Abstract
Introduction:
The response to vaccination against the virus that causes severe acute respiratory infection syndrome coronavirus 2 is lower in renal transplant recipients than in the general population. The data obtained from Latin America showed reduced immunogenicity under inactivated virus vaccination schedules and messenger ribonucleic acid platforms.
Methods:
A retrospective cohort study including renal transplant recipients from Colombia with a two-dose vaccination schedule against severe acute respiratory infection syndrome coronavirus 2 with Pfizer, AstraZeneca, Moderna, Jansen, and Sinovac vaccines between March 1, 2021 and December 1, 2021, was carried out with a follow-up period to evaluate outcomes until May 2022. The outcomes correspond to the titers of immunoglobulin G antibodies against the receptor binding domain of the severe acute respiratory infection syndrome coronavirus 2 spike and a composite outcome of mortality, general, and intensive care unit hospitalization.
Results:
In total, 215 renal transplant recipients with two doses of vaccination for severe acute respiratory infection syndrome coronavirus 2 during the predominance of the Omicron variant in Colombia were included, with the measurement of immunoglobulin G antibody titers against the receptor binding domain of the severe acute respiratory infection syndrome coronavirus 2 spike at 8 weeks of vaccination. The mean age was 52.1 years, and the standard deviation was ± 14.2; severe acute respiratory infection syndrome coronavirus 2 infection occurred in 20% of the population, of which 23.26% required hospitalization, 13.95% were under intensive care unit management, and four cases of mortality (9.3%) were reported. Of the total population, 52.5% had antibody titers higher than 0.8 IU/mL (median 0.77 IU/mL, interquartile range 0.4–131). Patients with severe acute respiratory infection syndrome coronavirus 2 infection had a median antibody titer of 0.4 IU/mL (interquartile range 0.4–3.45), and those without infection had a median antibody titer of 1.8 IU/mL (interquartile range 0.4–202) (
Conclusion:
Anti-severe acute respiratory infection syndrome coronavirus 2 antibody titers with a cutoff point less than 0.8 IU/mL are associated with increased risk of severe acute respiratory infection syndrome coronavirus 2 infection.
Introduction
In Colombia, the Omicron variant of severe acute respiratory infection syndrome coronavirus 2 (SARS-CoV-2) was documented for the first time in the first week of December 2021. By this time, the vast majority of the population, especially this cohort of patients, was vaccinated with a two-dose vaccination schedule under different platforms.
In Colombia, vaccines from three main platforms were used: CHADO X1-S/NCOV-19 from AstraZeneca, Ad26. S from Johnson & Johnson based on a viral vector platform; BNT162b2 from Pfizer-BioNTech, messenger ribonucleic acid (mRNA)-1273 from Moderna with a mRNA platform; and CoronaVac from Sinovac with an attenuated virus platform.
The immunogenicity of vaccines against viruses in solid organ transplant patients before the coronavirus pandemic had been mainly studied in the case of influenza where vaccination in renal transplant recipients is suboptimal, with antibodies developing at such low levels, reaching only 30% of those vaccinated1,2; an extra dose of vaccine is required to achieve protective antibody levels. 3
Solid organ transplant recipients infected with SARS-CoV-2 are at increased risk for severe disease manifestations, hospitalization, intensive care unit (ICU) admissions, and mortality. 4 Up to 84% of patients with symptomatic infection in this group require hospitalization compared to 45% of the general population.5,6 Not only do they have worse outcomes but the incidence of COVID-19 infection is two to five times higher than in the general population.6,7
In seven transplant centers in Bogota, Colombia, a 17% mortality of renal transplant recipients with SARS-CoV-2 infection prior to the start of vaccination schedules was found. 8
The different vaccination platforms generate a lower humoral response in solid organ transplant recipients in comparison with the general population1–4, which may be related to immunosuppression. 9
Under a mRNA platform in general, a response close to 50% has been found after two doses of vaccination 10 . Under platforms with viral vectors, specifically AstraZeneca, a response of 30%–45% has been found.11,12 A response of less than 30% has been found under platforms with inactivated viruses.13,14
Taking into account the above data, this study aims to evaluate the response of antibody titers after vaccination against SARS-CoV-2 measured in levels of anti-spike antibodies and clinical outcomes during the predominance of the Omicron variant in Colombia in renal transplant patients who have received the two-dose vaccination scheme in a Bogotá transplant center.
Methods
A retrospective cohort study, including renal transplant recipients from the Clinica Universitaria Colombia Colsanitas with a two-dose vaccination schedule against SARS-CoV-2 with Pfizer, AstraZeneca, Moderna, Jansen, and Sinovac vaccines on March 1, 2021 and December 1, 2021, was carried out with a follow-up period to evaluate outcomes until May 2022. The outcomes correspond to the titers of immunoglobulin G (IgG) antibodies against the receptor binding domain (RBD) of the SARS-CoV-2 spike in serum samples 8 weeks after receiving a vaccination schedule with at least two doses under the Elecsys platform (Roche, Indianapolis, IN, USA) with a cutoff point for positive response greater than 0.8 IU/mL and the relationship of titers with a composite clinical outcome given by infected patients, hospitalized patients requiring ICU admission, and death. The total number of renal transplant recipients who met the inclusion criteria was 215. Samples were collected from these recipients for measurement of antibody titers after receiving the second dose of vaccination, and all were included in the analysis. Patients living outside Bogota, those with limitations for central laboratory sample collection, and patients with active SARS-CoV-2 infection were excluded.
A descriptive analysis of the information was carried out, including the measures of central tendency and dispersion for quantitative variables; in the case of qualitative variables, frequency means, and percentage distribution, a descriptive analysis of the information was carried out, including the measures of central tendency and dispersion for quantitative variables; Qualitative variables, frequency means, and percentage distribution were calculated. Subsequently, a bivariate analysis was carried out using mean or median difference tests, such as Student’s
The research protocol was approved by the Clinica Universitaria Colombia Colsanitas on November 19, 2021, with approval number CEIFUS2259-21, and it complies with the international research standards. The authors considered that obtaining informed consent was not necessary for the type of study conducted, and the non-obtaining of consent was approved by the institutional ethics committee.
Results
In total, 215 renal transplant recipients received two doses of vaccination for SARS-CoV-2 with measurement of antibody titers 8 weeks after vaccination. The demographic characteristics of this population showed a mean age of 52.1 years with a standard deviation (SD) of ± 14.2; the predominant sex was male (59.1%,
Characteristics of the study population.

SD: standard deviation; Pred: prednisolone; Tac: tacrolimus; Bela: belatacept; MPA: mycophenolic acid analogs; mTOR: mTOR inhibitors.
The population distribution according to the time of transplantation was eight patients (3.72%) transplanted in less than 6 months and 207 (96.28%) patients transplanted more than 6 months ago, with no statistically significant differences in relation to the antibody titers achieved.
At the time of data collection, the most frequent immunosuppression schemes were mycophenolate (MPA) (
In each of the subgroups of patients who received immunosuppression schemes, it was found that patients who received belatacept and prednisolone (
Of the study population, 50 patients received renal transplantation in the last 3 years (23.25%), and at the time of data collection, the most frequent immunosuppression regimens were MPA (
Morbidity and mortality
Of the 43 patients who evidenced SARS-CoV-2 infection, 10 patients (23.26%) required hospitalization, 6 required ICU management (13.95%), and 4 cases of mortality (9.3%) (Table 2) were recorded. All patients requiring hospitalization were over 40 years old and 90% were men. The most frequent vaccination schedules in patients who presented the composite outcome were Pfizer: six patients; Sinovac: two patients; and AstraZeneca: two patients.
Clinical outcomes in patients with SARS-CoV-2 infection.

Antibody titers
Of the 215 patients included in the study, 52.5% (
Patients with SARS-CoV-2 infection had a median antibody titer of 0.4 IU/mL (IQR = 0.4–3.45), and those without SARS-COV-2 infection had a median antibody titer of 1.8 IU/mL (IQR = 0.4–202), with this finding being statistically significant (

Ratio of antibody titers in SARS-CoV-2-infected and uninfected patients.
Antibody titers in SARS-CoV-2-infected and uninfected patients.

Mann–Whitney median difference test.
From the antibody titers of patients who presented the clinical outcome of hospitalization, ICU requirement, and death, a median antibody titer of 0.4 IU/mL (IQR = 0) was found, and in those who did not present this outcome, the median antibody titer was 0.4 IU/mL (IQR = 0.4–10.5) without statistical differences (
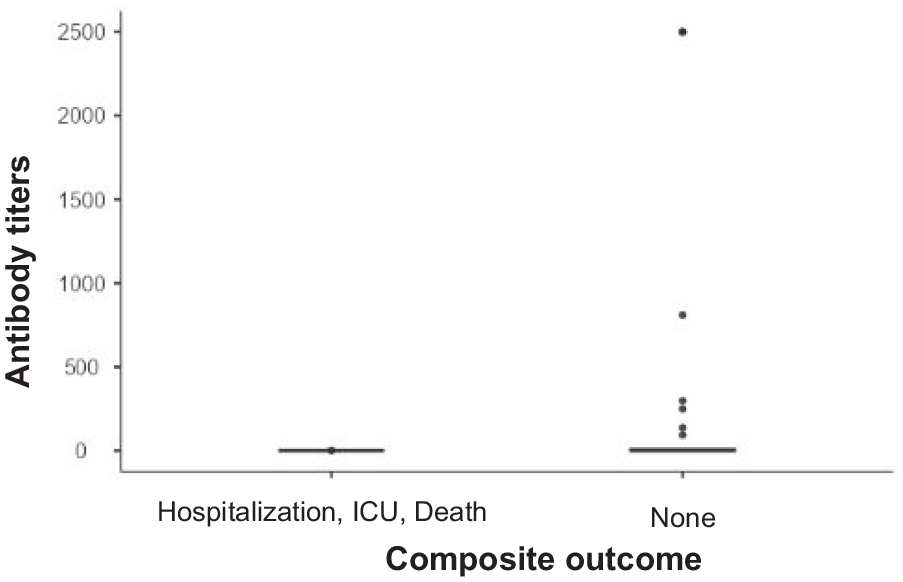
Antibody titers in relation to composite clinical outcomes.
Taking into consideration only the outcome of death, a median antibody titer of 0.4 IU/mL without statistical differences with respect to the group of non-deceased with SARS-CoV-2 infection was found (

Antibody titers in relation to mortality outcome.
Discussion
This study evaluated the clinical behavior associated with the humoral response 8 weeks after the application of two doses of vaccination against SARS-CoV-2 during the predominance of the Omicron variant in renal transplant patients using three different platforms: mRNA, inactivated virus, or vectors, thus carrying out anti-spike antibody titer measurement by Elecsys test (Roche) with a reference value of 0.8 IU/mL.
Lower effectiveness of vaccination against the Omicron variant compared to the Delta variant has been demonstrated in the general population 15 ; recent analyses suggest immunologic evasion of this variant 16 as well as the need for a third dose of vaccination to achieve neutralizing antibody titers associated with protection.17,18 In solid organ transplant recipients, different studies have shown a lower probability of humoral immune response to vaccination compared to the general population under different vaccination platforms such as BNT 162b2, mRNA 1273, CoronaVac, and ChAdOx1mRNA, although finding a better probability of response with mRNA vaccines compared to attenuated viruses or viral vectors.5–7,9,11–14,18–25
In this experience, it was found that 52.5% of patients included in the study had antibody titers higher than the reference value (0.8 IU/mL, Elecsys, Roche); a statistically significant difference between the median antibody titers of patients infected and not infected with SARS-CoV-2 (
From the immunosuppression regimens used by the patients in the study population, it was found that the use of therapies that included belatacept and prednisolone was related to antibody titers <0.8 IU/mL when compared to other regimens that did not include these agents; similarly, patients who received regimens that included mTOR presented mostly titers ⩾0.8 IU/mL with a statistically significant value. Findings were consistent with those found in previous studies in a similar population. 8
Previous infection with a different variant of Omicron was associated with a reduction in the risk of infection of approximately 50%, finding greater efficacy with hybrid immunity from previous infection and recent booster vaccination (up to 80%), a finding described in a national study in Qatar 30 ; unvaccinated people with previous infection may have infection-induced immunity, which represents a bias when evaluating the effectiveness of vaccination, even presenting higher levels of protection than those vaccinated without a history of exposure.31–33 In this study, previous infection with other variants was not taken into account for the analysis of adverse clinical outcomes, and it remains to be established whether protection against the Omicron variant is provided by immunity associated with previous infection, vaccination, or a combination of both.
Hospitalization rates in recipients of solid organ transplants during the predominance of the Omicron variant have been reported in up to 26% of confirmed cases 34 ; an analysis carried out in seven transplant centers in the city of Bogota, Colombia, found a case fatality rate of 17% and a hospitalization rate of 49.7% in renal transplant recipients with SARS-CoV-2 infection in the pre-vaccination era, 8 which is in contrast to a case fatality rate of 9.3% and a hospitalization rate of 23.26% found in this study, suggesting that vaccination is associated with a lower risk of hospitalization and death in solid organ transplant recipients as has been reported in heart transplant recipients with SARS-CoV-2 infection. 35 This protective effect of vaccination independent of antibody titers has been demonstrated in the transplant population in other types of vaccination strategies such as influenza, suggesting that not only humoral but also cellular immunity is involved in the protective effect of vaccination in this patient population. 36
The data from this study suggest that a substantial proportion of renal transplant recipients probably remains at risk of SARS-CoV-2 infection after two doses of vaccination regardless of the type of biologic utilized; therefore, it seems paramount to confirm in appropriately designed studies the protective effect of vaccination in solid organ transplant recipients by measuring IgG antibodies against the RBD of the SARS-CoV-2 spike in order to stratify the risk of complications associated with SARS-CoV-2 infection in patients with a poor response to vaccination such as those on belatacept26–29 so as to implement the use of early therapeutic strategies to reduce the risk of hospitalization and death in this population with low immunologic response to current vaccination platforms. 34
Conclusion
We evaluated the humoral response in renal transplant patients 8 weeks after receiving two doses of SARS-CoV-2 vaccination using three different platforms. The results showed that 52.5% of patients had antibody titers above baseline, but it was not possible to establish a cutoff point to discriminate patients at risk of hospitalization or death. The use of belatacept and prednisolone was associated with lower antibody titers, while mTOR was associated with higher antibody titers.
This study also found that prior infection with a different Omicron variant was associated with a reduced risk of infection. Vaccination was associated with a lower risk of hospitalization and death in solid organ transplant recipients, independent of antibody titers.
This research suggests that a substantial proportion of renal transplant recipients remains at risk for SARS-CoV-2 infection after two doses of vaccination, and further studies are needed to confirm the protective effect of two doses of vaccination or to explore booster dose protocols and stratify the risk of complications associated with SARS-CoV-2 infection in patients with a poor response to vaccination. Nevertheless, vaccination remains the safest and best tool for protection against infection, hospitalization, and death in solid organ transplant recipients with SARS-CoV-2 infection.
Limitations
This is a retrospective study without knowledge of the baseline antibody titers of the population, without comparative analysis of hospitalization and mortality against a control group of unvaccinated patients, and without viral genetic analysis to confirm infection by the Omicron variant. The sample size was not calculated since the available population that met the selection criteria for conducting the study was taken.
Footnotes
Acknowledgements
The authors are acknowledged for their efforts in conducting this study.
Author contributions
Rodolfo Torres, Camilo Montero, Maricely Reina, and Jonth Flechas researched the literature and conceived the study. Sebastián Moreno and Camila Granados participated in the development of the protocol, obtaining ethical approval, Camilo Montero carried out the recruitment of patients, and David Andrade participated in the data analysis. Maricely Reina, Camilo Montero, and Rodolfo Torres wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
The research protocol was approved by the Clínica Universitaria Colombia Colsanitas on November 19, 2021, with approval number CEIFUS2259-21, and it complies with the international research standards.
Informed consent
The authors considered that obtaining informed consent was not necessary for the type of study conducted, and the non-obtaining of consent was approved by the institutional ethics committee.
