Abstract
Presentation, management, and outcomes of COVID-19 infections among younger patients is an area of medicine with deficits in research, likely due to the lower incidence of severe COVID-19 disease among the younger population. Management can be challenging, and clinicians often guide their decision-making based on the ever-changing protocols that are tailored mostly to the elderly population. Even more underrepresented in COVID-19 research are patients with chromosomal abnormalities and trisomy syndromes, as they appear less frequently, but have risk of increased morbidity and mortality due to underlying medical conditions. We describe a case of severe COVID-19 infection in a young patient with mosaic trisomy 13 and pre-existing polycystic kidney disease, who developed severe acute hypoxic respiratory failure and acute chronic kidney injury. The patient was provided maximal pharmacological support and her clinical course helps to shape the understanding of COVID-19 infections in the setting of chromosomal abnormalities and complex medical history.
Introduction
The coronavirus disease 2019 (COVID-19) pandemic caused by SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2) continues to remain the focus of clinical and research efforts by health care professionals globally. A large proportion of COVID-19-related deaths have involved the elderly, with more than 70% of deaths in the United States involving adults over the age of 70 years. 1 However, the pediatric and young adult population has not been completely immune to SARS-CoV-2 infection, long-term sequelae, and mortality. Children and young adults with COVID-19 generally experience a milder symptom profile compared to high-risk elderly patients, with fever and cough being the most common presenting symptoms.2,3 However, even younger patients can have severe and life-threatening COVID-19 in the presence of pre-existing medical conditions.2,3 There are several congenital conditions and chromosomal abnormalities seen predominantly in pediatric and young adult populations. Coupling the rarity of these congenital diseases with the significantly lower incidence of severe SARS-CoV-2 in younger cohorts, 4 it is not surprising that there is a paucity of literature documenting COVID-19 in these populations. As COVID-19 moves toward endemic status globally, and new variants continue to evolve, it becomes paramount to understand and document the variable presentation and clinical course of COVID-19 in these rare populations.
Review of literature shows emerging evidence that children and adults with trisomy 21 (Down syndrome) are at higher risk for severe COVID-19 with life-threatening complications.5,6 However, there are no published data on other trisomy syndromes. Trisomy 13, also known as Patau syndrome, has a median life expectancy of 7–10 days, and 90% of children die before the age of 1 year. However, the mosaic form of trisomy 13 can have a variable clinical presentation, and be associated with relatively longer survival. 7 We present a case of a young patient with complex medical history of mosaic trisomy 13 and polycystic kidney disease (PKD), who developed severe COVID-19 and subsequently died secondary to complications.
Case presentation
A 20-year-old female with a complex medical history of mosaic trisomy 13, PKD, asthma, seizure disorder, gastric-tube dependence, and osteoporosis presented to our tertiary medical center emergency department with respiratory distress, fever, vomiting, and diarrhea in setting of SARS-CoV-2 exposure through sibling. Her highest pre-hospitalization temperature was 39.4°C, and her mother reported a home pulse oximetry reading of 86% prompting presentation on the fifth day of symptoms. The patient’s older brother who lived in the same household had been feeling fatigued and febrile for last 7 days, and tested positive for SARS-CoV-2 on the same day as the patient’s presentation to our hospital. Since the patient was restricted to home due to her chronic medical condition, her mother was concerned that the patient may have contracted COVID-19 from her brother. At baseline, this young woman was non-verbal and wheelchair bound, and seizures were controlled with oxcarbazepine (600 mg twice daily) and levetiracetam (600 mg twice daily). Patient also had chronic urinary incontinence at baseline requiring urinary tract infection (UTI) prophylaxis with weekly alternating nitrofurantoin (50 mg once daily) and trimethoprim/sulfamethoxazole (80/400 mg once daily).
Upon presentation, she was tachycardic (heart rate 107 beats/min) and hypoxic (oxygen saturation 86%), with respiratory rate of 24 breaths/minute, blood pressure of 143/84 mmHg, and temperature 36.4°C. She was acutely distressed, with decreased breath sounds and crackles bilaterally. Her oxygen saturation improved with initiation of high flow nasal canula and she received a dose of piperacillin–tazobactam (1 g piperacillin and 0.125 g tazobactam) for empiric coverage due to concerns for potential aspiration. The emergency medicine team presumably suspected aspiration based on her presentation with respiratory distress and fever in setting of vomiting in addition to chronic disabilities that could potentially compromise airway protection. Initial labs reflected leukocytosis (white cell count 15.4 K/µL), abnormal urinalysis concerning for UTI, and elevated inflammatory markers (ferritin 950 ng/mL, C-reactive protein 33.90 mg/dL, procalcitonin 6.59 ng/mL). In addition, she demonstrated metabolic derangements including mild hyponatremia (130 mmol/L), hypochloremia (95 mmol/L), and elevated blood urea nitrogen/creatinine ratio (35.0). Chest x-ray (Figure 1) demonstrated diffuse ground glass opacity appearance, suggestive of acute respiratory distress syndrome (ARDS). Her nasopharyngeal SARS-CoV-2 PCR (polymerase chain reaction) test resulted positive, and she was admitted to the intensive care unit for management of acute hypoxic respiratory failure secondary to COVID-19. Piperacillin–tazobactam was not continued during inpatient stay as positive SARS-CoV-2 test sufficiently explained her clinical presentation. During her admission, electrolyte levels were monitored daily due to her PKD (Figure 2 and Table 1).
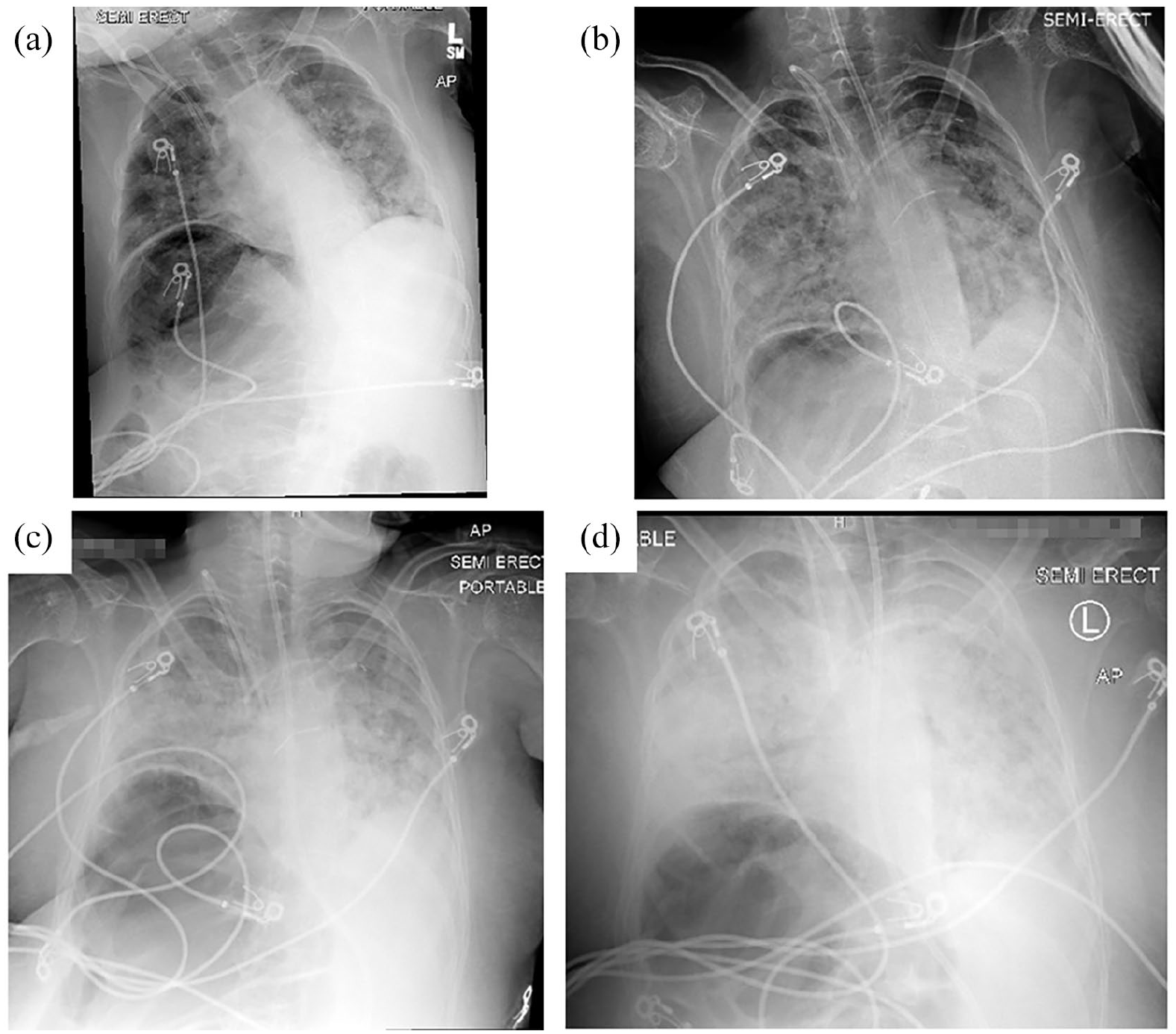
Chest radiographs showing progression of COVID-19-related lung disease: chest x-ray on (a) presentation, (b) inpatient day 3, (c) day 6, and (d) inpatient day 11.
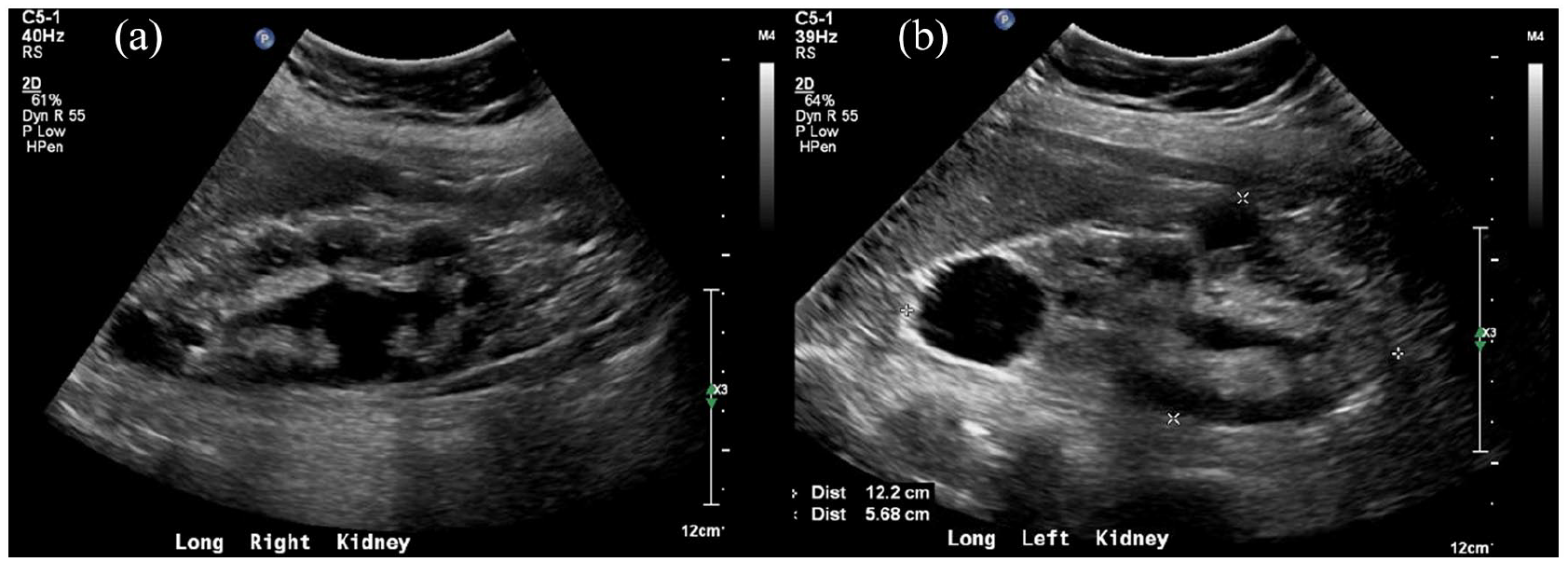
Long axis ultrasound images of (a) Right, and (b) Left sided polycystic kidneys.
Notable laboratory markers and therapies during hospital course.

E. coli: Escherichia coli; UTI: urinary tract infection; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; IV: intravenous.
N/A indicates not collected.
Based on COVID-19 protocols at the time (August 2020), she received remdesivir 100 mg from days 1 to 5, and convalescent plasma on day 2 of hospitalization. Dexamethasone 10 mg was also administered from days 1 to 5 and again from days 8 to 12. For respiratory support, she was initiated on high-flow nasal cannula at 40 L/min with FiO2 80%. This was escalated to non-invasive mechanical ventilation with bi-level positive airway pressure (BiPAP) on day 2 of admission due to worsening hypoxic respiratory failure (O2 saturations 87%), worsening chest x-ray findings, and persistent tachycardia. Alternating prone positioning was also initiated on day 2, but discontinued on day 5 of hospitalization due to patient discomfort, and perceived lack of physiological improvement.
Dexmedetomidine infusion was initiated to optimize patient comfort and improve BiPAP synchrony. She received enoxaparin 20 mg twice daily starting on day 1, and was increased to 50 mg twice daily per COVID-19 protocols when her
Despite above treatments and supportive care, the patient’s clinical status and labs continued to worsen (Table 1). By day 9, she had developed worsening acute kidney injury (AKI), presumed secondary to kidney damage from SARS-CoV-2 infection in setting of pre-existing PKD. In setting of progressively worsening clinical status, on day 10, her parents elected to redirect care toward comfort measures only. By day 12, she progressed to severe oliguria unresponsive to diuretics or fluid resuscitation; parents elected not to proceed with hemodialysis when offered the option. Her respiratory status continued to worsen with progressive hypercapnia (pCO2 86 mmHg) and worsening lung infiltrates on chest x-ray (Figure 1) until her death on day 13 of hospitalization.
Discussion
Trisomy 13 is found in 1 in 10,000 to 20,000 births and is the third most common autosomal trisomy in live births. 7 These children may be born with microcephaly, midline facial defects, and characteristic facial features such as small malformed ears, anophthalmia/microphthalmia, and micrognathia. Central nervous system abnormalities such as alobar holoprosencephaly, and congenital heart defects such as atrio-ventricular septal defects and tetralogy of Fallot may be present. In addition, lungs, liver, kidneys, and gastrointestinal tract may also be involved. Common limb abnormalities such as polydactyly, talipes equinovarus, or rocker-bottom feet are commonly present. 8 While there is variability in life expectancy, majority of affected infants die within the first weeks of life. However, mosaicism occurs in 5% of trisomy 13 cases, thus above-mentioned clinical features can vary from mild to profound.9–11 This report describes SARS-CoV-2 infection in a 20-year-old patient with severe clinical features of mosaic trisomy 13, and the rarity of this condition particularly at this age, makes this is a valuable addition to the fund of COVID-19 literature.
Children and young adults have a much lower COVID-19-related death rate than elderly counterparts, 4 likely due to their healthier status and fewer underlying medical conditions. Our patient, however, does not fall into this category, as she had many co-morbid factors that may have resulted in worsened disease process. While there is little information on the interaction between COVID-19 and trisomy 13 patients, we can extrapolate from children with trisomy 21 who have increased mortality rates from COVID-19 infections (as do other children with intellectual/developmental disabilities). 5 Similar to trisomy 21, trisomy 13 can result in abnormalities in upper airway anatomy and hypotonia, both of which can result in worsening of respiratory infections. 8 In addition, cardiopulmonary status of trisomy 13 patients is an important consideration in that patients may be particularly vulnerable to pathology as a result of structural malformations. 8
Finally, dysregulation of an individual’s immune system could play a role in developing severe infections; it has been shown that patients with trisomy 21 can have variations in immune functioning (lymphopenia, impaired chemotaxis, etc.) that may contribute to the morbidity of respiratory infections. 6 While these data are specific to trisomy 21, it is possible that similar challenges are contributing to disease process in trisomy 13. Notably, our patient had persistently low levels of total IgG diagnosed at age 5 years, which was managed with monthly intravenous immunoglobulin (IVIG) infusions when she was younger. The pediatric immunology team had briefly trialed her off IVIG at age 10 years, but subsequently restarted due to persistently low levels. At the time of presentation, our patient was receiving weekly subcutaneous immunoglobulin. Interestingly, the rapidly expanding COVID-19 literature now includes several reports on successful use of IVIG as an adjuvant therapy, with favorable outcomes in severe COVID-19 pneumonia. 12 This should perhaps also be considered as an adjuvant agent in COVID-19 patients with chromosomal abnormalities and trisomy syndromes, particularly in cases with documented immunoglobulin deficiency.
Respiratory support management was a particular challenge in the care of this patient, especially as her disease progressed. Because of persistently low oxygen saturation levels and worsening respiratory distress, patient required support via BiPAP, but only helped transiently. Trial of non-invasive mechanical ventilation as initial support for COVID-19-associated respiratory failure is standard therapy at this time. However, it should be noted that our patient presented during early stages of the pandemic, at a time when experts were instead erring on the side of early intubation. 13 In the case of our patient, intubation was considered; however, parental guardians decided against full code measures including invasive mechanical ventilation via intubation. Our patient had a history of chronic, well-controlled asthma; however, a review of available literature does not point to a clear link between asthma and trisomy 13. It can be argued that the underlying asthma contributed to her disease severity, but we would like to emphasize that she did not manifest any signs of obstructive lung disease or status asthmaticus. Instead, her pulmonary mechanics were consistent with poor compliance secondary to parenchymal disease and ARDS.
Anticoagulation is an important consideration in the management of patients with COVID-19. It has been shown that
At the time of our patient’s hospitalization, there had been two randomized clinical trials comparing 10-day treatment of remdesivir, an antiviral agent, versus placebo; one of these studies showed reduction of recovery time in patients receiving the antiviral. 16 Similarly, a preliminary report at the time supported the use of dexamethasone in hospitalized patients receiving respiratory support, as it was shown to reduce 28-day mortality. 17 This process is hypothesized to be related to reduction of inflammatory-mediated lung injury, thus improved respiratory status. 17 Both agents have subsequently showed benefit in severe COVID-19 pneumonia in setting of end-stage renal disease, similar to our patient. 18 Finally, the use of convalescent plasma has been studied in the context of previous viral outbreaks, including Ebola virus, Influenza A H1N1, and Middle East respiratory syndrome coronavirus. 19 Because of this information, convalescent plasma was identified relatively early in the COVID-19 pandemic as a possible treatment option in severe cases due to a potential protective effect from antibodies of recovered patients.
Another major complication of this patient’s disease process was the rapid decline in kidney function. It is believed that the SARS-CoV-2 infection directly resulted in AKI. As found in other studies, AKI is a common result of this particular infection and is significantly more likely to occur in COVID-19 positive patients. 20 It is hypothesized that a variety of processes may contribute to renal decline, including systemic collapse, pro-thrombotic states resulting in micro-emboli, and cytokine storms that can occur with SARS-CoV-2. Our patient also had PKD, and although her renal function was within normal limits prior to admission, it is recognized that polycystic kidneys have underlying renal dysfunction and an increased susceptibility to AKI induced by ischemic-reperfusion injury. 21 Therefore, it would be reasonable to assume that her PKD could have exacerbated the COVID-19-associated AKI. Since 50%–60% of patients with trisomy 13 can have structural renal abnormalities, and more than one-third of these are cystic kidney diseases, 22 physicians caring for COVID-19 in trisomy 13 population should be mindful of the theoretically higher risk of AKI.
Conclusion
Although vaccination efforts are spreading globally, new variants of SARS-CoV-2 continue to emerge, and patient populations with predisposing medical conditions will continue to be at high risk for severe disease and mortality. Younger individuals with trisomy syndromes certainly belong in this group, and we present one of the first reported cases of SARS-CoV-2 in a patient with mosaic trisomy 13, shedding light on the medical fragility of these patients and their susceptibility to severe COVID-19.
Footnotes
Acknowledgements
We would like to thank the nursing staff within the pediatric ICU who contributed to the management, and provided compassionate end-of-life care for this young patient.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing charges related to the publication of this article were supported by The University of Kansas (KU) One University Open Access Author Fund sponsored jointly by the KU Provost, KU Vice Chancellor for Research & Graduate Studies, and KUMC Vice Chancellor for Research and managed jointly by the Libraries at the Medical Center and KU - Lawrence.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
