Abstract
Diagnosis, management, and treatment of Dieulafoy’s lesion have been complicated since its first mention in 1884. With increased awareness over the decades, Dieulafoy’s lesions are being managed differently based on the physician’s expertise. Herein, we report a case of a 58-year-old female who was found to have a Dieulafoy’s lesion on endoscopy, with her treatment complicated by gastric necrosis and perforation after multiple embolizations and limited surgical resection.
Introduction
In the United States, upper gastrointestinal (GI) bleeding accounts for up to 20,000 deaths per year. 1 Believed to be responsible for only 1%–2% of GI bleeds, Dieulafoy’s lesion can cause obscure GI bleeding potentially resulting in life-threatening GI hemorrhage. 2 Dieulafoy’s lesion is a histologically normal but abnormally large arteriole which fails to decrease in diameter as it runs a tortuous course within the submucosa wall and protrudes through a small mucosal defect. 3 These lesions are commonly found in the stomach 4 and are two times more likely found in men than women in their fifth decade of life. 2 While there is no consensus on the treatment of these lesions, therapeutic endoscopy and angiography have been described. Surgical intervention is reserved as the last option. Here, we report a case of a 58 year-old female with Dieulafoy’s lesion who was found to have full-thickness necrosis of her stomach after embolization of her short gastric arteries and left gastric artery.
Case presentation
A 58-year-old female with a past medical history of hypertension and no past surgical history who presented with four episodes of dark, bloody stools over the course of 1 month. She was previously seen at an outside hospital and underwent multiple endoscopies for upper GI bleeding. At this time, patient was found to have a Dieulafoy’s lesion; however, this was not bleeding at the time of endoscopy. She subsequently underwent an angiography with interventional radiology (Figures 1–3). A blush was seen around a branch of the short gastric arteries, and coil embolization of two short gastric arteries without evidence of ongoing bleeding was performed. Patient was stable the day after the procedure and was discharged home.

Blush from short gastric arteries on angiography.

Bleeding from patient’s Dieulafoy’s lesion on endoscopy.

Hemoclips placed during endoscopy.
She returned to the hospital approximately 24 h later with complaints of dark tarry stools. Patient reported no previous history of GI bleeds, ulcers, alcohol abuse, renal or cardiac diseases, or abdominal surgery in the past. She denied chronic use of nonsteroidal anti-inflammatory drug (NSAIDS) or blood thinners. She denied constipation, diarrhea, light headedness, dizziness, nausea, or vomiting. Upon admission, the patient was tested for Helicobacter pylori and found to be negative.
On presentation, she had no abdominal pain; however, she was tachycardic with a hemoglobin of 6.3. The patient received a blood transfusion and re-admission for further workup. Endoscopy was performed and the patient was found to have a bleeding Dieulafoy’s lesion in the gastric body, which was treated with hemoclips and epinephrine injection endoscopically. This was unable to control bleeding, so interventional radiology was consulted. Patient then underwent an angiography and her left gastric artery was embolized with Gelfoam (Pfizer, New York, NY, USA) and coil embolization (Figure 4). The patient was initially stable after her procedure. On hospital day 2, she developed hypotension and her hemoglobin was found to be 6.1 down from 7.7. She was transfused and underwent a second endoscopy procedure. She was noted to have old clotted blood in her stomach, but no source of bleeding was identified. The previous Dieulafoy’s lesion was inspected and clips were noted to be in place without active bleeding. The patient was stabilized after her procedure; however, the patient had hematemesis and another episode of hypotension on hospital day 4. Endoscopy was again performed and she was found to have a substantial amount of clotted blood with identification of bleeding from her Dieulafoy’s lesion. The lesion was clipped, and general surgery was consulted.
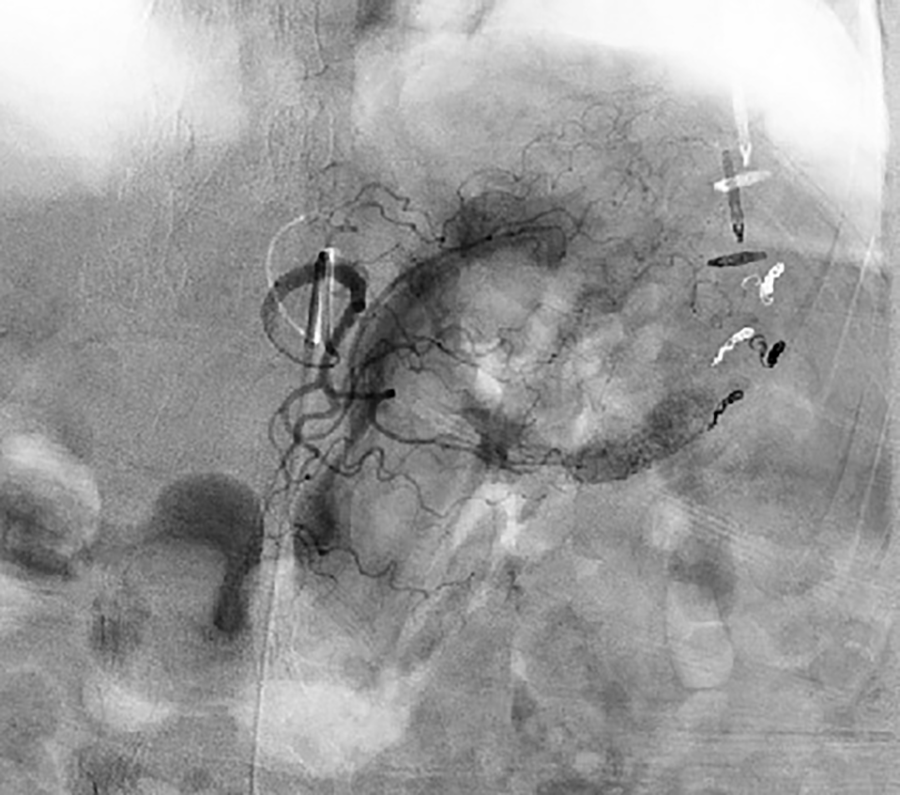
Blush from LGA on angiography.
The patient was taken to the operating room for gastric wedge resection. Intraoperative endoscopy was performed and Dieulafoy’s lesion was identified. No bleeding from Dieulafoy’s lesion was seen; however, the patient was noted to be bleeding from a visible vessel on the lesser curve of the stomach. With the assistance of a Da Vinci Surgical System, a gastric wedge resection was performed. The specimen was removed and inspected in the operating room. Necrotic gastric mucosa and Dieulafoy’s lesion were identified in the specimen. She was transferred back to the floor for post-op care.
On post-op day 3, the patient became hypotensive and tachycardic. Hemoglobin was noted to be 6.9 down from 9.3. CT scan was performed and suspicious for a perforation around her previous wedge resection (Figure 5). Patient’s upper endoscopy perforation of the gastric body was identified (Figure 6). She was taken back to the OR for an exploratory laparotomy. Upon entering the abdomen, a large portion of the anterior stomach extending to the lesser curvature, upward to the esophagogastric junction, was found to be necrotic. A small perforation was noted just medial to the previous staple line of her wedge resection. An additional Dieulafoy’s lesion was identified in the patient’s stomach at this time. All areas of necrotic tissue were resected, including a substantial portion of the lesser curvature of the stomach extending from the gastroesophageal junction to the incisura. A pyloroplasty was performed during this procedure. A 46 French bougie was passed down the esophagus and the stomach was reconstructed. Using a 2-0 vicryl suture in a canal stitch, the antrum was approximated to the remaining viable tissue of the gastric body. This extended approximately 2 cm distal to the GE junction inferiorly to the greater curvature. This was reinforced with 3-0 vicryl with Lembert sutures. A leak test was performed, and an omental patch was placed over the suture line. The abdomen was closed, and the patient was returned to the floor. A nasogastric tube was maintained until an upper GI study was performed on post-op day 5 and no leak was seen. She was maintained on a liquid diet for 2 weeks and has recovered well postoperatively (Figure 7). Pathology of the specimen showed chronic gastritis with ulceration and perforation and acute serostitis. Specimen was negative for H Pylori or malignancy. At 30- and 60-day follow-up, patient was doing well. She was re-admitted to the hospital for a single episode of hematemesis at POD14, but endoscopy at the time found no source of bleeding and an intact suture line without ongoing signs of necrosis. Patient reported she is eating normally at 60-day follow-up with no ongoing symptoms.
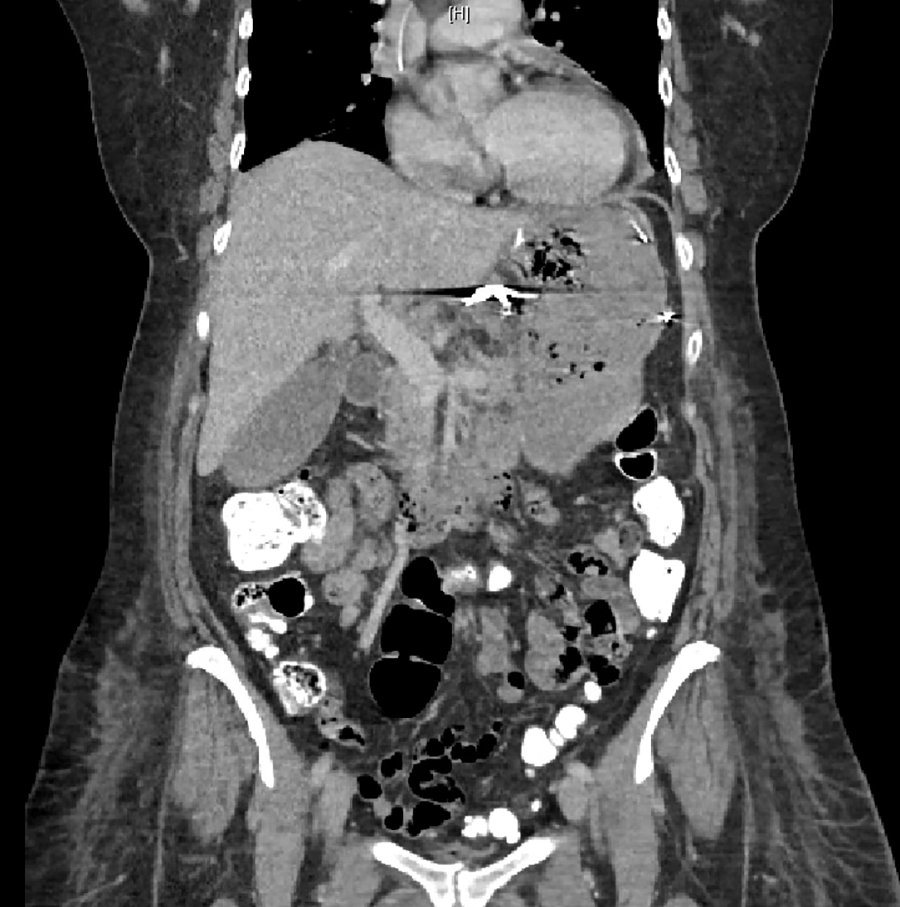
CT showing extraluminal gas around lesser curvature of stomach suspicious for perforated stomach.

Endoscopy showing perforated gastric lumen.
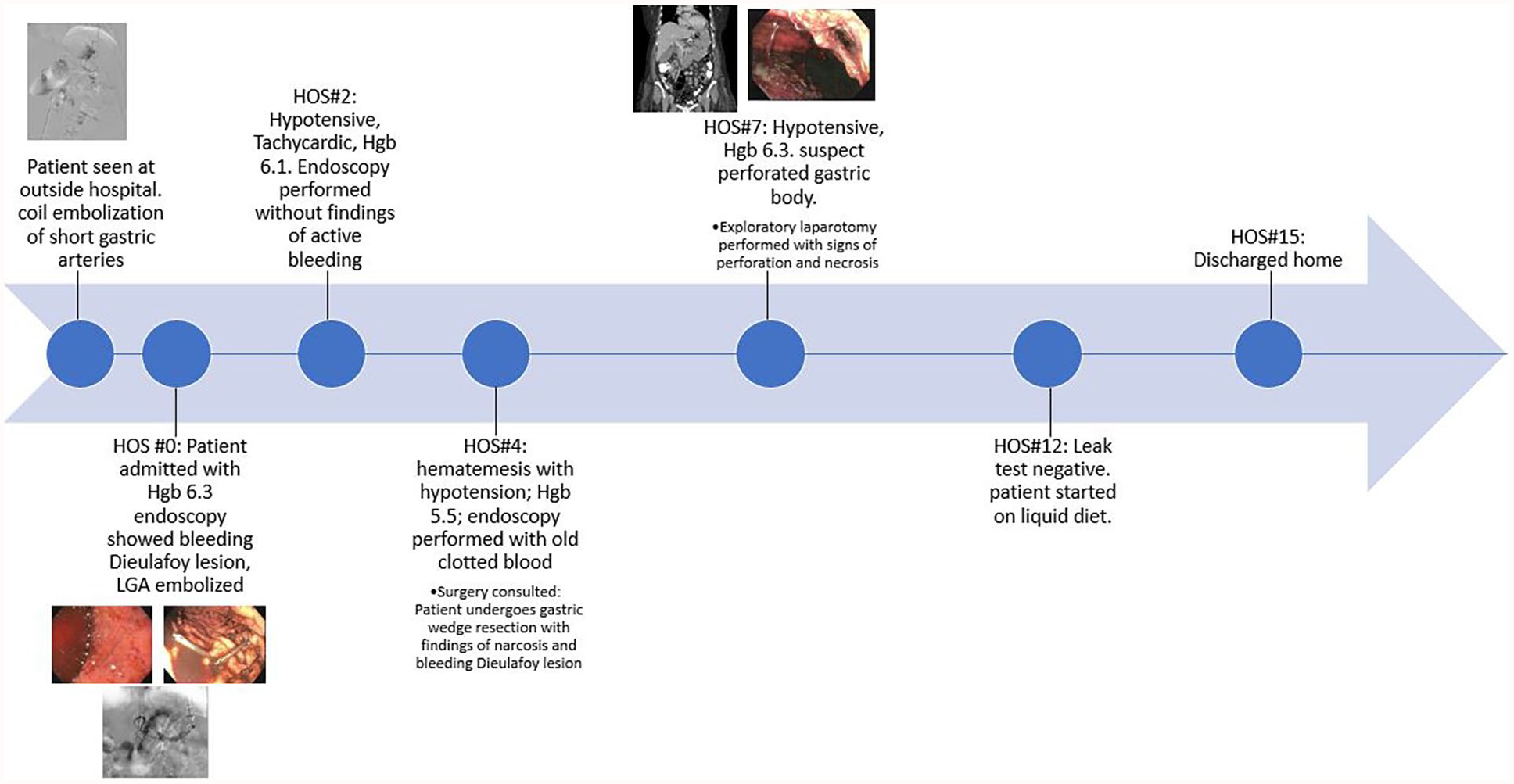
Timeline of major events for patient during the hospital stay.
Discussion
The total number of hospitalizations with Dieulafoy’s lesion has increased sixfold from 2002 to 2011, while the cost of care per hospitalization has increased from US$14,992 to US$25,294 during the same period. This may be due to increase in medication use such as NSAIDS, anticoagulation, and antiplatelet medications which can cause erosive gastritis and rupture. Conditions that cause epithelial damage and formation of aberrant vessels that are prone to bleeding are also becoming more commonly diagnosed. 6 Before 1990, bleeding from Dieulafoy’s lesion was treated surgically. However, there is no current consensus regarding treatment, and the decision is based on the patient’s clinical presentation, site of lesion, and physician’s expertise in treating GI bleeding. Current endoscopic modalities include epinephrine injections, sclerotherapy, and hemostatic clipping. Surgery may ultimately be necessary if bleeding cannot be controlled. Di Nardo et al. 7 studied pediatric cases of Dieulafoy’s lesions over the last 10 years and found hemostatic clipping to be successful in many cases. Many patients require multiple interventions due to a bleeding Dieulafoy’s lesions leading to complications and a prolonged hospital stay.
In the patient presented here, patient initially underwent endoscopy at an outside hospital, where she was found to have bleeding from branches of her short gastric arteries. Dieulafoy’s lesion was identified at time of initial endoscopy, but no intervention was performed. Dieulafoy’s lesions carry a mortality of up to 80%. 2 We suspect no intervention was performed initially on the lesion due to bleeding from another source having been identified. It is unclear if embolization or endoscopy itself causes Dieulafoy’s lesion to bleed. Patient ultimately needed operative intervention. There is no one consensus on operative technique for management of Dieulafoy’s lesions. Both open and laparoscopic techniques have been described. Laparoscopic techniques rely on successful identification of location of bleeding. In this case, bleeding was identified endoscopically and a wedge resection was performed with a Da Vinci Surgical System. Usage of the robot was physician preference. Eisenberg and Bell 8 described four successful cases of interoperative endoscopy with laparoscopic wedge resection; however, they admit that a larger series needs to be performed to establish efficacy.
In our case, therapeutic endoscopy was unsuccessful and embolization of two short gastric arteries and the left gastric artery (LGA) was attempted. We suspect her gastric necrosis was a result of her embolizations, specifically the LGA embolization. LGA embolization has been previously attempted with no complications for the treatment of Dieulafoy’s lesion. 9 In addition, there are ongoing clinical trials to evaluate LGA embolization for weight loss and no adverse effects from embolization have been reported. 10
It is not uncommon to embolize an artery in the stomach, as it has an extensive network of collateral blood supply. As such, embolization of the LGA does not typically cause extensive gastric necrosis seen in this case. We were only able to find one similar case where embolization of LGA led to full-thickness gastric necrosis; however, it was thought to include a combination of factors that were not present in our patient such as severe atherosclerotic diseases and a history significant of alcohol abuse and liver disease. 11
Conclusion
Dieulafoy’s lesions can typically be controlled with endoscopic techniques. This patient shows only endoscopic techniques may not be enough. She ultimately needed multiple embolizations and surgical management. The stomach has an extensive network of collateral blood supply and embolization of the left gastric artery does not typically cause necrosis. Physicians treating upper GI bleeding should know the various ways to control bleeding and risks associated with each. Our case is an example of necrosis and perforation of the stomach following embolization of multiple gastric arteries and limited surgical resection.
Footnotes
Acknowledgements
The views expressed in this publication represent those of the authors and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
