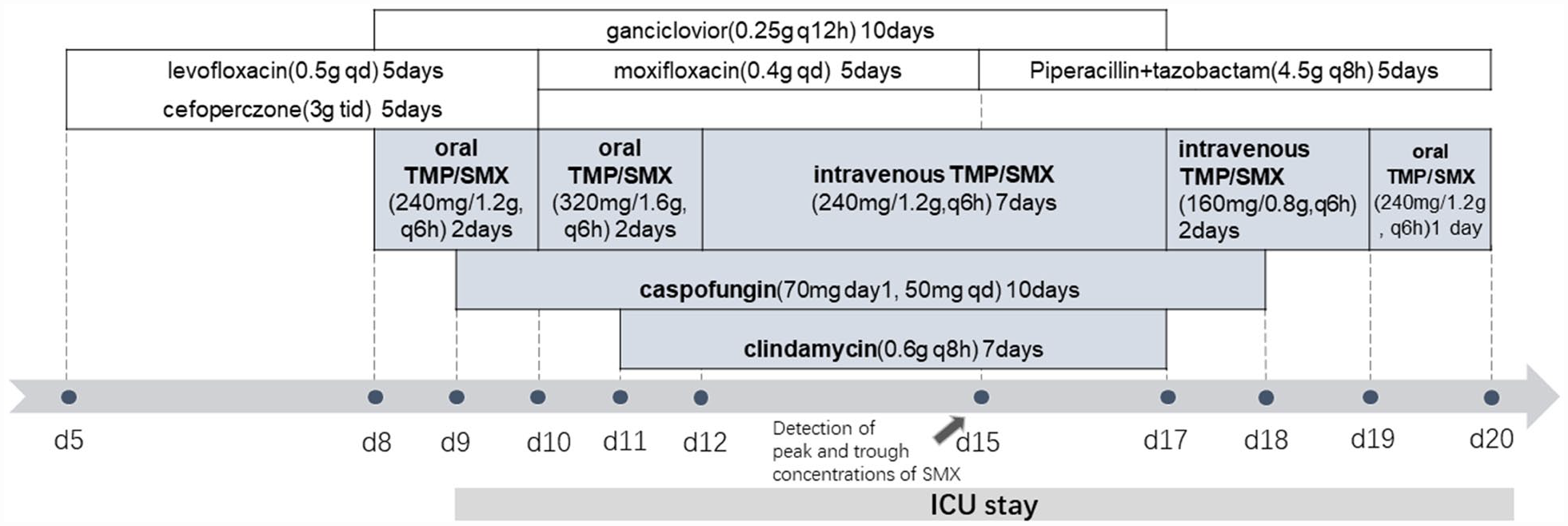
Antimicrobials course before and during the patient’s ICU stay.
On day 8 of admission, the patient developed dyspnea and hypoxemia, with an oxygen saturation of 90% on room air. Ganciclovir and oral trimethoprim/sulfamethoxazole (240 mg/1.2 g q6 h) were administered to cover viruses, bacteria and P. jirovecii (Figure 2). However, the patient’s respiratory condition rapidly deteriorated, and he was transferred to the intensive care unit (ICU) for ventilatory support. He received noninvasive ventilation (NIV), under which the oxygenation index (PaO2/FiO2 ratio) was around 100 ± 17, strongly suggesting acute respiratory distress syndrome (ARDS) and severe PJP. Bronchoalveolar lavage (BAL) liquid was obtained and sent for bacterial culture and metagenomic next-generation sequencing (mNGS). BALF-mNGS yielded abundant P. jirovecii DNA. Meanwhile, laboratory tests revealed increased levels of lactate dehydrogenase (LDH) (536 U/L) and 1-3-beta-D-glucan (564.6 pg/mL) (Table 1), and complete blood cell counts showed the CD4+ T cell count was 189 cells/µL. In addition, the chest CT showed multiple ground glass opacities (GGOs) in both lungs (Figure 3). Based on the above evidence, the diagnosis of severe PJP in our DLBCL patient was confirmed. The echinocandin, caspofungin (70 mg d1, 50 mg qd) was early added to the standard TMP/SMX (dose increased to 320 mg/1.6 g, q6 h) regimen to eliminate both the cysts and trophic forms of P. jirovecii. Piperacillin + tazobactam, moxifloxacin, and ganciclovir were concurrently administered to cover possible bacterial and viral infections (Figure 2). Systemic methylprednisolone was used to alleviate pneumonia.
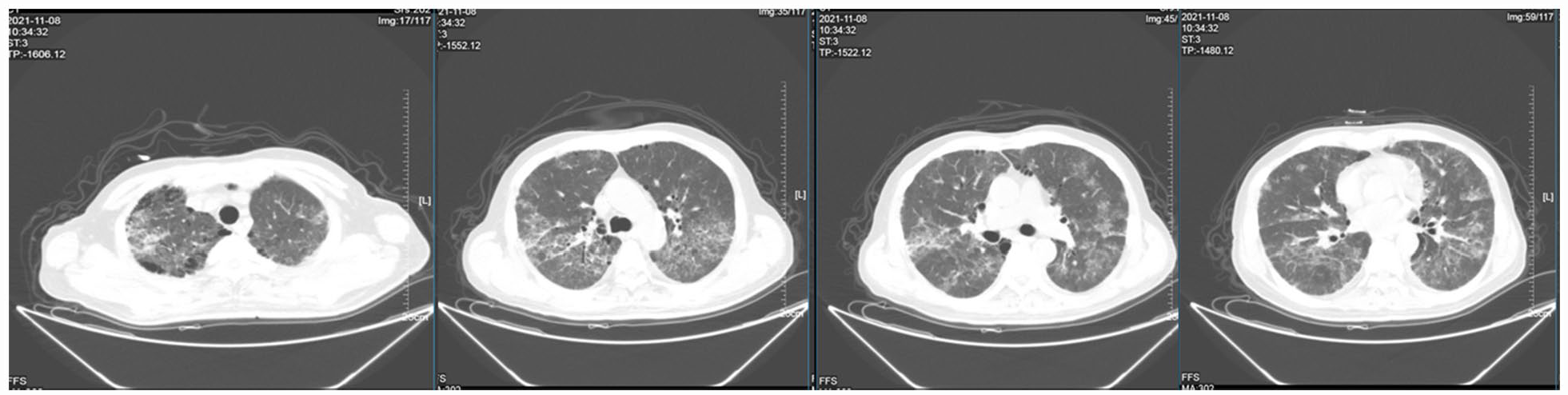
Chest CT scan on day 8 revealed diffuse stripe-like and ground-glass high-density shadows with honeycomb structures in both lung fields.
On day 11, the patient was intubated and placed on invasive mechanical ventilation due to severe dyspnea. Prone position ventilation was also attempted to improve gas exchange in the lungs. However, repeated chest X-ray findings showed no significant improvement but dilated bowel loops (Figure 4). Since gastrointestinal dysfunction could impair the absorption of oral TMP/SMX and decrease the plasma drug concentration, the patient was switched to intravenous TMP/SMX, supplemented with clindamycin (0.6 g q8 h) (Figure 2).

Chest X-rays on day 11 showed bilateral diffuse ground glass-like opacities with honeycomb changes. The abdominal radiograph revealed dilated bowel loops.
After treatment with a combination of TMP/SMX, caspofungin, and clindamycin, the oxygenation index was significantly improved, and the levels of LDH and 1-3-beta-D-glucan began to decline (Table 1). With an improvement of the patient’s respiratory condition and resorption of pulmonary infiltrates on the repeat chest CT (Figure 5), clindamycin and caspofungin were discontinued after 7 and 10 days of use, respectively, to avoid potential side effects. As the patient’s general condition improved, he then was switched to oral TMP/SMX and the dosage was decreased when a mild increase in hepatic transaminases was observed (Table 1). The blood samples were sent to the company (BGI Co., Ltd, Shenzhen) for determination of blood TMP/SMX concentrations sponsored by the pharmaceutical manufacturer, given that intravenous TMP/SMX has only recently become available in mainland China. On day 15, after 3 days of us-ing intravenous TMP/SMX, the trough and peak levels of sulfamethoxazole were, respectively, 152.92 mg/L and 177.98 mg/L, which was reasonable for treating severe PJP, which reportedly requires an effective blood concentration of 150–200 mg/L.

Repeated chest CT on hospital day 15 showed diminished patchy, grid-like, strip-like shadows in both lungs, compared to CT findings on day 8 (Figure 3).
The patient was successfully extubated on day 16 and received high-flow oxygen therapy. After a 10-day ICU stay, he was transferred back to the Oncology Department, where TMP/SMX was prescribed for another 3 weeks. During that period, the patient had no fever or dyspnea, with the ground-glass opacities disappearing on chest X-rays (Figure 6). At follow-up 2 months later, his chest CT revealed no ground-glass opacities, demonstrating a full recovery from PJP and no relapse of pneumonia.

Chest X-rays showed haziness and increased vascular markings. The ground-glass opacities gradually decreased, although some opacities remained in the apical parts.
Discussion
Corticosteroids are frequently used in chemotherapy regimens for their anti-neoplastic, anti-emetic, and anti-allergic effects. Here in our case, the DLBCL patient developed severe PJP after R-CHOP chemotherapy, including the monoclonal antibody rituximab and the corticosteroid, prednisolone. Corticosteroid use is a well-studied risk factor for PJP in HIV-uninfected patients. There is a growing consensus that prolonged high-dose corticosteroid exposure (with a cumulative dose of prednisolone over 1.8 g) could significantly increase the risk of P. jirovecii infection in a dose-dependent manner. 10 Our DLBCL patient was administered prednisolone 100 mg for 5 days for 5 cycles before acquiring PJP (total cumulative dose 2.5 g). Prednisolone use alone markedly increased his susceptibility to P. jirovecii infection. Rituximab has also been identified as an independent risk factor for severe PJP. 11 Rituximab was shown to induce an additional risk of PJP by B-cell depletion. Although CD4+ T cells play a critical role in PJP eradication, B cells are required to generate protective effector and memory CD4+ cells in response to P. jirovecii infection,12–14 and for the early priming of CD4+ T cells for pneumocystis clearance in mice model. 15 It is well-established that a CD4+ T lymphocyte count of fewer than 200 cell/µL correlates with a high incidence of developing PJP, and prophylaxis of PJP is recommended for patients with CD4+ T cell count below this figure. 16 Based on our patient’s high-risk factors for PJP, the rapid progression of respiratory symptoms, the nonspecific diagnostic laboratory indexes and GGOs on chest CT films, PJP was highly suspected.10,17–20
The standard microbiological diagnosis of PJP is based on the detection of P. jirovecii in respiratory samples, including induced sputum, bronchoalveolar lavage, or lung biopsy specimens, through chemical staining. 16 However, the Gomori’s methenamine-sliver staining of the sputum or BAL for P. jirovecii is time-consuming, costly, and unavailable in our hospital. Given the high mortality associated with severe PJP, it is very important to diagnose PJP correctly and commence the anti-PJP treatment early.21,22 To identify the pathogenic organism in our patient as quickly as possible, BAL-mNGS technology was selected.23–25 In recent years, NGS technology has rapidly developed and attracted much attention due to its fast results and high sensitivity to a full range of pathogenic agents, including uncommon ones. Its application in the diagnosis of PJP has been increasingly reported.23,26 In our case, the BAL-mNGS results confirmed the suspected diagnosis of PJP and substantiated that empiric anti-PJP treatment was indicated.
TMP/SMX is the first-line antibiotic for anti-PJP treatment. By interfering with folate’s metabolism, it inhibits the cysts and trophozoite forms of P. jirovecii. 27 Caspofungin is an echinocandin that blocks the formation of pneumocystis cysts by inhibiting the synthesis of β-(1,3)-D-glucan in the cell wall; thus, it has no direct activity against trophozoite forms.28–30 However, caspofungin acts faster than TMP/SMX, which requires more time to achieve a therapeutic steady-state plasma concentrations.31–34 Usually after 3–4 days of TMP/SMX monotherapy for treating suspected PJP, if patient’s clinic condition does not improve, an alternative diagnosis or secondary infection should be immediately considered. 35 Thus, combining caspofungin with TMP/SMX can quickly inhibit the entire life cycle of this fungus. Combination caspofugin with TMP/SMX was recommended as the first-line treatment for severe PJP.27,36 In our case, 48 h after the combined therapy, the patient’s hypoxia under NIV did not ameliorate, and he had to be intubated. Clindamycin was then added to enhance the inhibition of P. jirovecii. The mechanism of action of clindamycin against P. jirovecii is still unclear, though it was shown to target the protein synthesis and replication of bacteria protozoa. 37 Clindamycin combined with TMP/SMX was shown to increase the efficacy for treating severe PJP. 38 The addition of clindamycin to TMP/SMX may be associated with a more rapid clinical response to treatment and facilitate the use of a lower dose of TMP/SMX, reduce the risk of toxicity. Clindamycin may also be used in combination with caspofungin in patients with TMP/SMX intolerance or treatment failure. 39 Caspofungin, clindamycin and intravenous TMP/SMX have been used to treat moderate-to-severe PJP in immunocompromised patients.18,20,27,38 We successfully treated the severe P. jirevocii infection in our DLBCL patient with all three agents.
The common adverse effects of TMP/SMX include rash, nausea, vomiting, liver injury, nephrotoxicity, hyperkalemia, and in more serious cases, neutropenia, thrombocytopenia, and toxic epidermal necrolysis are observed. 40 It has been reported that the estimated incidence of TMP/SMX-related hepatotoxicity is 16.4% among HIV-infected PJP patients in Chinese Taiwan. 41 Our patient developed mild hepatic dysfunction on day 5 of TMP/SMX therapy, so the peak and trough blood TMP/SMX concentrations were measured. The blood concentrations of SMX showed it reached an effective concentration against severe PJP. 32 Considering the concentration-dependent toxicities of TMP/SMX, we decreased the drug dosage and switched to the oral form as the patient’s general condition improved. To summarize, in order to achieve rapid control of the disease and improve the prognosis, we used caspofungin and clindamycin in combination with TMP/SMX to treat severe PJP. Although this strategy seemed aggressive, it has already been trailed in the treatment of HIV-infected patients with PJP in a pilot study. 42 In this small pilot study, combination therapy with caspofungin, clindamycin, and TMP/SMX was associated with reduced mortality in HIV-infected patients with PJP compared with TMP/SMX monotherapy, although the reduction in mortality only achieved statistical significance for patients with mild-to-moderate PJP.
Conclusion
We used triple combination therapy with caspofungin, clindamycin, and TMP/SMX to treat severe PJP in a patient with DLBCL following R-CHOP chemotherapy. Given the high mortality in severe PJP in HIV-uninfected immunosuppressed patients, we adopted an aggressive anti-PJP regimen. With this triple drug regimen, our patient’s respiratory condition was apparently improved, and he was successfully transferred out of the ICU after 1-week of mechanical ventilation support. Importantly, patients with DLBCL receiving R-CHOP chemotherapy are at high-risk of developing PJP and this diagnosis should be considered early in patients with clinical and radiological features suggestive of PJP.
Footnotes
Acknowledgements
We are grateful for the approval from the patient and his family and for the useful comments and suggestions from the anonymous referees.
Data Sharing Statement
The data sets and images supporting the conclusions of this article are included in the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The procedures used in this study adhere to the tenets of the Declaration of Helsinki. We conducted the study based on a protocol approved by the Ethics Review Board of Guangdong Second Provincial General Hospital (Guangzhou, China. IRB number 2022-KY-KZ-314-01).
Informed Consent
Written informed consent was provided by the patient for the publication of the case details and images. Details of the case is published with the approval the Ethics Committee of our hospital.
