Abstract
The predominant organic system involved in multisystem inflammatory syndrome in children associated with COVID-19 is the gastrointestinal system, which is observed in almost 90% of patients. Gastrointestinal symptoms can mimic acute appendicitis. There have been a few cases of misdiagnosed multisystem inflammatory syndrome in children associated with SARS-CoV-2 as appendicitis, and a few concomitant cases of a multisystem inflammatory syndrome associated with acute appendicitis during the COVID-19 pandemic. Here, we present the case of an 11-year-old girl who presented to our Intensive Care Unit with a 2-day history of fever, generalized abdominal pain, and vomiting. The clinical findings resulted in a clinical suspicion of acute appendicitis and subsequent surgery. While postoperatively, she became critically ill, and she was diagnosed with the multisystem inflammatory syndrome in children associated with COVID-19. When diagnosing children with acute appendicitis, healthcare professionals, especially pediatricians and surgeons, must pay attention to the multisystem inflammatory syndrome linked to the SARS-CoV-2 infection.
Introduction
The multisystem inflammatory syndrome in children (MIS-C) type of SARS-CoV-2 infection is now a well-known condition, 1 which was first thought to be comparable to atypical Kawasaki disease (KD). 2 Accordingly, this new disease entity has been called Multisystem Inflammatory Syndrome associated with COVID-19 (MIS-C) as per the World Health Organization (WHO) 3 and Pediatric Inflammatory Multisystem Syndrome temporally associated with COVID-19 (PIMS-TS) as per the Royal College of Pediatrics and Child Health case definition (RCPCH). 4
A systemic review and meta-analysis found that overall gastrointestinal (GI) symptoms in the MIS-C were 0.82 (0.71–0.89) and abdominal pain was 0.68 (0.62–0.74). 5 Interestingly, a presentation that resembles acute appendicitis (AA), mesenteric adenitis, and terminal ileitis has only been described on rare occasions and may be the initial sign of the illness, increasing the risk of misdiagnosis.
KD and MIS-C share many clinical and pathological features associated with AA. KD can have more severe abdominal features mimicking AA. Neither of the children requiring appendectomy had fecoliths, implying vasculitis or inflammation. 6
It is found that AA, SARS-CoV-2 infection, and the MIS-C virus share a correlation. This topic has been discussed extensively during the COVID-19 pandemic. 7
Moreover, early diagnosis of MIS-C is recommended to prevent needless surgery owing to a higher risk of perioperative morbidity when patients have a clinical presentation matching a surgical abdomen. As it was also reported from Wuhan, China, postoperative patients with COVID-19 infection have a 20.5% chance of dying. 8 Children with COVID-19 have reported receiving non-operative therapy for simple AA with antibiotics.9,10
A recently observed new variant, mimicking AA,11–13 prompts abdominal imaging and surgical evaluation. Pediatric surgeons must first be aware of the GI symptoms of MIS-C to diagnose it correctly. MIS-C should be suspected even when it appears similar to simple appendicitis, especially in areas severely affected by the pandemic or in patients previously exposed to the disease. 14
Although there has been no national data published regarding MIS-C and associated conditions or manifestations, we have found that GI manifestations are present in almost all cases treated as MIS-C at our Pediatric Clinic, a referential center in Kosovo. In a previously reported case of MIS-C and aseptic meningitis, vomiting and diarrhea were also noted as clinical manifestations. 15
To the best of our knowledge, the present case is the first reported case of coincidental or mimicking AA in children in Kosovo. Therefore, the present report aimed to describe a case of an 11-year-old Kosovo girl who presented with coincidental or mimicking MIS-C linked with SARS-CoV-2 infection.
Case
A previously healthy 11-year-old girl presented to our intensive care unit (ICU) with a fever (38.7°C), vomiting, and generalized abdominal pain. She had no unusual history of drug allergies, medical history, or surgical procedures. Her family lives in Rahovec, a town located in the district of Prizren in western Kosovo. The rest of the patient’s history was unremarkable.
On admission, the patient had a fever of 38.7°C, looked unwell, and was lethargic, and she had a rash on her palms and trunk with no conjunctivitis. No lymphadenopathy was noted.
The patient had severe drowsiness and could be aroused only by moderate stimuli. Meningeal signs were negative. The patient’s respiratory rate was 25/min, and her oxygen saturation was 93% in ambient air. On auscultation, the patient’s breathing was harsh with crackles during inspiration. She was tachycardic (heart rate (HR) 135 beats per minute) with no associated murmurs at heart auscultation, but she had hypotension (blood pressure (BP) 80/42 mm Hg).
The patient’s abdomen was tender to palpation, especially in the right lower quadrant. No other specific findings were noted on the systemic examination.
The initial laboratory tests showed a white blood cell count of 13.5 × 103/μL (segmented neutrophils, 94.5%) and a red blood cell count of 3.5–5.8 × 106/mm3. The erythrocyte sedimentation rate, serum C-reactive protein (CRP), and procalcitonin concentrations were significantly increased to 82 mm/h, 298.5 mg/L, and 18.45 mcg/L, respectively (Table 1). The patient tested negative for SARS-CoV-2 by both rapid PCR and RT-PCR.
Laboratory values and trends during hospital stay.

Abdominal ultrasound showed an enlarged appendix of 11.2 mm in diameter associated with a moderate amount of fluid in the pelvis. Due to suspected appendicitis, the surgeon was consulted, and the patient underwent an emergency laparotomy appendectomy. The patient tolerated the procedure well and was transferred back to the ICU. Her initial postoperative course was remarkably toxic in an acute distress condition and with hemodynamic implications. She had tachycardia (139 beats per minute) and hypotension (BP 76/39 mmHg), and fractional shortening was observed in the echocardiogram. Because there was no improvement in her BP despite aggressive fluid resuscitation (60 mL/kg), dopamine, a vasoactive drug, was used, and dosages were adjusted according to the patient’s condition. Due to the low oxygen saturation in ambient air (88%–90%), the patient was placed on oxygen therapy (2 L/min O2) via a nasal cannula.
Because homogeneous bilateral paratracheal and paracardial spots were noted on the chest X-ray, suggesting bronchopneumonia (Figure 1), the patient was started treatment with ceftriaxone and amikacin, which was later switched to imipenem.

Chest X-ray shows area of reticulo-nodular opacities, indicating the presence of bronchopneumonia.
Despite the negative tests for SARS-CoV-2, we found that the patient had a positive history of contact. Based on more detailed anamnesis, we found that her grandfather was diagnosed with COVID-19 and was treated a month earlier. She was not vaccinated and had no personal or family history of allergic reactions, vasculitis, autoimmune disorders, cardiac disease, diabetes, or hereditary disease.
Several features of our patient’s presentation raised concerns for MIS-C associated with COVID-19. Therefore, a serologic test was performed, resulting in a positive serology of SARS-CoV-2 (immunoglobulin G (IgG)). In addition, ferritin, IL6, high-sensitivity troponin, and D-dimer were all elevated (Table 1). Because the patient had a high D-dimer level, enoxaparin, an anticoagulant, was initiated. Due to our concern for MIS-C-like illness, she was treated with IV immunoglobulin (IVIG; 2 g/kg) divided between Days 2 and 3 of the hospital stay. According to the suggested treatment courses for pediatric patients with MIS-C, 325 mg of aspirin per day was also administered.
On the second day of hospitalization, the patient’s general condition worsened, and she continued to be febrile and became anemic and more toxic. Because her hemoglobin level decreased from 11.5 to 6.9 g/dL, a red blood cell transfusion was administered. In the absence of a response to IVIG, a pulse dosage of systemic corticosteroids (30 mg/kg daily methylprednisolone) was given as a second-line treatment. Re-evaluation of the emerging shock revealed an aggravation of heart dysfunction. An echocardiogram revealed a slight decrease in left ventricle (LV) function, septal hypokinesia, and an ejection fraction (EF) of 30%. Dobutamine was added as the second vasoactive drug.
On the following day, the patient had favorable progression; the vasoactive drugs were discontinued after 72 and 48 h. On hospital day 4, the patient was afebrile, and her clinical symptoms improved. Arterial pressure was stable without inotropes.
No pathogenic agents were detected in the patient’s blood, sputum, feces, or urine cultures.
The histopathological examination resulted in the diagnosis of catarrhal appendicitis (Figure 2).
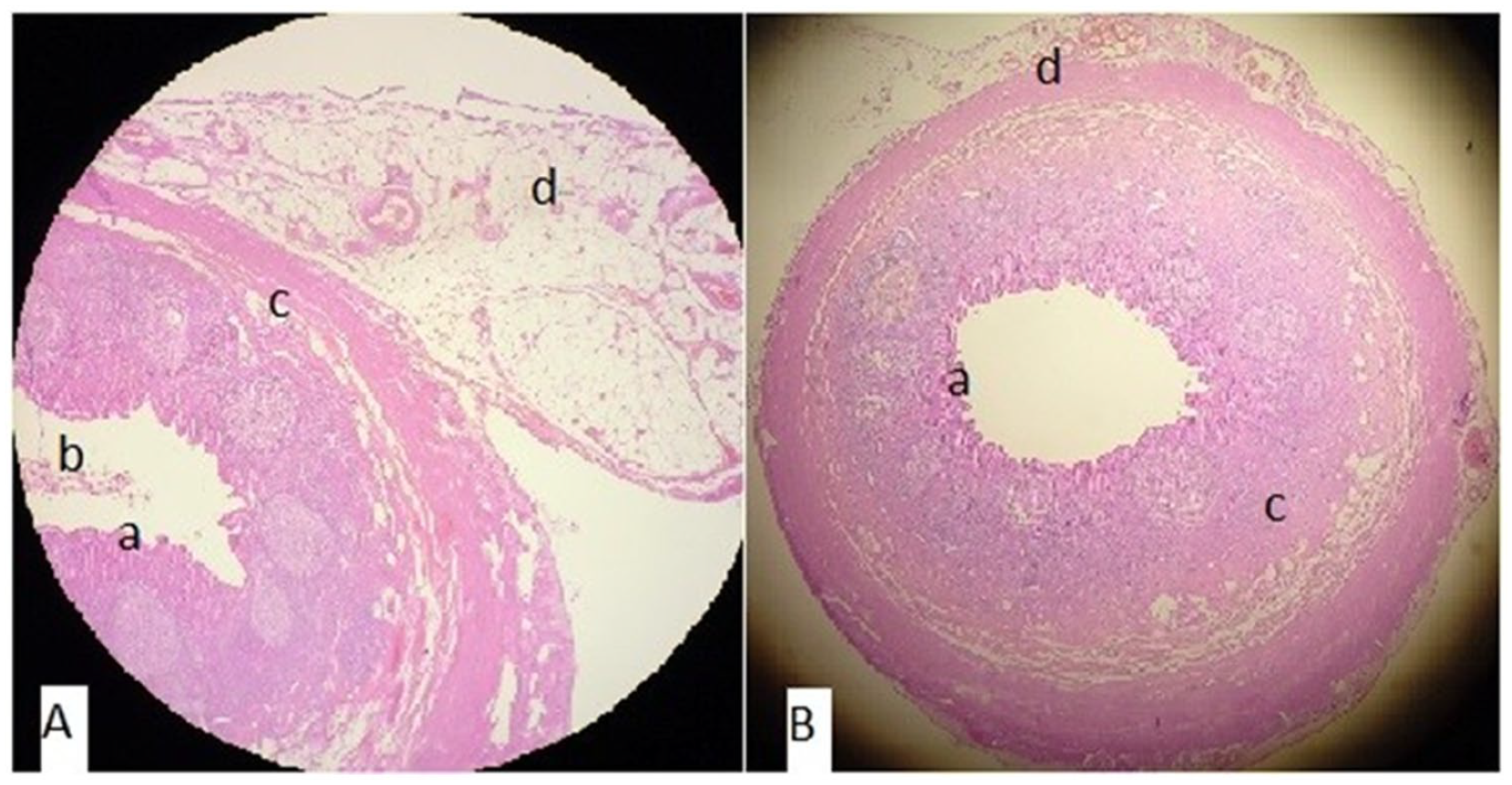
Microscopic images of catarrhal appendicitis. A and B cross-section of vermiform appendix showing (a) focal mucosal erosion, (b) luminal debris, (c) minimal inflammatory infiltrate only on the submucosal layer, and (d) serosal vascular congestion. (Magnification x100).
Moreover, the patient’s D-dimer showed a downward trend, and her troponemia had resolved. At the time of discharge, the patient’s inflammatory parameters were normal. According to the patient’s echocardiogram, her LV function was improved, demonstrating normal biventricular function. No aneurysms were observed in the proximal coronary artery system.
On hospital day 12, the patient was discharged after the improvement of symptoms with advice to take a low dosage of aspirin (3 mg/kg). During a follow-up outpatient visit after 2 weeks, the patient’s blood tests had normalized, while the COV-2 IgG was elevated to 84 (reference level of 1.0). The patient’s abdominal and cardiac ultrasounds were normal. Follow-up outpatient visits were planned every 3 months for 1 year, including echocardiography, urine analysis, and evaluation of her general condition. The patient has been doing well since discharge without any short-term complications.
Discussion
The case series reported first in London and then in Bergamo helped to define MIS-C and to differentiate it from KD.1,2 Even though cardiovascular system affection is the most life-threatening, the predominant organ system involved in MIS-C is the GI system, which is observed in approximately 90% of patients. 7 According to the available literature, it has been shown that up to 30% of patients with MIS-C present with an acute abdomen with clinical features suggestive of appendicitis.16,17 Our patient presented with clinically suspected AA, and an appendectomy was performed. The presence of postoperative worsening with depressed cardiac function, multiorgan involvement, and higher inflammatory markers suggested the possibility of MIS-C.
After conducting an immunological study in patients with AA, Rubér et al. described the pathophysiological relationship between AA and SARS-Cov-2-linked MIS-C; they reported that patients with histopathologically AA are characterized by an increase in inflammatory markers, which leads to neutrophil attraction to the site of infection by promoting epithelial, endothelial, and stromal cells to produce activating cytokines and chemokines.18,19 In the present case, microscopic findings of the vermiform appendix showed focal mucosal erosion, luminal debris, inflammatory infiltrate on the submucosal layer, and serosa vascular congestion.
During the COVID-19 pandemic, many cases of MIS-C associated with AA and a few cases of MIS-C misdiagnosed as appendicitis have been reported. 6 Yock-Corrales et al. 20 reported a small percentage of children who underwent surgery but had no intraoperative findings of appendicitis, suggesting that the inflammatory response is the cause of pain rather than appendicitis.
In the present case, the findings resulted in a clinical suspicion of AA with subsequent surgery. As in the present case, Sturgill et al. 21 presented a series of three children diagnosed with appendicitis and MIS-C related to SARS-CoV-2; all patients were initially admitted with a diagnosis of appendicitis and had an appendectomy before the further diagnosis of MIS-C. One and two cases from two-case series reported from South Africa and Indonesia, respectively, were treated nonsurgically and only received systemic antibiotics and antiviral medication.22,23
Tullie et al. 24 presented a case series and highlighted that children with MIS-C associated with COVID-19 may exhibit clinical characteristics suggesting appendicitis. Using advanced radiological procedures, however, a differential diagnosis was made for the children in the London series, and operative intervention was subsequently abounded. Anyway, both sonography and CT scanning are unable to accurately distinguish between MIS-C and appendicitis in children with abdominal symptoms and MIS-C. 25
Similar to the present case, Jackson et al. 26 presented a case of a 9-year-old girl who was hospitalized for AA and underwent a laparotomy distal ileum resection, but she was diagnosed with MIS-C post resection.
Vansevičienė et al. 27 aimed to distinguish regular appendicitis cases from MIS-C and to suggest the best clinical and laboratory criteria for this differentiation; they found that MIS-C can be suspected even when clinical data and performed tests suggest AA.
By presenting two cases, Anderson et al. 28 provided additional insight into which concomitant infections with AA and COVID-19 may result in MIS-C. In addition, Grynspan and Rabah 29 reported that 94 out of 877 appendectomies had AA before surgery, but the pathology report showed no signs of mucosal ulceration or acute inflammation.
Epidemiological, serological, and immunofluorescence studies have suggested a viral role in AA. Even though the diagnostic tests for identification of viral infection of the appendix by immunohistochemistry (IHC) and RT-PCR were lacking in the present case, Serra et al. 12 have highlighted the importance of these diagnostic tests in reinforcing the hypothesis that the direct effect of SARS-CoV-2 on tissues is involved in the pathogenesis of MIS-C.
The predominance of GI manifestations and the lack of systemic signs in some patients with MIS-C during the initial presentation may produce difficulties in recognizing MIS-C in patients who have concomitant AA.
In the present case, based on clinical presentation, laboratory findings, and additional examinations, the diagnosis was achieved, and the patient was successfully treated with therapy according to clinical guidelines for MIS-C associated with SARS-CoV-2. 30
Conclusion
For the present case study, it was difficult to infer that coincidental or mimicking AA was associated with SARS-CoV-2-related MIS-C due to the lack of IHC and RT-PCR analyses of appendix tissues. During the COVID-19 pandemic, surgeons should consider this new alternative diagnosis for MIS-C in their evaluation of young patients with abdominal symptoms that point to AA.
Footnotes
Author contributions
A.R.-M. contributed to all parts of the manuscript such as conception, design, data collection, analyses and interpretation, literature review, and writing.
A.K.-S. contributed to the design, data collection, analyses, and interpretation.
A.B. contributed to the literature review and writing.
P.G. contributed to the conception and data collection.
A.P. contributed to analyses and interpretation, literature review, and writing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent for the publication of this case report and accompanying images was obtained from the patient’s father. A copy of the written consent is available for review from the editor of this journal.
