Abstract
Human umbilical cord mesenchymal stem cell-derived exosomes have gained attention as a promising, cell-free regenerative therapy due to their immunomodulatory and tissue-repair properties. This study retrospectively evaluated the safety and efficacy of nebulized human umbilical cord mesenchymal stem cell exosome therapy in three patients with asthma and/or chronic obstructive pulmonary disease who underwent weekly nebulized exosome therapy for 5 weeks at ALPS Medical Centre, Kuala Lumpur, Malaysia. Clinical outcomes were assessed using pulmonary function tests, inflammatory markers, and laboratory parameters. Post-therapy, pulmonary function improved, with increases in forced expiratory volume in 1 s from 2.59 to 3.4 L (p = 0.07) and forced vital capacity from 2.48 to 3.32 L (p = 0.11), while the forced expiratory volume in 1 s/forced vital capacity ratio significantly increased from 82.57% to 92.9% (p = 0.05), indicating reduced airflow limitation. Inflammatory markers, including C-reactive protein and eosinophil count, remained stable, and no significant changes were observed in hepatic, renal, or haematological parameters. Additionally, no adverse reactions or safety concerns were reported. In conclusion, nebulized human umbilical cord mesenchymal stem cell-derived exosome therapy demonstrated potential in improving pulmonary function and alleviating respiratory symptoms in patients with asthma and chronic obstructive pulmonary disease while maintaining a favourable safety profile. These preliminary findings support further investigation of exosome therapy as an adjunctive treatment for chronic respiratory diseases.
Introduction
Human umbilical cord mesenchymal stem cells (hUCMSCs) have emerged as a promising tool in regenerative medicine due to their multilineage differentiation potential and potent immunomodulatory properties. 1 Isolated from Wharton’s jelly and other umbilical cord components, hUCMSCs offer several advantages over other stem cell sources, including non-invasive procurement, minimal ethical concerns, and low immunogenicity, making them ideal for clinical applications. These cells have been widely explored for their therapeutic potential in a range of conditions, including inflammatory, autoimmune, 2 and degenerative diseases. 3
In recent years, increasing attention has been directed toward exosomes derived from hUCMSCs (hUCMSC exosomes) as a cell-free alternative to stem cell therapy. Exosomes are nanosized extracellular vesicles that serve as intercellular messengers, delivering bioactive molecules such as proteins, lipids, and microRNAs to modulate immune responses and promote tissue regeneration. 4 Unlike direct cell transplantation, exosome-based therapies circumvent risks such as immune rejection and uncontrolled cell proliferation while retaining the regenerative and anti-inflammatory benefits of their parent stem cells. 5 Studies have demonstrated that mesenchymal stem cells (MSCs) exosomes can enhance cellular repair, inhibit oxidative stress-induced apoptosis, and modulate inflammatory pathways, making them a compelling option for treating chronic and acute diseases, particularly those affecting the lungs. 6
Nebulized exosome therapy has shown potential in respiratory medicine, particularly in conditions such as acute lung injury, acute respiratory distress syndrome, chronic obstructive pulmonary disease (COPD), and asthma. 7 Preclinical studies indicate that exosomes can mitigate pulmonary fibrosis, reduce airway inflammation, and promote epithelial repair by modulating immune cell activity and suppressing excessive collagen deposition.8,9 Moreover, clinical reports suggest that stem cell-derived exosomes administered via inhalation can improve lung function and alleviate respiratory symptoms in patients with chronic lung diseases.10,11 Their ability to modulate inflammatory responses and enhance alveolar repair positions nebulized exosome therapy as a novel and potentially transformative approach in pulmonary medicine.
Mechanistically, hUCMSC-derived exosomes are enriched with immunosuppressive and reparative molecules, including soluble tumour necrosis factor receptors (sTNFRI and sTNFRII), interleukin-1 receptor antagonist (IL-1Ra), and soluble receptor for advanced glycation end products (sRAGE). These molecules have been shown to attenuate pro-inflammatory cytokine signalling (e.g. TNF-α and IL-1β), inhibit leukocyte infiltration, and promote anti-inflammatory immune cell phenotypes such as M2 macrophages, regulatory dendritic cells, and IL-10-producing regulatory T (Treg) cells, thus mitigating airway inflammation and preserving alveolar integrity in preclinical COPD models.12–15
The inhalational route offers a direct and efficient means of delivering exosomes to the injured pulmonary microenvironment, allowing targeted modulation of local immune responses with minimal systemic exposure. This is particularly important in COPD and asthma, where the local immune dysregulation and epithelial injury are central to disease progression. 16
Given the role of nebulized exosomes in improving lung function and alleviating respiratory symptoms in patients with lung diseases without significant safety concerns, they have been used in clinical settings to manage COPD. Therefore, the present study retrospectively evaluated the effects of nebulized hUCMSC exosomes in three patients with asthma and COPD who underwent this therapy at ALPS Medical Centre, Kuala Lumpur, Malaysia. Clinical outcomes were assessed based on pulmonary function tests (PFTs), inflammatory markers, and overall systemic safety. The findings demonstrated improvements in pulmonary function parameters, particularly in forced expiratory volume in 1 s (FEV1) and the FEV1/forced vital capacity (FVC) ratio, with no significant adverse effects on haematological, inflammatory, hepatic, renal, or metabolic markers. These results provide preliminary evidence supporting the feasibility and safety of nebulized hUCMSC exosome therapy as a potential adjunctive treatment for chronic respiratory diseases.
Methodology
Isolation and preparation of exosomes
The exosomes used in this study were provided by Celestialab Sdn Bhd, Kuala Lumpur, Malaysia. They were isolated from hUCMSCs obtained from Wharton’s jelly, following ethical and regulatory protocols. The isolation process was conducted in a Good Manufacturing Practice (GMP)-grade facility using GMP-certified reagents and media to ensure safety, purity, and consistency. Exosomes were isolated using filtration techniques, and their characteristics were confirmed through nanoparticle tracking analysis for particle size and concentration. They were also evaluated for sterility, mycoplasma contamination, and endotoxin levels (results are provided in Supplementary Table 3).
Treatment plan
Each exosome treatment dose consisted of 1 ml of exosome solution, containing a defined range of exosome particle numbers, mixed with 5 ml of saline, as described in previously published protocols. 10 The treatment was administered via inhalation using a medical-grade nebulizer (Electronic nebulizer; Vega Technologies Inc, Guangdong, China). Patients inhaled this solution for 30 min once a week for 5 weeks (weeks 1–5). This method ensured direct delivery of the therapeutic agents to the lungs, targeting the affected areas efficiently.
Assessment schedule
Baseline assessments were conducted in week 0, before the initiation of treatment, and included lung function tests, C-reactive protein (CRP) levels, complete blood count (CBC), and respiratory evaluations such as shortness of breath and chest tightening. Additional measurements included heart rate, oxygen saturation (SpO2) levels, lactate dehydrogenase (LDH), alanine aminotransferase (ALT), and creatinine levels. Post-treatment assessments were conducted in week 6, replicating the pre-treatment evaluations to determine the treatment’s impact.
Outcome measures
The primary outcome measures included improvements in lung function, reductions in CRP levels, and changes in blood count and respiratory values. Secondary outcome measures focussed on the reduction of symptoms such as shortness of breath and chest tightening, stabilization or improvement in heart rate and SpO2 levels, and decreases in LDH and ALT levels. Creatinine levels were monitored to assess any potential adverse effects on kidney function.
Data analysis
Statistical analysis was performed on the data collected from pre- and post-treatment assessments. Descriptive and inferential statistics were used to evaluate the significance of changes in the measured parameters, providing a comprehensive analysis of the therapeutic potential of nebulized hUCMSC-derived exosome therapy for patients with asthma and COPD.
Case description
The present clinical study was registered with the National Medical Research Register (ID-25-00600-F4A). Data collection and analysis were conducted following the ethical principles outlined in the Declaration of Helsinki and Malaysian Good Clinical Practice. This retrospective case series involved three non-consecutive patients who received nebulized exosome therapy derived from hUCMSC at ALPS Medical Centre. To evaluate the safety and efficacy of the nebulized hUCMSC exosome therapy, clinical data were collected from 2024 to 2025. Patient data were traced from the centre’s database and selected based on specific inclusion and exclusion criteria.
In this study, the inclusion criteria included individuals aged 18 years or older who had received nebulized hUCMSC exosome therapy at ALPS Medical Centre and provided informed consent for data publication. Exclusion criteria included individuals who had received other MSC-based therapies or products, individuals with known allergies or hypersensitivities to any component of exosome therapy, individuals with a history of cancer or other severe medical conditions, and individuals who were pregnant or breastfeeding. As part of the standard care protocol, each patient underwent a comprehensive evaluation before receiving exosome therapy, which included a detailed medical history review, physical examination, and laboratory testing.
The medical history review assessed patients’ overall health status and identified any contraindications for exosome therapy. The physical examination included an evaluation of general health, vital signs, and any acute exacerbations of existing medical conditions. Baseline laboratory tests, including a full blood count, white cell differentiation, diabetic screening, lipid profiling, liver function profile, renal function tests, gout screening, sexually transmitted disease screening, hepatitis B and C screening, cancer markers, and urinalysis, were conducted before therapy initiation. Pre-therapy PFTs were also conducted to establish baseline respiratory status. These tests included spirometry (Spirolab; Medical International Research, Viale Luigi Schiavonetti, Roma, Italy) values such as FEV1, FVC, and FEV1/FVC ratio, providing objective measures of lung function. Eligible patients were then scheduled for nebulized exosome therapy using the MyCelest hUCMSC-derived exosome product. Throughout the treatment process, patients were closely monitored to ensure safety and detect any adverse reactions.
This case series involved three patients aged 57–60 years, comprising two males and one female. All patients underwent nebulized exosome therapy for respiratory conditions such as asthma and COPD. Patients’ health statuses were carefully evaluated to confirm the absence of contraindications for therapy. Post-therapy, patients were observed for any acute hypersensitivity reactions before being discharged from daycare. Patients were instructed to report any new symptoms, such as fever, vomiting, insomnia, redness or pain at the administration site, rash, or shortness of breath, to the medical team. Follow-up visits were scheduled 2 weeks post-therapy for physical examination, and laboratory tests were conducted after 30 days to monitor blood parameters and evaluate the safety and efficacy of the nebulized exosome therapy.
Case series
Case 1: A 60-year-old male patient with a history of smoking, asthma, and chronic hypertension presented for nebulized hUCMSC exosome therapy. The patient was clinically categorized as having moderate persistent asthma. At baseline, the patient’s vital signs included a blood pressure (BP) reading of 134/94 mmHg, a pulse rate (PR) of 72/min, and SpO2 of 97%. Pre-therapy PFTs showed an FEV1/FVC ratio of 82%. Pre-therapy laboratory results showed stable blood parameters, and no contraindications were identified. The patient underwent nebulized exosome therapy without any immediate adverse reactions. During follow-up visits, the patient reported a noticeable improvement in breathing capacity and a reduction in the frequency of asthma exacerbations.
Case 2: A 60-year-old female patient with a moderate long-standing diagnosis of COPD was evaluated for exosome therapy. Baseline assessments indicated a BP of 134/87 mmHg, a PR of 78/min, and SpO2 of 97%. Pre-therapy PFTs revealed an FEV1/FVC ratio of 73%. The patient received the nebulized hUCMSC exosome therapy. No immediate hypersensitivity reactions were observed post-therapy. At the 2-week follow-up, the patient reported improved exercise tolerance and reduced dyspnoea. Subsequent laboratory tests at 30 days confirmed improved lung function, stable blood parameters, and no safety concerns.
Case 3: A 57-year-old male patient with a history of severe asthma and sinusitis underwent evaluation for nebulized exosome therapy. Baseline vitals included a BP of 119/75 mmHg, PR of 89/min, and SpO2 of 99%. Pre-therapy PFTs showed an FEV1/FVC ratio of 92%, indicating normal airflow. The patient’s pre-therapy blood tests showed no abnormalities or contraindications. Following the therapy, the patient was closely monitored and exhibited no immediate side effects. During follow-up evaluations, the patient reported fewer asthma attacks, improvement in sleep quality, and an overall improvement in quality of life, with enhanced respiratory function and reduced reliance on bronchodilator medications and inhalers.
Outcome, safety, tolerability, and efficacy measures
The present study aimed to evaluate the safety, tolerability, and potential efficacy of nebulized exosomes derived from hUCMSCs in patients with asthma and COPD. The study focussed on both objective laboratory indicators and patient-reported outcomes to assess the therapeutic effects of hUCMSC-derived exosomes.
Pre-administration standard laboratory tests were conducted for all patients, including a CBC with white cell differential, glucose and lipid profiles, liver function tests, and renal function profiles. PFTs were also performed before treatment to establish baseline respiratory status, including FEV1, FVC, and FEV1/FVC ratio, key parameters in assessing obstructive airway diseases. 17 Following the administration of nebulized hUCMSC-derived exosomes, patients underwent a comprehensive follow-up at 2 and 4 weeks post-treatment. Safety and tolerability were assessed through physical examinations, patient-reported symptoms, and repeat laboratory tests conducted 30 days after the initial treatment. Any reported adverse events, such as fever, shortness of breath, chest discomfort, or hypersensitivity reactions, were documented and managed accordingly.
Efficacy measures were evaluated based on improvements in respiratory function and symptomatology. 18 Repeat PFTs were performed 30 days post-treatment, with particular attention to changes in FEV1, FVC, and FEV1/FVC ratio, compared to baseline values. Subjective outcomes, including reduced frequency of exacerbations, improved exercise tolerance, and relief of respiratory symptoms such as wheezing and dyspnoea, were recorded through patient feedback.
The results obtained from the experimental protocol of the present study were analyzed using SPSS; IBM Corp., Armonk, NY, USA) for statistical analysis. A Shapiro–Wilk test was conducted to assess the normality of the data. For normally distributed data, a paired t-test was used to compare pre- and post-values within each case. For non-normally distributed data, the Wilcoxon signed-rank test was applied. This clinical case study involved three different individuals with a mean age of 59 years (SD = 1.7 years), including one female and two males, who received hUCMSC-derived exosome therapy between 2024 and 2025 (Table 1).
Subject characteristics for the study.

Across the patient cohort, notable improvements in pulmonary function and symptom relief were observed following nebulized exosome therapy. The parameters and their pre-and post-therapy values are presented in Table 2. As part of the assessment of tolerability, the serum albumin-to-globulin (A/G) ratio was also monitored as a marker of inflammation. The association between the A/G ratio and inflammatory processes for various conditions has been described in the literature. 19 The A/G ratio of all patients remained similar, both at baseline (mean = 1.46) and post-therapy (mean = 1.8) assessments, suggesting no significant increase (p = 0.21) in inflammatory activity in the patients.
Individual values of baseline and post-exosome therapy assessments.
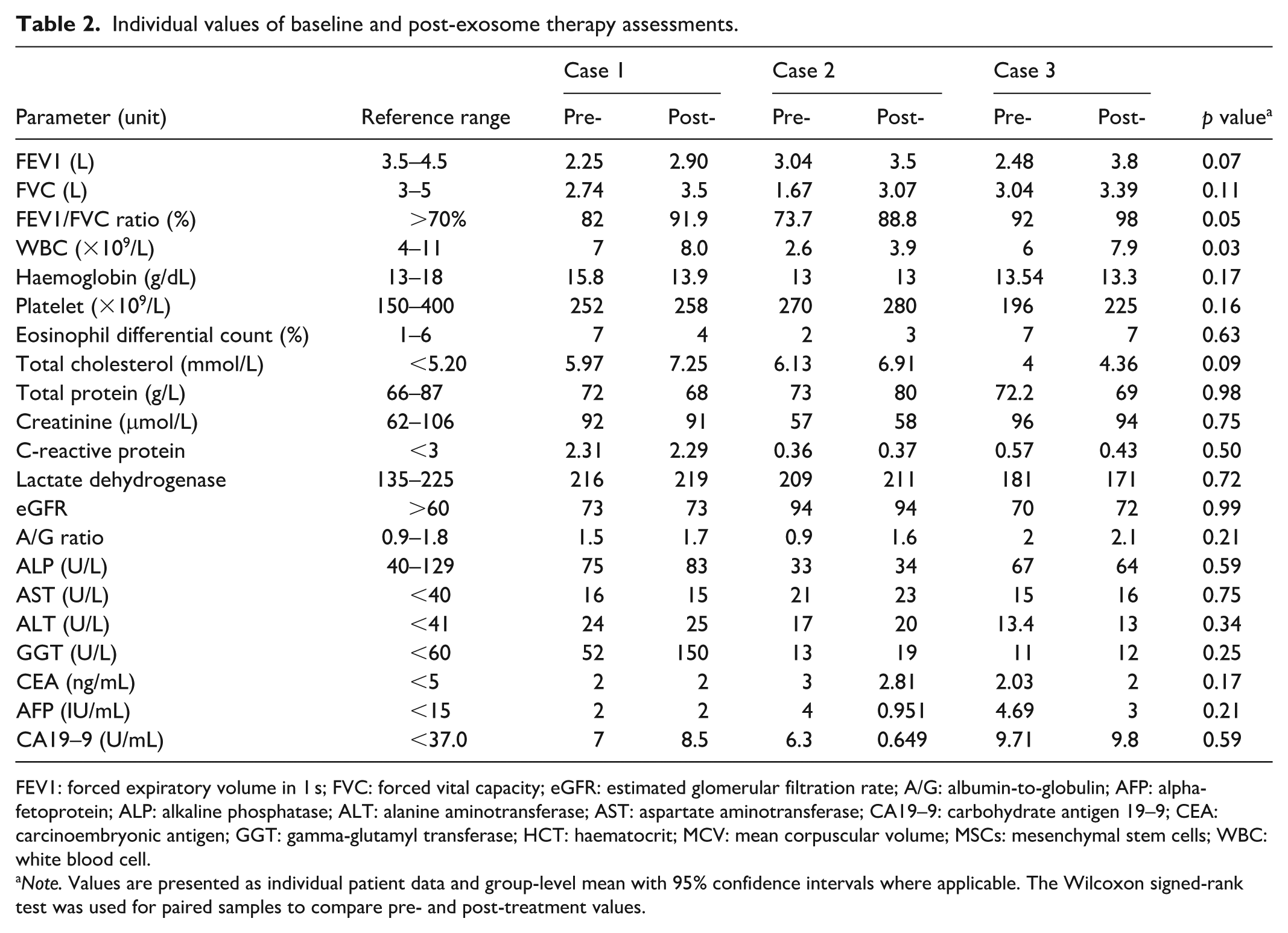
FEV1: forced expiratory volume in 1 s; FVC: forced vital capacity; eGFR: estimated glomerular filtration rate; A/G: albumin-to-globulin; AFP: alpha-fetoprotein; ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; CA19–9: carbohydrate antigen 19–9; CEA: carcinoembryonic antigen; GGT: gamma-glutamyl transferase; HCT: haematocrit; MCV: mean corpuscular volume; MSCs: mesenchymal stem cells; WBC: white blood cell.
Note. Values are presented as individual patient data and group-level mean with 95% confidence intervals where applicable. The Wilcoxon signed-rank test was used for paired samples to compare pre- and post-treatment values.
Tumour markers were also monitored at baseline and post-exosome therapy. At baseline, the mean carcinoembryonic antigen (CEA), alpha-fetoprotein (AFP), and carbohydrate antigen 19–9 (CA19–9) levels were within the normal range, and post-therapy assessments showed no significant changes (p = 0.59).
Discussion
Exosome therapy, derived from hUCMSCs, has gained increasing attention for its potential regenerative and immunomodulatory properties, particularly in pulmonary diseases. 20 We previously reported the safety of hUCMSCs, 21 the present study aimed to evaluate the effects of nebulized exosome therapy on pulmonary function, inflammatory markers, and overall systemic safety in a small cohort of patients. The findings suggest that exosome therapy may contribute to improved lung function while maintaining a favourable safety profile, with no significant adverse effects on inflammatory, haematological, hepatic, renal, or metabolic parameters (Table 2). The results are discussed in the context of pulmonary function improvements, immune response modulation, and overall tolerability.
The PFTs demonstrated notable improvements following exosome therapy. The mean FEV1 increased from 2.59 L pre-treatment to 3.4 L post-treatment (p = 0.07), suggesting enhanced airway function and improved ventilatory capacity. 22 Similarly, FVC improved from 2.48 to 3.32 L (p = 0.11), reinforcing the potential for the lungs to expand and contract, which is essential for efficient gas exchange. 23 The FEV1/FVC ratio, a key marker for assessing obstructive airway diseases, 24 showed a significant increase from 82.57% to 92.9% (p = 0.05), indicating a reduction in airflow limitation. Individually, all three cases demonstrated an upward trend in PFT parameters, with improvements in both FEV1 and FVC (Table 2), supporting the potential of nebulized exosome therapy in enhancing pulmonary function, particularly in patients with compromised respiratory capacity due to asthma or COPD. These improvements are consistent with previously published clinical observations in COPD patients treated with Exo-d-MAPPS, a formulation rich in soluble anti-inflammatory factors such as sTNFRI, sTNFRII, IL-1Ra, and sRAGE, which are also naturally present in hUCMSC-derived exosomes.25,26 In those studies, nebulized or intraperitoneally delivered MSC-exosome therapies significantly enhanced FEV1, peak expiratory flow, and 6-min walking distance, while reducing computed tomography (CT) markers of emphysema and lung hyperinflation. 25
Beyond pulmonary function, systemic inflammatory and haematological markers were evaluated to assess immune response and overall tolerability. The total white blood cell count increased from 5.2 to 6.6 × 109/L (p = 0.03), which may suggest a mild transient immune activation post-therapy. However, this increase remained within physiological limits and did not indicate an overt inflammatory reaction (Table 2). The eosinophil differential count, a marker for allergic inflammation often elevated in asthma, decreased slightly from 5.33% to 4.67% (p = 0.63), suggesting that the treatment did not exacerbate eosinophilic activity or allergic responses. This trend aligns with preclinical findings where hUCMSC exosomes suppressed Th2 cytokine activity, reduced IL-5 and IL-13 production, and promoted Treg cell expansion in asthma models.27,28 Similarly, Exo-d-MAPPS reduced the infiltration of TNF-α-, IFN-γ-, and IL-17-producing CD4+ and CD8+ T cells, neutrophils, and NK cells in cigarette smoke-induced COPD murine models, thereby mitigating airway inflammation and epithelial cell injury. 14
Similarly, CRP, a general marker of systemic inflammation, 29 remained stable, showing only a minor change from 1.08 to 1.03 (p = 0.50), further supporting the safety profile of nebulized exosome therapy in terms of inflammatory regulation.
Liver and renal function parameters were monitored to evaluate potential systemic toxicity. Liver enzymes, including ALT, aspartate aminotransferase (AST), alkaline phosphatase (ALP), and gamma-glutamyl transferase (GGT), remained within normal ranges post-treatment, with no statistically significant fluctuations (Table 2). ALT increased slightly from 18.13 to 19.33 U/L (p = 0.34), while AST showed minimal variation, rising from 17.33 to 18 U/L (p = 0.75), indicating no substantial hepatic stress or dysfunction. ALP and GGT also exhibited minor, non-significant changes, demonstrating the hepatic safety of the intervention. Similarly, renal function indicators, including creatinine and estimated glomerular filtration rate (eGFR), remained stable (creatinine: 81.67–81 μmol/L, p = 0.75; eGFR: 79–79.67, p = 0.99), suggesting that the therapy had no detrimental effects on kidney function. These findings are reassuring, given the known low immunogenicity and absence of teratoma formation risk associated with exosome-based treatments, in contrast to MSC transplantation, which carries concerns of ectopic differentiation and immune rejection.30,31
Lipid and protein metabolism markers were assessed to determine any metabolic impact of exosome therapy. Total cholesterol levels exhibited a mild increase from 5.37 to 6.17 mmol/L (p = 0.09), which, while not statistically significant, warrants further investigation in larger cohorts to assess potential metabolic effects. In contrast, total protein levels remained virtually unchanged (72.4 to 72.33 g/L, p = 0.98), suggesting that the therapy had no substantial impact on protein homeostasis or systemic nutritional balance.
In terms of safety and tolerability, no significant haematological disturbances were observed. Haemoglobin levels showed a slight decrease from 14.11 to 13.4 g/dL (p = 0.17), while platelet counts increased modestly from 239.33 to 254.33 × 109/L (p = 0.16), both remaining within normal physiological ranges. Importantly, tumour markers, including CEA, AFP, and CA19–9, remained stable and within normal limits, with no significant changes post-treatment (CEA: 2.34 to 2.27 ng/mL, p = 0.17; AFP: 3.56 to 1.98 IU/mL, p = 0.21; CA19–9: 7.67 to 6.32 U/mL, p = 0.59), further demonstrating the safety profile of nebulized exosome therapy and providing no indication of pro-tumourigenic activity.
Mechanistically, hUCMSC-derived exosomes are known to carry key immunoregulatory factors, such as IL-10, TGF-β, and miRNAs (e.g. miR-21, miR-146a) that shift immune cell polarization from proinflammatory M1 macrophages and Th1/Th17 cells toward anti-inflammatory M2 macrophages and Treg cells.32,33 This immune reprogramming was evident in previous animal models where lung leukocyte infiltration and proinflammatory cytokine levels (TNF-α, IL-1β) were significantly suppressed following exosome treatment. 14 Our current findings support a similar anti-inflammatory effect in a human clinical setting.
Collectively, these findings suggest that nebulized exosome therapy derived from hUCMSCs may have a beneficial role in improving pulmonary function in patients with asthma or COPD while maintaining a favourable safety and tolerability profile. The significant improvement in the FEV1/FVC ratio, combined with the absence of substantial inflammatory responses or adverse effects on haematological, hepatic, renal, or metabolic parameters, highlights the potential of exosome therapy as an adjunctive treatment for airway diseases. These results are consistent with growing evidence from both animal models and early-phase clinical studies of MSC-derived exosomes. While these preliminary findings are promising, larger-scale studies with longer follow-up periods are needed to further establish efficacy, optimal dosing, and long-term safety.
Conclusion
This retrospective case series provides preliminary evidence that nebulized exosomes derived from hUCMSCs may offer therapeutic benefits in patients with asthma and COPD. Improvements in pulmonary function, stability in inflammatory markers, and the absence of significant adverse effects suggest that this therapy is well-tolerated. While these findings are promising, larger-scale, controlled studies are necessary to confirm these results and establish standardized treatment protocols.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X251386547 – Supplemental material for A case report on nebulized human umbilical cord mesenchymal stem cell-derived exosome therapy for asthma and chronic obstructive pulmonary disease: A preliminary clinical evaluation
Supplemental material, sj-docx-1-sco-10.1177_2050313X251386547 for A case report on nebulized human umbilical cord mesenchymal stem cell-derived exosome therapy for asthma and chronic obstructive pulmonary disease: A preliminary clinical evaluation by Basmullah Yusof, Rui Qing Foo, Manickam Ravichandran, Mohammad Auwal Sa’ad and Seng Kong Tham in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
The authors would like to acknowledge the nurses from ALPS Medical Centre for their contribution to this study and their efforts and assistance in the collection of data.
Ethical considerations
This case series was registered and approved by the National Medical Research Register (NMRR; ID-25-00600-F4A), Ministry of Health Malaysia. Data were extracted per the ethical principles of the Declaration of Helsinki and Malaysian Good Clinical Practice.
Consent for publication
Written informed consent was obtained from all participants for publication of this case report.
Author contributions
Conceptualization: B.Y., R.Q.F., M.R., M.A.S., S.K.T. Study design: B.Y., M.R., M.A.S. Data curation: B.Y., M.R. Investigation: B.Y. Formal analysis: M.A.S. Methodology: R.Q.F. Project administration: M.R., S.K.T. Resources: S.K.T. Supervision: M.R., S.K.T. Validation: B.Y., M.R., S.K.T. Visualization: B.Y., R.Q.F., M.R., M.A.S. Writing – original draft: M.A.S. Writing – review and editing: R.Q.F., M.R., M.A.S., S.K.T. All authors reviewed the text and approved the final version of the manuscript for publication.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data from the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
