Abstract
A 66-year-old woman presented to the hospital with cutaneous necrosis of her right ankle and foot. Her symptoms began immediately after an intra-articular injection of hyaluronic acid for ankle osteoarthritis, which was performed 6 days before. Histopathology showed an intra-vascular hyaluronic acid embolus. The initial treatment approach was conservative, but the patient’s clinical state degraded. She was thus treated with sub-cutaneous hyaluronidase, the enzyme that degrades hyaluronic acid, which yielded a moderate improvement even though it was administered 22 days after the initial hyaluronic acid injection. Although hyaluronic acid embolism and subsequent cutaneous necrosis are well-known complications of dermal fillers, there are few reported cases of embolism following intra-articular injection. To our knowledge, this is the first time hyaluronidase has been used in this setting.
Introduction
In a dermatological setting, hyaluronidase (HYAL) is primarily used for treatment of skin necrosis caused by hyaluronic acid (HA) dermal fillers.1,2 The exact mechanism of this serious complication is understood to be caused by direct HA embolization, with intra-vascular occlusion and chemical damage to the endothelial lining, and extra-vascular factors such as external vascular compression by HA and inflammation-induced edema.1,3 Similarly, a handful of cases4–6 of skin necrosis caused by intra-articular (IA) injection of HA have been described, but recognition of this complication is often delayed and treatment is not well established. This report presents a case of cutaneous necrosis following IA HA injection successfully treated with HYAL.
Case report
A 66-year-old woman received a fluoroscopy guided IA injection of 3 mL of non-animal stabilised HA (Durolane®, 20 mg HA/mL, 0.5%–1% cross-linked HA) and 20 mg of triamcinolone acetonide in her right ankle. She presented to the hospital 6 days later with edema and painful violaceous patches with livedoid purpuric macules of her foot, ankle and shin (Figure 1(a)) which began a few hours after the procedure. Her vital signs, neurovascular exam and duplex ultrasonography were normal. All her blood analyses were normal, including blood cell count, biochemical assay, C-reactive protein, coagulation profile, autoantibodies and antiphospholipid profiles. The computed tomography angiography showed no visible emboli or signs of vasculitis. The fluoroscopic images were reviewed, confirming that the injection technique was intra-articular. Given the rapid development of symptoms following her IA injection, our most likely diagnosis was HA arterial embolism. The punch biopsy showed intra-vascular HA embolus (Figure 1(c) and (d)). She was initially managed with analgesics. Two days later, she was hospitalized due to increasing pain causing inability to walk, and expansion of the rash.
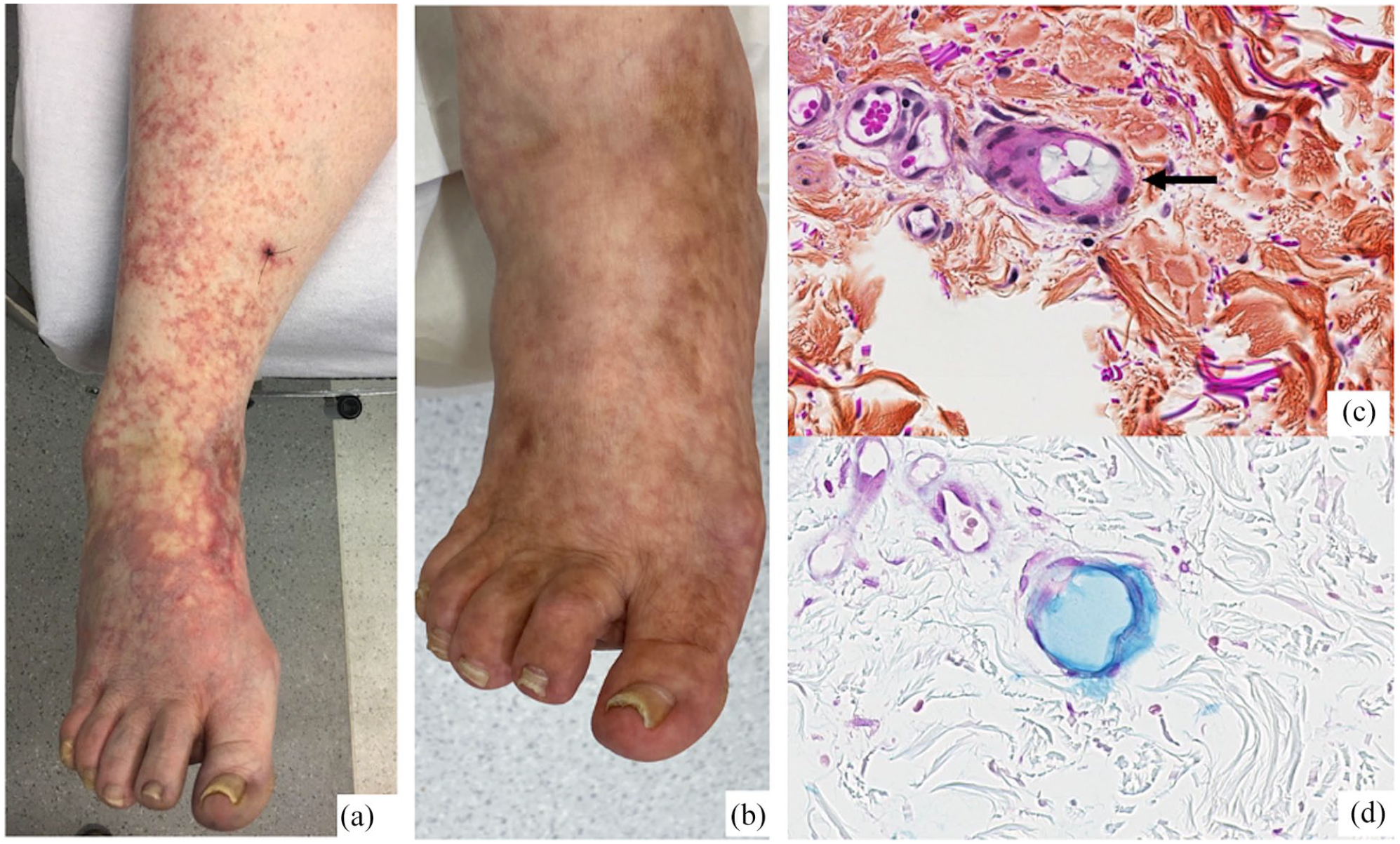
Clinical and histopathological images of our patient. (a) Anterior view of right foot and calf on day of first dermatology visit (Day 7 post intra-articular hyaluronic acid injection). (b) Anterior view at follow-up, 5 months later. (c) Small, rounded blood vessel in the reticular dermis showing mucinous slightly basophilic material in the vessel lumen (HPS stain) (black arrow). (d) Hyaluronic acid in blood vessel lumen positive with Alcian blue stain at pH of 2.5.
With the diagnosis confirmed by histopathology, given the patient’s pain, which was not controlled with increasing doses of opioids, and the prognosis of degradation of HA emboli over 4–6 months, 4 we opted to treat her with HYAL, which degrades HA. With the patient’s consent, 22 days after the initial IA injection, we proceeded to regularly spaced (1 cm) subcutaneous (SC) injection of 3 mL of HYAL (150 IU/mL, total of 450 IU), divided between the two most painful areas, the dorsum of her foot and internal malleolus. A small but notable lessening of the pain and of the violaceous patches was observed immediately after the treatment. No adverse events were noted. Her lesions resolved within 5 months (Figure 1(b)).
Discussion
Although HYAL is well described in the management of vascular complications of HA fillers,1,2 it has not been described previously in the treatment of IA HA vascular embolism. Few articles (Table 1) report HA cutaneous embolism following IA injection, but none used HYAL, although many suggested it as a potential treatment.4–6 The proposed mechanism of HA embolism, apart from inadvertent intra-arterial injection, is that HA escapes the joint through a vessel wall defect due to microtrauma or endothelial activation.4,5 Conservative treatment was our preferred initial approach. Due to the patient’s deteriorating clinical state, we decided to treat her with HYAL 22 days after the IA injection to hasten the degradation of the HA emboli even though animal and in vitro data suggest limited benefits after a delay of 4–24 h.1,7 Administration route was based on studies showing that SC HYAL can penetrate vessels to dissolve HA and is preferable to intra-arterial injection.2,7,8 The SC approach is also technically more feasible. Regarding the amount of HYAL injected, our decision to administer a conservative dose (450 IU) was based on the quantity of HA injected, the novelty of this treatment approach and the tolerability of multiple SC injections for the patient. However, given that stabilised HA is more difficult to degrade, that the ischemic zone was extensive and that the quantity of HYAL used for vascular complications of cosmetic HA fillers can be much higher, a larger dose could have been considered and might have yielded better results.1–3,7
Summary of cases of cutaneous hyaluronic acid embolism following intra-articular injection.

M: male; F: female; PAS: periodic acid–Schiff; LMWH: low molecular weight heparin.
This case highlights the importance for clinicians to be cognizant of this rare, albeit severe complication of IA HA injection. To our knowledge, this is the first time HYAL has been used in this setting. We observed clinical improvement and no side-effects. Early recognition and treatment of this adverse event will likely yield better outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Patient consent
The patient has provided an informed written consent for publication of her clinical information and images.
