Abstract
Diffuse large B-cell lymphoma is the most common histologic subtype of non-Hodgkin lymphoma. Secondary involvement of the heart is seen late in advanced cases, it is uncommon for diffuse large B-cell lymphoma to present as intracardiac mass. A 26-year-old female patient presented with progressive shortness of breath, facial swelling, and lower limb edema. Imaging investigations by echocardiography and computed tomography showed a large right atrial mass that was obstructing the tricuspid valve. Open biopsy was taken, histopathology showed diffuse large B-cell lymphoma. She received six cycles R-CHOP chemotherapy (rituximab, cyclophosphamide, epirubicin, vincristine, and prednisone) with complete resolution of the mass. Diffuse large B-cell lymphoma can present with atypical and uncommon sites such as the heart as the first presentation. Early diagnosis and appropriate management is crucial given the poor outcome with late presentation. A high index of suspicion and the proper investigations is recommended to allow for early intervention and favorable outcomes as what happened with the case under discussion.
Introduction
Diffuse large B-cell lymphoma (DLBCL) is a lymphoid neoplasm originating from B-cells, it is an aggressive form of non-Hodgkin lymphoma (NHL), 1 and accounts approximately 25% of all NHLs, which make it the most common histologic subtype of NHL. 2 Nodal presentation is the most common presentation including lymphonodus, Waldeyer’s ring, and mediastinum. 3 However, mediastinal mass presentation accounts for only 2%–4% of all NHLs. 4
Despite that secondary involvement of the heart is seen in 8.7%–27.2% of documented clinical cases of lymphoma, 2 it occurs mostly in advanced disease or remains silent and often diagnosed post-mortem. It is a very rare scenario for DLBCL to present as intracardiac mass. Many imaging modalities are used for further evaluation including computed tomography (CT), echocardiography, and cardiac magnetic resonance imaging (MRI).4,5 Diagnosis established after histopathology confirmation to start immediately with appropriate treatment. This is a case report of a 26-year-old immunocompetent female patient presenting with gross intracardiac mass and signs of right side heart failure.
Case presentation
A 26-year-old immunocompetent female patient with unremarkable medical history presented to our hospital with progressive shortness of breath, facial and upper limb swelling, and mild lower limb edema for 3 months. She had no history of weight loss, night sweats, or fever. The rest of medical history was unremarkable.
On admission, she was hemodynamically unstable, blood pressure was 85/60 mm Hg, heart rate 116 beats/min, temperature 36.3 °C, and saturation 96% on room air. She was tachypneic with respiratory rate of 30 breath/min. She had facial swelling, raised jugular venous pressure of 6 cm above sternal angle, along with upper limb swelling, and mild lower limb edema. She had distant heart sounds, a diastolic murmur best heard in the left lower sternal border, lung exam revealed decreased breath sounds with dullness on percussion on bilateral basal lung zones. Chest radiograph showed bilateral pleural effusion, wide mediastinum, and bilateral hilar congestion. Electrocardiogram showed sinus rhythm, Q-wave in V1, and low voltage QRS complex with no electrical alternans.
Transthoracic echocardiograph showed a right large atrial mass obstructing the tricuspid valve with severe pericardial effusion. She underwent urgent pericardiocentesis which drained around 1000 cc of serous fluid, glucose 86, negative acid-fast bacilli stain, and no malignant cells were present. Also, thoracentesis from bilateral pleural effusion was done, they drain around 1 L from each side, and it was serous and transudate in analysis. After that, transesophageal echocardiography was done for evaluation of the mass. It confirmed the presence of a large rounded shape mass with a maximum dimension: 6.3 cm × 6.4 cm as seen in the attached picture obstructing the tricuspid valve (shown in Figure 1). Whole-body contrast CT showed a large soft tissue density filling defect almost occupying the right atrium and almost occluding the distal superior vena cava, with no clear cleavage line on to the adjacent mediastinal structure. It also showed a large anterior mediastinal mass 7 cm × 4 cm, another heterogeneous mass measuring 13 cm × 13 cm ×7.5 cm was seen in the right pelvic cavity likely to be originated from the right adnexa (shown in Figure 2).

Transesophageal echocardiography showed presence of a large rounded shape mass with maximum diameter: 6.3 cm × 6.4 cm on the right atrium obstructing the tricuspid valve.
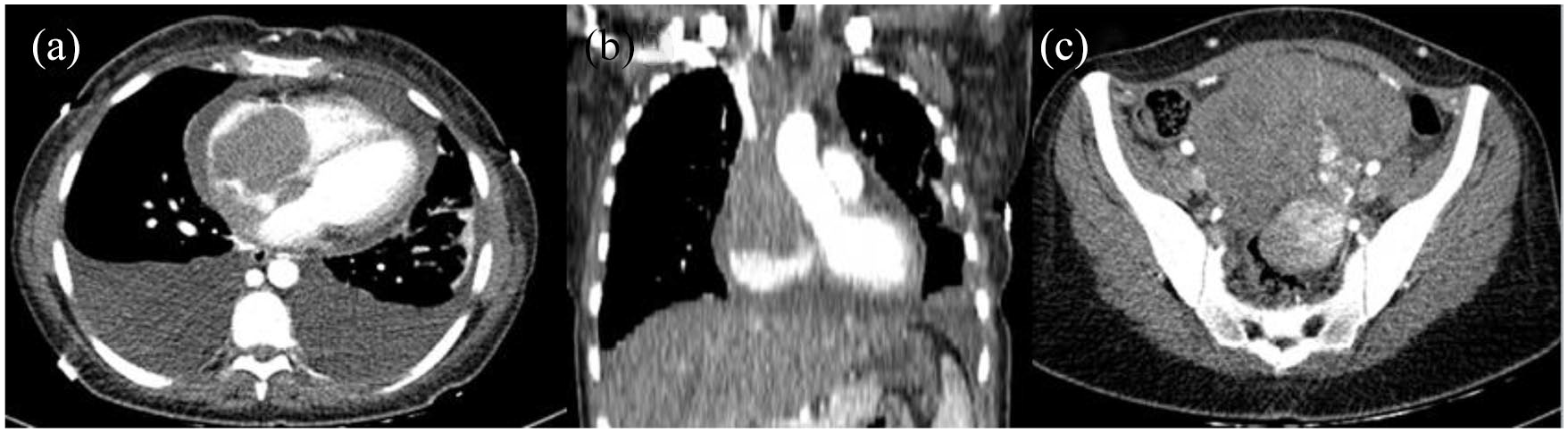
CT scan showed a large soft tissue density filling defect almost occupying the right heart and overriding the cardiac root, indenting the pulmonary veins and almost occluding the distal SVC as seen in (a) and (b). (c) About 13 cm × 13 cm × 7.5 cm heterogeneous mass lesion seen in the right pelvic cavity.
Open biopsy via thoracotomy was taken from the anterior mediastinal mass and sent for histopathology which revealed DLBCL. We defer further investigations including adnexal mass biopsy, positron emission tomography (PET) scan, and bone marrow as the patient needs cardiac stabilization before proceeding for further investigations.
The patient was started on R-CHOP (rituximab, cyclophosphamide, epirubicin, vincristine, and prednisone) chemotherapy in an urgent basis as its chemosensitive. She received the first cycle during hospitalization with a good response evident by improvement of respiratory distress, facial and upper limb swelling within few days, along with a dramatic reduction in the size of cardiac mass and adnexal mass. She received six cycles with echocardiograph follow-up between the cycles showing resolution of the cardiac mass. The last cycle of chemotherapy was in May 2019, during which she received four doses of intrathecal methotrexate as prophylaxis for central nervous system (CNS) involvement. It was given due to high CNS-international prognostic index score with high lactate dehydrogenase (LDH) (468 U/L), multiple lymph nodes, and ovarian involvement. Follow-up imaging showed a residual mass in the mediastinum and pelvis. PET scan showed no evidence of hypermetabolic lymphomatous disease. After that, she received radiotherapy for both the mediastinum and the pelvis, about 40 gray equivalents delivered for both (2 gray with 20 fractions). Currently, the patient is in complete remission, in serial follow-up every 1–2 years. Last follow-up 2 years after diagnosis with PET scan showed no evidence of any hypermetabolic active lymphomatous lesion in the body.
Discussion
This case represents a rare presentation of DLBCL, as mass inside the right atrium was the first presentation. In our case, early echocardiography was diagnostic for the right atrial mass blocking the tricuspid valve, although other cardiac imaging could be of diagnostic value. Echocardiography is generally non-invasive, simple, and extremely useful cardiac test for the diagnosis of multiple cardiac pathology. 6 Initial clinical picture before CT scan was compatible with many differential diagnosis. It includes atrial myxoma as our initial thought, rhabdomyosarcoma, atrial thrombus, and lymphoma. Clinical presentations of intracardiac mass are not specific; it includes dyspnea, lower limb edema, signs of right-sided heart failure, intracardiac thrombus, peripheral embolization, valvular heart disease, and pericardial effusion.7,8 Other signs and symptoms suggestive of malignancy such as weight loss, night sweats, and fever could be presented. However, it was absent in this case which guided us to think of a benign tumor rather than malignancy.
Physical exam showed picture of right-sided heart failure along with diastolic murmur best heard in the tricuspid area; this gave us a hint that the level of obstruction in the area of tricuspid valve which was confirmed by echocardiography. According to the clinical presentation and initial transthoracic echocardiography of pending cardiac tamponade and hemodynamic instability, our first decision was to stabilize the patient by removing pericardial and pleural fluid. It was followed by transesophageal echocardiography. It is well-known that transesophageal echocardiograph gives more information of intracardiac masses; it gives better visualization, extension, and attachment of the atrial mass. 4 CT scan is another modality which can demonstrate the primary malignancy invading the heart as in our case. 7 Imaging investigations confirmed primary mediastinal mass invading the heart forming intracardiac mass in the right atrium. Direct extension and infiltration of the mass to the heart from the mediastinum is a well-known mechanism. 9
Tissue biopsy was the core element in the diagnosis of this patient; thoracotomy was done previously for this purpose. However, less invasive procedures like percutaneous or fluoroscopy-guided biopsy are preferred for pathological examination. 7 In our case, there were no accessible lymph nodes seen on CT scan. Pericardial and pleural fluid was negative for malignant cells. After multidisciplinary team discussion, and taking consideration of hemodynamic instability of the patient mainly from the mass obstructing the tricuspid valve. Decision was to go for open heart surgery to stabilize her hemodynamic state by relieving the obstruction and also to get biopsy. Intraoperatively, it was unresectable tumor; biopsy was taken. Histopathology showed large cell diffuse lymphoma, non-germinal center B-cell, it was positive for cluster of differentiation (CD)20, leukocyte common antigen (LCA), and CD23. It was negative for CD30 and placental alkaline phosphatase (PLAP) immunostain. Morphologic features and CD23 were consistent with primary mediastinal DLBCL. Lymphoma presenting as intracardiac mass is a very rare. It could be a primary cardiac lymphoma or mediastinal mass with direct extension and infiltration into the right atrium giving a picture of intracardiac mass.3,4,10,11 The prognosis of both subtypes is poor due to rapid progression and invasion of the disease with detection usually in the late stages.12,13
Chemotherapy is considered as the standard treatment of lymphoma presented with superior vena cava syndrome which is very close to our case, and it has equivalent outcome in achieving symptomatic relieve when it compared with chemoradiotherapy, especially in this chemosensitive tumor. 6 Fertility preservation before chemotherapy was thought. However, the patient declined any method, and her critical condition precludes any fertility preservation. First-line treatment regimen with R-CHOP has shown a good response rate.14,15 Other possible option was R-EPOCH chemotherapy. 16 However, as she was symptomatic and after discussion with the patient and family about the side effects and expected outcome, the decision was to start with R-CHOP chemotherapy. Surgery is usually indicated for a patient who is hemodynamically unstable by improving blood flow to the lungs in case of right ventricle outflow obstruction. 17 Also, surgical salvage is considered in case of advance intrathoracic disease when chemotherapy or radiotherapy failed to achieve response. 6
The patient also presented with a pelvic mass seen on CT scan. Ideally, the biopsy should be taken from the mass. However, as we started the patient on urgent chemotherapy as she was severely symptomatic, we deferred further investigation until follow-up. The mass completely resolved by chemotherapy which was expected with the same histopathology etiology.
DLBCL with heart involvement has a very poor prognosis; it has a 10% survival rate of 9–12 months if not treated. 8 Although it has a high mortality rate, it could be cured with intense chemotherapy. Post-treatment surveillance should include PET/CT to confirm response and further follow-up imaging as clinically indicated. 18 Early relapse or refractory disease is managed by further chemotherapy and allogeneic stem cell transplantation. 4 Last guidelines recommended routine follow-up by history, physical exam, and laboratory investigations every 3–6 months for 5 years, while imaging follow-up no more than every 6 months for 2 years, except if clinically indicated. 19
Conclusion
Intracardiac mass is very rare presentation of primary mediastinal DLBCL. It is very important to establish an early diagnosis and starting treatment as soon as possible as it affects outcomes. In this case, conventional chemotherapy without surgery with good follow-up was effective and lead to complete response.
Footnotes
Acknowledgements
The authors are very thankful to the cardiac surgery team, pathology team, and radiology department team at our hospital for providing support for completing this report. The authors also acknowledge research square that the article has been accepted for publication as preprint.
Author contributions
Y.D., Y.I., and S.M. managed her cardiac condition, pericardiocentesis, and transthoracic and transesophageal echocardiography. They contributed to the diagnosis and management of this patient. R.O., a hemato-oncologist who followed up her treatment protocol and put a management plan. All authors participated in the drafting of the manuscript. All authors read and approved the final manuscript.
Availability of data and materials
All data supporting the study are presented in the manuscript or available upon request from the corresponding author of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
