Abstract
Thromboangiitis obliterans, or Buerger’s disease, is a relatively rare nonatherosclerotic, segmental inflammatory and obliterative vascular disease that affects the small- and medium-sized arteries, veins, and nerves. In the acute phase, the lesion presents as an inflammatory, nonsuppurative panarteritis or panphlebitis with vascular thrombosis without necrosis. In the late stage of the disease, the thrombus becomes organized leading to varying degrees of recanalization and subsequent gangrene and amputation. There have been rare reports of thromboangiitis obliterans with involvement of the gastrointestinal trace and even more unusual is the occurrence of this manifestation of disease in women. Here, we report a case of a 45-year-old female patient with a history of thromboangiitis obliterans who presented with ischemic colitis.
Keywords
Introduction
Thromboangiitis obliterans (TAO), or Buerger’s disease, is a non-arteriosclerotic, segmental, progressive, inflammatory vaso-occlusive disorder that primarily affects arteries and veins of small to medium caliber and surrounding neural elements. The disease usually occurs in young male smokers.1,2 TAO is more prevalent in the Middle East and Far East than in North America and western Europe. 2 The incidence of TAO in women is low 3 and involvement of the gastrointestinal (GI) tract is exceedingly rare. The rise of incidence of TAO in women is attributable to the increased incidence of women cigarette smokers in the past few decades.3,4 Use of or exposure to tobacco is central to the initiation and progression of the disease.5,6 The strong correlation with cigarette use is thought to involve a direct idiosyncratic toxicity caused by a component of tobacco or an immune response to the same agents that have modified host vascular wall proteins. 7 Lesions in the brain, heart, and abdominal viscera are rare. TAO differs from other forms of vasculitis in its pathophysiology: the resultant thrombus is highly cellular and inflammatory with relative sparing of the blood-vessel wall with normal acute-phase reactants and autoantibodies. 8 The lesion starts as an inflammatory, nonsuppurative panarteritis or panphlebitis with vascular thrombosis without necrosis. The vascular thrombus becomes organized at the late stages in the disease, leading to varying degrees of minor recanalization of the thrombi with subsequent possible gangrene and amputation. The histopathological findings vary according to the duration of disease and findings are most likely to be diagnostic in the acute phase of the disease, as in the end-stage phase of the disease, only an organized thrombus with associated fibrosis is seen.9–11 Throughout all stages of the disease, the normal structure of the vessel wall, including the internal elastic lamina, generally remains intact, which distinguishes TAO from other types of systemic vasculitis, in which the internal elastic media and lamina are disrupted. 12 Signs and symptoms of the disease are directly proportionate to the degree of vessel and neural involvement and include pain, edema, recurring episodes of thrombophlebitis, claudication, skin changes, tissue malnutrition, and gangrene of the extremity. 13 The Allen test in patients with leg ulceration is used to assess the circulation in the hands and fingers, and an abnormal test is highly suggestive of TAO.12,14 However, an abnormal result can be indicative of other types of small-vessel occlusive disease of the hands. A complete serological profile with acute-phase reactants and a full antibody and hypercoagulability panel helps to differentiate TAO from these entities. Traditional diagnosis is based on the fulfillment of Shionoya’s five criteria: smoking history, onset before the age of 50 years, infrapopliteal arterial occlusive disease, upper limb involvement or phlebitis migrans, and absence of atherosclerotic risk factors other than smoking. 15 The diagnostic criteria of Olin 12 include age below 45 years, current or recent history of tobacco use, presence of distal-extremity ischemia indicated by claudication, pain at rest, ischemic ulcers or gangrenes and documented by non-invasive vascular testing, exclusion of autoimmune diseases, hypercoagulable states and diabetes mellitus, exclusion of proximal source of emboli by echocardiography or arteriography, and consistent arterographic findings in the clinically involved and non-involved limbs. However, these five diagnostic criteria are not universally accepted, and angiographic finding is not pathognomonic. In addition, as there is no specific diagnostic test or serologic finding, the diagnosis of TAO is primarily clinical. Discontinuation of cigarette smoking is the only proven tertiary prevention,4,16–18 as no forms of therapy are definitive. The rare cases of TAO with involvement of the GI tract have manifested in the small intestine,19–21 and cases of TAO causing ischemic colitis with perforation are scarce. 22 The majority of confirmed cases of visceral intestinal TAO occur in men13,19,21–52 (Table 1). We present a unique case of a 45-year-old female patient with a history of TAO who presented with ischemic colitis.
Comprehensive comparison of case reports of Buerger’s disease with gastrointestinal manifestations.
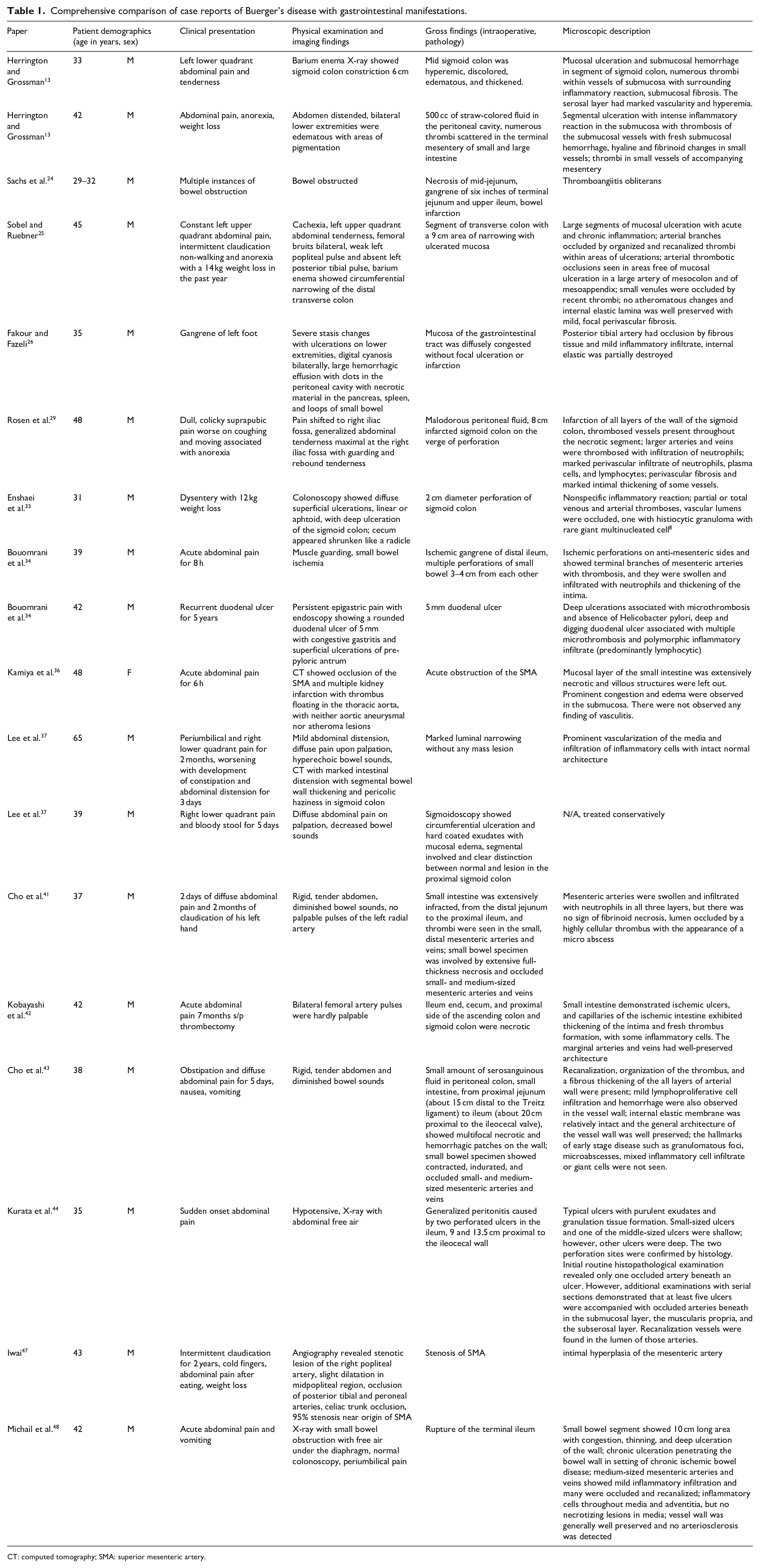
CT: computed tomography; SMA: superior mesenteric artery.
Case report
A 45-year-old Caucasian female with a past medical history significant for Buerger’s disease complicated by toe and finger amputations, possible systemic lupus erythematosus/rheumatoid arthritis, chronic obstructive pulmonary disease, peptic ulcer disease, hypothyroidism, and drug and tobacco abuse (one pack per day since her teenage years) presented with altered mental status and was found to be hypoxic and hypotensive. Laboratory work was significant for hypoglycemia, hyponatremia, hypocalcemia, lactic acidosis, and acute kidney injury. The patient was initially diagnosed with Buerger’s disease at an outside institution in 2017 with a right second finger wound which required amputation and first presented to our institution with complications of her disease in 2019 after partial amputation of her fingers. At this time, her right wrist brachial index and forearm Pulse Volume Recording (PVR) waveforms indicated mild arterial occlusive disease with non-pulsatile digits and left forearm PVR waveforms indicated mild arterial occlusive disease despite normal wrist brachial index with non-pulsatile digits. Per chart review, the patient endorsed bilateral calf claudication after walking a few hundred feet, relieved by rest. Chest X-ray was significant for pneumoperitoneum. The patient was taken immediately for exploratory laparotomy and was found to have a transverse colon perforation which was repaired primarily. She had other suspicious lesions, so was left open for re-evaluation. On take back, the patient was found to have multiple punctate lesions of full-thickness necrosis every few centimeters throughout the colon, most prominently in the ascending colon (Figure 1). A total abdominal colectomy with end ileostomy was performed given these findings. Upon gross examination, full-thickness transmural elliptical-shaped perforations were noted 12 cm from the proximal margin with nearby areas of ulceration (Figure 2). There were longitudinal areas of dimpling with fibrosis near the anastomotic site (the patient had a previous primary repair of the colonic perforation). Adjacent to the anastomotic site was an area of ulceration with overlying granulation tissue, and several similar punctate ulcers were noted distal to this area. Microscopic examination confirmed the presence of multiple foci of ulceration with granulation tissue as well as scattered medium and small-sized vessel vasculitis, consistent with the patient’s known Buerger’s disease (Figures 3 and 4). The remainder of the unaffected colon showed signs of early TAO with panarteritis and inflammatory infiltrate, whereas the areas of ulceration with overlying granulation tissue were indicative of end-stage disease. Grocott’s methianine silver special stain for fungi was negative and elastic stain highlighted the involved vessels (Figures 5 and 6).

(a, b) The patient was found to have multiple punctate lesions of full-thickness necrosis every few centimeters throughout the colon, most prominently in the ascending colon (black arrows).

Macroscopic examination after formalin fixation reveals transmural elliptical-shaped perforations were noted 12 cm from the proximal margin with nearby areas of ulceration (black arrows).
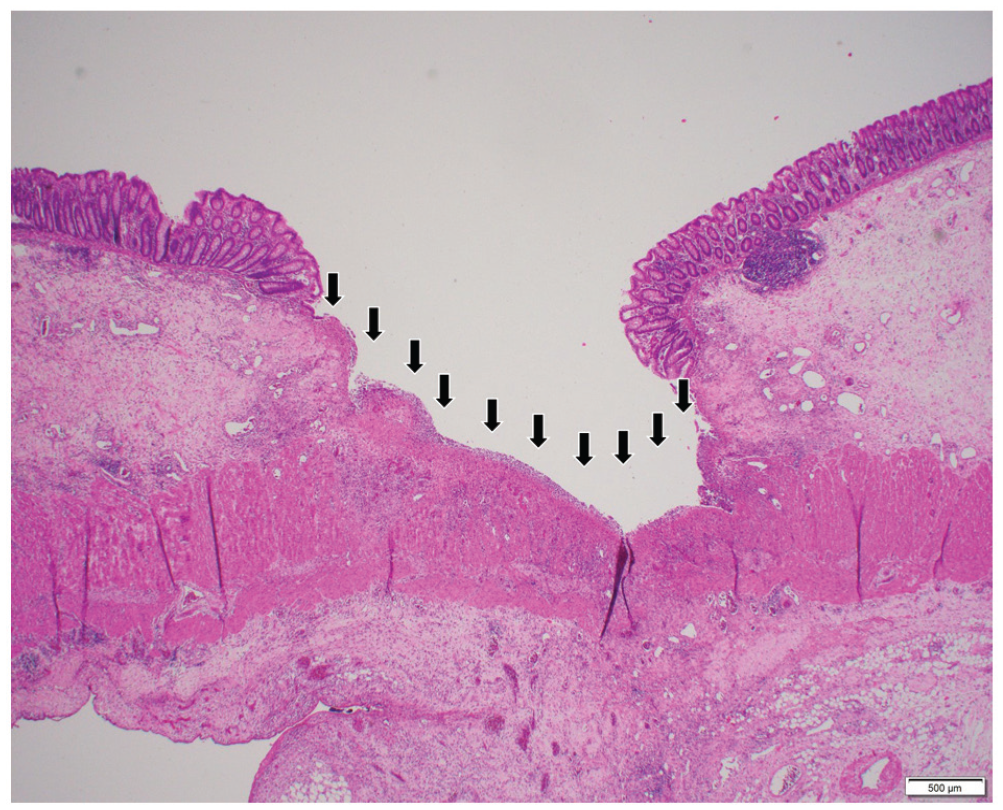
Focus of ulceration with granulation tissue (black arrows).

(a) Vasculitis (20× magnification), (b) arteritis (40× magnification), and (c) venulitis (40× magnification).

(a, c) Mucosal ulcer with vasculitis (black arrows, 4× and 10× magnification, respectively). (b, d) Elastic special stain highlights the involved vessels (black arrows, 4× and 10× magnification, respectively).

(a) Vasculitis (arteritis) almost occluded (40× magnification) with elastin staining shown in (b, c). Venulitis (20× magnification) with elastin staining shown in (d).
Discussion
TAO, or Buerger’s disease, is a segmental, progressive, inflammatory vaso-occlusive disorder that primarily affects small- and medium-sized arteries, veins, and nerves of the extremities in young male smokers. This type of vasculitis differs from other systemic vasculitidies, involving small or medium-sized vessels that can manifest in the colon in its segmental nature and lack of immune complexes or vascular wall involvement. Histologically, systemic vasculitis presents as an inflammatory infiltrate in the wall of dermal or subcutaneous vessels, which may be neutrophilic, lymphocytic, or granulomatous mediated by immune complexes. Often, there is microscopic red blood cell extravasation and variable fibrinoid necrosis of vessel walls as well secondary changes in overlying epidermis and sweat glands. The development of TAO in the acute phase is characterized by a lesion that presents as an inflammatory, nonsuppurative panarteritis or panphlebitis with vascular thrombosis without necrosis. The thrombus becomes organized in the late stage of the disease leading to varying degrees of recanalization and consequent gangrene and amputation. The incidence of TAO in women is low 3 and involvement of the GI tract is exceedingly rare. The rare cases of TAO with involvement of the GI tract have manifested in the small intestine,19–21 and cases of TAO causing ischemic colitis with perforation are scarce, 22 and the majority of confirmed cases of visceral intestinal TAO occur in men.13,19,21–32,34–52
Our patient is unique with features of TAO. While only the upper extremities were reported to be affected and we do not have the full diagnostic workup to support the provided history of TAO, given the histology and fulfillment of Olin’s criteria, we can attribute the patient’s diagnosis to Buerger’s disease after excluding other causes of her vasculitis. However, she may have suffered from another unknown vasculitis which mimicked TAO. The patient has an unconfirmed medical history of two inflammatory and autoimmune diseases (rheumatoid arthritis and lupus) and a history of intravenous (IV) drug abuse and tobacco use with transverse colon perforation with subsequent septic shock. However, the histological findings and serological studies support TAO as the cause of this patient’s ischemic colitis rather than other types of systemic vasculitis given the intact elastic lamina, the lack of serological findings and immune complexes, the segmental nature, and lack of necrosis. Given the brief interval between surgeries, the patient had few serosal adhesions or areas of advanced ischemia that could have led to the multifocal ischemic colitis. The patient had an open wound that could have contributed to her initial clinical presentation. She is one of the first reported female patients with TAO with intestinal manifestation and subsequent colonic perforation. The patient passed away 2 months after the surgery from septic shock.
Conclusion
This case demonstrates the importance of clinical correlation with gross findings especially in rare presentation of a disease.
Footnotes
Acknowledgements
The authors thank the surgical team for their contributions.
Author contributions
M.B. performed the macroscopic examination of the resected specimen and drafted the manuscript. R.D.B. performed the surgery and edited the manuscript. S.A.D. confirmed the histologic diagnosis and led the team.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
