Abstract
Ectopic pancreas refers to pancreatic tissue that lacks vascular or anatomical connection with the main body of the pancreas. Superior mesenteric artery syndrome involves the compression of the third part of the duodenum between the aorta and the superior mesenteric artery. We present a case of a 12-year-old female with a 4-year history of recurrent episodes of severe epigastric pain, improved by sitting and leaning forward, and occurred monthly for nearly 48 h. Radiological findings confirmed the diagnosis of superior mesenteric artery syndrome. However, further investigation revealed the presence of ectopic pancreas. Surgical intervention successfully treated both conditions. The patient presented with severe abdominal pain, and although there was a diagnosis of superior mesenteric artery syndrome, the pain was not entirely attributable to this condition, as there was no correlation with food intake, and the degree of obstruction was mild. Further investigations revealed the presence of ectopic pancreas, which provided an explanation for the severity of the pain. This case highlights the need for further research on the potential association between superior mesenteric artery syndrome and ectopic pancreas.
Introduction
The ectopic pancreas (EP) is a pancreatic tissue without vascular or anatomic connection with the essential body of the pancreas. According to autopsy investigations, the incidence ranges from 0.5 to 13.7%. 1
The stomach, duodenum, and proximal jejunum are the parts of the upper gastrointestinal (UGI) tract where the EP most frequently develops. 2 The condition is typically asymptomatic, and most instances are discovered by accident. Rarely, it may occur suddenly with thoracic discomfort, vomiting, nausea, and abdominal pain. Endoscopic ultrasonography (EUS) is the most effective diagnostic method for submucosal gastric lesions. Surgery can be a consideration for possible treatment in symptomatic patients and definitive diagnosis. 3
Superior mesenteric artery (SMA) syndrome is the compression of the third part of the duodenum between the aorta and the SMA. It is a rare situation with an incidence between 0.13 and 0.3% and a preference for young, skinny females. 4
Potential causes of SMA syndrome may include fast weight loss, certain metabolic conditions that reduce the amount of mesenteric and retroperitoneal adipose tissue, trauma, a high degree of lumbar lordosis, neoplastic masses close to the SMA radix, a dissecting aortic aneurysm, and certain types of surgeries. 5
The most common symptoms are abdominal pain, nausea, and vomiting. Treatment may be conservative or surgical, depending on the severity of the clinical presentation. 6
With the similar manifestations of these two conditions, we face a challenge in diagnosing and treating them both. This is a case report of a 12-year-old child who has both situations simultaneously.
Case presentation
A 12-year-old female presented to our hospital with a 4-year history of recurrent episodes of severe epigastric pain associated with nausea and mild fever but without any history of vomiting or changes in bowel habits. The pain was exacerbated by movement, relieved by sitting and leaning forward, radiated to the back, and occurred almost monthly with an onset of 48 h.
The pain was slightly responsive to antispasmodics. Physical examination revealed abdominal tenderness.
The patient’s medical history revealed that her mother had a history of migraines that were under preventive control, but there was no history of blood diseases in the family. The patient did not report any symptoms of anemia or a history of blood transfusion.
During these 4 years, outside our hospital, the patient had previously been misdiagnosed with peptic ulcer and treated for helicobacter pylori infection, as well as food allergy, with no improvement.
The patient’s recurrent episodes of abdominal pain required hospital admission, and management with strong analgesics was necessary. Abdominal migraine was ruled out as a potential explanation, as the absence of headache in the child was inconsistent with this condition.
To investigate the cause of the patient’s symptoms, laboratory tests were performed, and imaging studies were conducted. Serum amylase and lipase levels were within normal limits (pancreatitis was excluded). Abdominal multislice computed tomography (MSCT) with injection—which was done before she came to the hospital—revealed an acute angle between the superior mesenteric artery and the aorta with a slightly marked compression on the third segment of the duodenum and a gastric dilatation (Figure 1). The radiological findings corresponded with the SMA syndrome. Additional investigations were pursued due to the persistence of severe pain despite the mild obstruction and lack of association between pain and food intake. An endoscopic ultrasound (EUS) noted a submucosal polypoid swelling in the prepyloric antral region, which appeared to be a heterogeneous formation with a micro duct (Figure 2 and Figure 3). The patient underwent an exploratory laparotomy that displayed an expansion in the stomach and duodenum, which was partially compressed by the superior mesenteric artery. A small mass of 13 × 7 × 5 mm was located very close to the pylorus of the stomach. The mass was completely resected, and a diamond-shaped duodenal–jejunal anastomosis was performed at 20 cm from the Traits ligament. Histopathological examination of the specimen revealed pancreatic acinar cells and ducts (Figure 4). The patient’s recovery after surgery was uneventful, with a regression of GI symptoms observed 6 months after the procedure.

UGI endoscopy showing submucosal dimpled polyp in the prepyloric antrum (red arrow).

UGI endosonography showing a mass lesion measuring 7.52 × 3.96 mm in the pylorus of stomach (red arrow).

Sagittal CT shows a narrowing of the distance between the superior mesenteric (red arrow) artery and the aorta (blue arrow) measuring 6 mm and an acute angle between the two arteries measuring less than 20° with a slight impression on the third segment of the duodenum (purple arrow) and gastric dilatation, corresponding to the mesenteric artery syndrome.
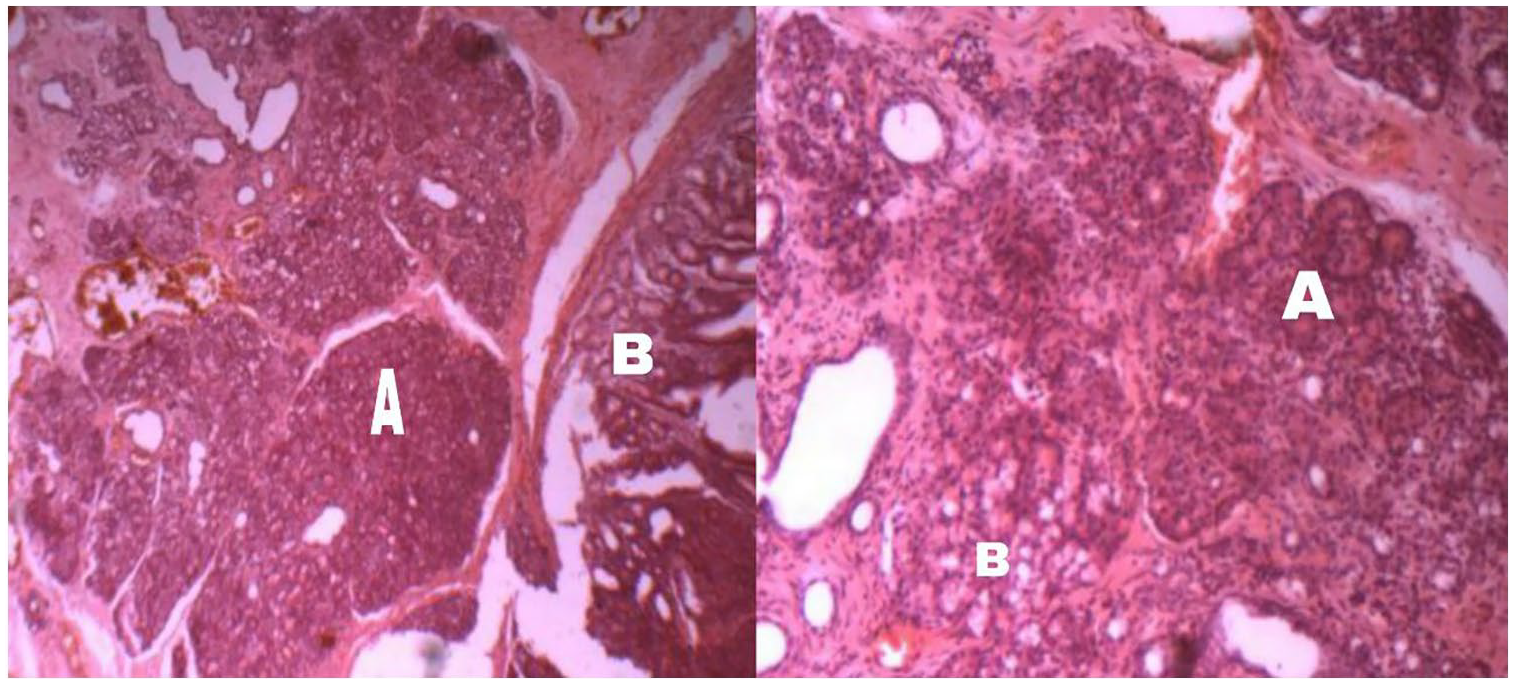
Sections of gastric mucosa show mild chronic inflammatory cell infiltrates in lamina propria “Brunner glands” (B) along with pancreatic tissue (A).
Discussion and literature review
The EP is a congenital abnormality in which the pancreas’s tissue is anatomically distinct from the main gland without vascular or ductal continuity. 7 In autopsy studies, the prevalence of EP in the general population ranges from 0.55 to 13.7%, while in recent surgical studies, the prevalence ranges from 0.18 to 5.3%. 8 It is most common between the ages of 30 and 50, with male predominance. 9 Malignant transformation of EP is rare; it happens in 0.7–1.8% of patients.1,7
The etiology remains unclear, although several theories have been put forth. 10 Studies have documented instances of ectopic pancreatic tissue in children, with the most common locations being the stomach or Meckel diverticulum. However, the tissue has also been found in other areas of the GI tract, such as the duodenum, jejunum, ileum, and esophagus, as well as in the biliary tree, gut mesentery, liver, spleen, appendix, omentum, and umbilicus. 11 In cases of EP, the submucosa layer is the most common location (54–75% of cases), with some instances where it extends beyond the submucosa layer to cross the muscularis propria (23% of cases). The muscular layer is affected in only 8% of cases, while the serous layer is involved in 11–13%. 1 In our case, EP was located in the submucosa in the prepyloric antral region of the stomach.
Most cases with EP were asymptomatic, whereas the symptomatic cases manifested with abdominal pain, which is the most common symptom. In some cases, there was GI bleeding, dyspepsia, pancreatitis, and gastric ulceration.7,8
The majority of cases are discovered by chance during endoscopy, but since other lesions might seem similar to EP by esophagogastroduodenoscopy, EUS with fine-needle aspiration might be a safe technique to diagnose EP and can distinguish it from the other lesions. 9 In the present case, the EP was not visualized on the abdominal MSCT, which was performed externally. This was likely due to the small size of the EP and the average quality of the MSCT device. At the time of the scan, there was no indication to review the CT image as the diagnosis of SMA syndrome had already been made.
However, an EUS revealed a submucosal polypoid swelling in the prepyloric antral region, which ultimately led to the diagnosis of the EP. The final diagnosis was confirmed by histological examination after the mass resection.
The primary differential diagnosis of EP includes GI stromal tumors, gastric carcinoids, lymphomas, GI autonomic nerve tumors, and gastric carcinomas. 2
According to Heinrich’s 1909 classification and the following the Gasper-Fuente update in 1973, EP can be classified into four types7,12,13:
Type 1: EP consists of all parts of the normal pancreas: acini, ducts, and islet cells.
Type 2: EP consists of only pancreatic ducts.
Type 3: EP consists of only acinar tissue (exocrine pancreas).
Type 4: EP consists of only islet cells (endocrine pancreas).
Our case findings are consistent with type 1 (acinar cells and ducts are in the specimen).
Ectopic pancreatic tissue is vulnerable to the same pathologic diseases that can afflict the orthotopic pancreas, such as pancreatitis, pseudocyst development, and both benign and malignant neoplasms.
GI hemorrhage, bowel obstruction, and intussusception are additional complications specific to EP. 7
As some authors have suggested, we do not advise resection for children with incidentally detected asymptomatic lesions and typical endoscopic characteristics. 11 For symptomatic patients, localized surgical excision is appropriate and the preferred treatment option in the absence of malignant transformation. 9 Surgery was performed in this case due to the patient’s recurrent episodes of severe abdominal pain.
Rokitansky was the first to identify the SMA syndrome, describing it in 1842. The incidence of SMA syndrome ranges between 0.20 and 0.7%.
When the third segment of the duodenum is compressed between the SMA and aorta, it becomes symptomatic.14,15
Although it can happen at any age, the illness is more likely to affect women and young adults (18–35 years). This age and sex distribution may reflect the condition’s underlying factor, namely eating disorders. 16 Normal aorto-mesenteric angles range between 25 to 60° and mean aorto-mesenteric distances vary from 10 to 28 mm. However, SMA syndrome may affect patients with an angle of less than 25 and an aorto-mesenteric distance of less than 8–10 mm. 18
Patients have been characterized by this syndrome falling into one of two categories: those who experience it after surgery or compression and those who do so in conjunction with extreme weight loss. The first category comprises patients who have recently undergone corrective spinal surgery, which has historically been the most often mentioned cause. The second group comprises catabolic problems such as burns, wasting diseases like malabsorption, cerebral palsy, cancer, AIDS, and eating disorders like anorexia nervosa. 16 The child, in this case, was underweight, likely due to refraining from eating during episodes of abdominal pain. Given the absence of other obvious causes for the weight loss, it is possible that the underlying condition responsible for the pain may have contributed to the weight loss that caused SMA.
Depending on the cause of the SMAs or the degree of duodenal compression, patients with SMAs typically experience acute or persistent abdominal pain. If the obstruction is severe, patients will complain of vomiting, vomiting bile, and losing body mass. Whereas, patients with mild obstruction complain of postprandial epigastric pain, esophageal reflux, and an early feeling of fullness after meals.5,17 In the present case, MSCT revealed mild obstruction, and the patient did not report vomiting. Additionally, the patient’s epigastric pain was not triggered by eating.
The compression is confirmed by duodenography and CT; both were considered the gold standard diagnostic investigations. While duodenography revealed diagnostic findings during the symptomatic period, CT revealed compression and a decreased angle during the symptomatic and asymptomatic periods. 18
The SMAs are frequently treated with conservative techniques, such as maintaining a healthy weight and situating the patient (on the abdomen or left side of the body with knees bent to the chest) could be a helpful aspect of treatment if the symptoms suddenly worsen. When symptoms do not get better with conservative treatment, surgery is recommended. 5 The patient’s pain was observed to decrease when sitting and leaning forward, but it did not completely subside except with the use of analgesics. The severity of the pain made conservative therapy an inappropriate option.
The following surgical procedures can treat SMA syndrome: gastrojejunostomy, duodenojejunostomy, Treitz ligament lysis, or the strong surgical technique. 5 In the present case, due to the presence of two potential causes of the patient’s abdominal pain, the surgical solution was chosen. This approach allowed for the removal of both possible sources of pain through a single surgical procedure, which may have minimized the patient’s discomfort and recovery time.
We conducted a literature search of the PubMed database using filters for English language case reports published in the last 10 years. The search used the keywords “Superior,” “mesenteric,” “artery,” “syndrome,” and “children.” We excluded obviously irrelevant articles and those that were inaccessible and ultimately reviewed 12 articles. We summarized the characteristics of these articles and compared them with the overall characteristics of superior mesenteric artery syndrome in (Table 1).
Describes the Pt age (Ys)/sex, symptoms, diagnostic tests, tumor size, treatment, follow-up, and recurrence for reviewed articles.

Pt: patient; Ys: years; CT: computed tomography; MRA: magnetic resonance angiography; UGI: upper gastrointestinal; GI: gastrointestinal.
We found that the age at diagnosis of superior mesenteric artery syndrome ranged from 6 to 12 years.19–30
Various symptoms were recorded in the literature, such as abdominal pain and early satiety, but the most common symptom, which was detected in all these cases, was vomiting.19–30
In our case, the patient didn’t have vomiting as the duodenal obstruction was mild. Thus, the absence of vomiting does not negate the disease.
Diagnostic tests used included CT scan, fluoroscopy, upper gastrointestinal endoscopy, abdomen magnetic resonance angiography, upper gastrointestinal (UGI) contrast study, and abdominal X-ray. The diagnostic method used in almost all cases, including ours, and each time showed and confirmed the SMA presence was CT.19–30
Despite the success of conservative treatment in a few cases, the recurrence made surgery the best option in most cases. This type of operation is almost always accompanied by improvement, as we noticed.19–30
Distinguished note that SMA was associated in most cases with another pathological condition associated with weight loss, such as celiac disease, which was observed in two of the cases.20,30
We conducted a literature search of the PubMed database using filters for English language case reports published in the last 10 years. The search used the keywords “ectopic,” “pancreas,” and “children.” We excluded obviously irrelevant articles and those that were inaccessible and ultimately reviewed eight articles. We summarized the characteristics of these articles and compared them with the overall characteristics of EP in (Table 2).
Describes the Pt age (Ys)/sex, symptoms, diagnostic tests, size, location, treatment, follow-up, and recurrence for reviewed articles.
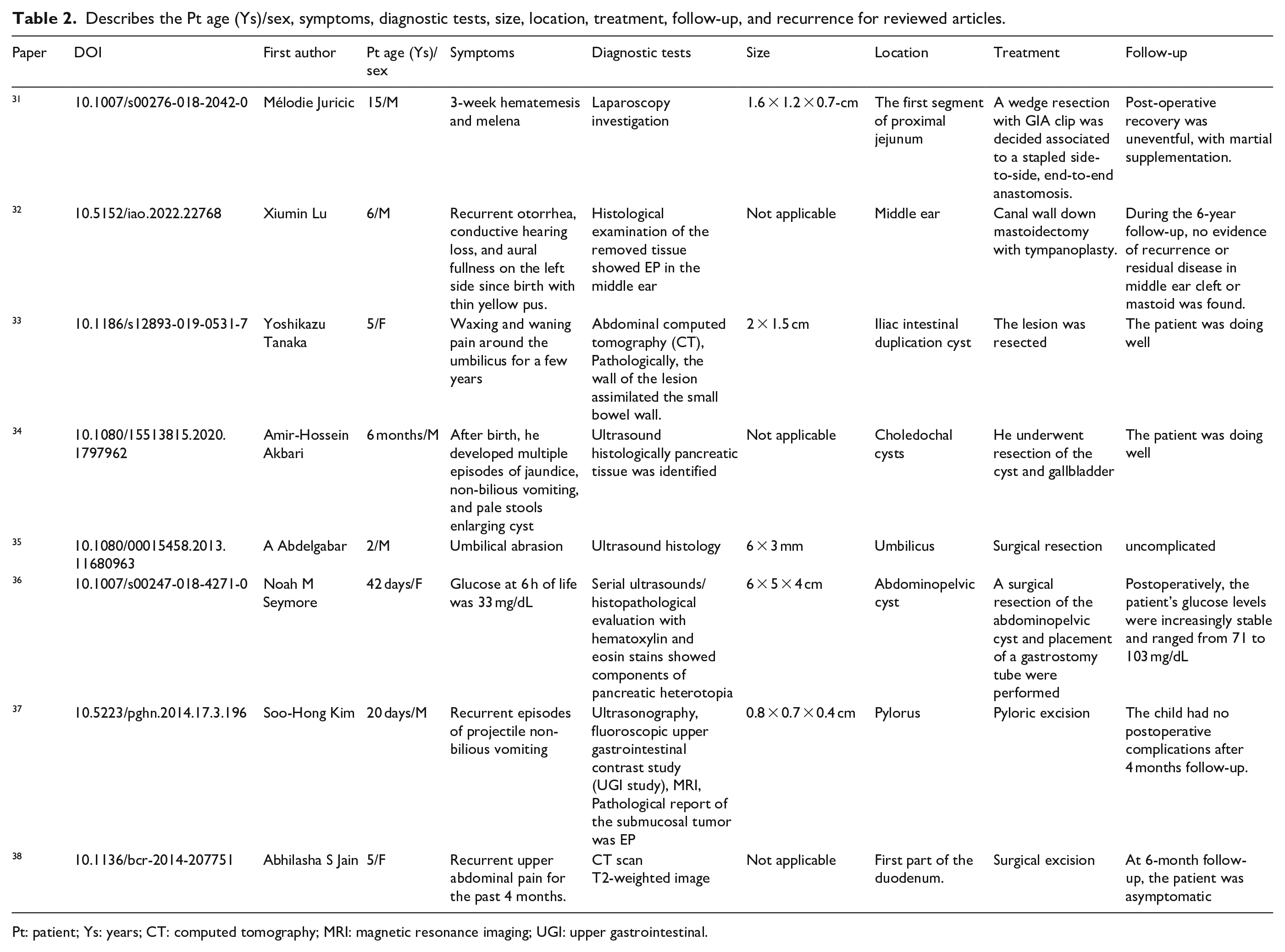
Pt: patient; Ys: years; CT: computed tomography; MRI: magnetic resonance imaging; UGI: upper gastrointestinal.
We noticed that the size of the EP did not reach large measurements, and each time, it ranged from several millimeters to several centimeters.31,38
We noticed that the subject of age did not follow a specific rule, as the EP was discovered in children of weeks and days old, as well as children of older ages. This is likely because it is in specific cases present in visually clear organs that helped to quickly detect it, such as the umbilicus 35 and the middle ear (by noting the tympanic membrane). 32 In other cases, the EP was also associated with another clinically glaring disease in the same place, which made its discovery earlier as the 6 months child who developed multiple episodes of jaundice, non-bilious vomiting, and it turns out later that he has choledochal cysts containing EP. 34
In most cases, we noticed the presence of the EP within the GI tract,31,33–37 and this does not negate the possibility of its presence outside it. 32
Symptoms varied between irritation (bleeding, abrasions),31,35 and obstruction, such as gastric outlet obstruction, 37 or even systemic hypoglycemia. 36 But abdominal pain remains the most common symptom,37,38 The presence of the EP in our patient was in the stomach, and it caused abdominal pain, in the absence of any obstructive symptoms.
Multiple diagnostic methods helped to head for the diagnosis of the EP (like CT, ultrasound), but each time, we need a histological study to confirm the diagnosis.31–38.
The only method of treatment used was surgical resection. Improvement and non-recurrence was the rule.31–38
Conclusion
In this study, our aim is to encourage physicians to consider the severity of symptoms when making a diagnosis and to conduct further investigations if the symptoms do not align with the initial diagnosis. The presence of additional accompanying diseases, as in the case presented, may complicate the diagnosis and require additional investigations to identify all underlying pathologies. While there is no established direct link between SMA syndrome and EP, both conditions can cause similar symptoms. Therefore, additional investigation is advised to assess any potential connections between the two conditions. It is recommended to seek further medical evaluation and consultation with a qualified healthcare professional to determine the appropriate course of action.
Footnotes
Acknowledgements
We wish to show our appreciation to Stemosis for Scientific Research, a Syria-based scientific research youth Official team managed by Dr Nafiza Martini, for the scientific environment they provided. A great thanks to that doctor who walks like a queen in the corridors of the hospital and walks behind her children’s calls on her name as if she is their friend. Who believes in her students. She gives for free. She makes everything easy. The wonderful Dr Lina Khouri. We also thank Dr Mahmoud Ayash and Dr Ayman Samoun for their great efforts in caring for the patient.
Author contributions
DB contributed to drafting, reviewing, editing, and approving the final manuscript. SH contributed to drafting, reviewing, editing, and approving the final manuscript. SM contributed to drafting, editing, and approving the final manuscript. SZ contributed to drafting, editing, and approving the final manuscript. SH contributed to drafting, editing, and approving the final manuscript. NM contributed to drafting, reviewing, editing, corresponding, and approving the final manuscript. LK contributed to reviewing, supervising, and approving the final manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the Legally Authorized Representative for the anonymized information to be published in this article.
Data availability statement
Not applicable.
Provenance and peer review
Not commissioned, externally peer reviewed.
