Abstract
We reported four cases of intramural haematoma of the descending thoracic aorta. Four patients, aged 55–82 years, hypertensive, were transferred from the emergency room of other hospitals due to the appearance of epigastric pain and left thorax pain. All patients underwent computed tomography angiography reporting the presence of intramural haematoma. Three patients underwent a drug therapy to maintain a controlled hypotension. A computed tomography revaluation was performed documenting (1) an increase in the thickness of the intramural haematoma, (2) the appearance of a penetrating ulcer within the haematoma and (3) the appearance of several penetrating lesions throughout the thoracic aorta. Patients required the placement of one or two thoracic aorta endoprosthesis. For the fourth patient, the hyperdense appearance of the intramural haematoma and the presence of pleural effusion suggested an urgent treatment intervention. All patients underwent a placement of cerebrospinal fluid catheter and drainage before treatment. All patients were treated with endovascular intervention with 100% technical success and absence of migration or retrograde type A dissection. There were no complications related to femoral surgical access or access routes. Perioperative mortality was null; no patient had paraplegia. No strokes, transient ischemic attack or perioperative myocardial infarction were observed. The average hospitalization was 5 days. After 3 months, angio-computed tomography reported for all patients a complete reabsorption of the intramural haematoma and a complete exclusion of the penetrating ulcer of the aortic wall present at the time of the intervention. There have been no cases of distant thoracic aortic tears. Endovascular treatment must be considered the preeminent treatment for thoracic aortic haematoma. Best timing to perform the endovascular procedure depends on the patient clinical picture and on stability of hemodynamic parameters.
Introduction
The descending thoracic aorta diseases contribute to significantly increase mortality from cardiovascular diseases.1,2 Nowadays, thoracic aortic pathologies have been identified that are different from classic aortic dissection but having a similar thoracic symptomatology, and classified more generally as acute aortic syndromes (AASs).3,4 AASs include five forms of dissection. Type I includes the classic dissection with true and false lumen; type 2 includes the intramural haematoma (IMH) of the thoracic aorta, which is a less common form but a precursor of the classic dissection; the type 3 includes the dissection in patients with Marfan syndrome; the type 4 includes the presence of a penetrating ulcer of the aortic wall (PAU) and the type 5 includes the post-traumatic dissection of the thoracic aorta usually at the level of the aortic joint.
The IMH of the thoracic aorta represents up to 6%–10% of AASs and can be defined as ‘a dissection without intimal tear’.5,6 It is a haematoma confined within the aortic wall whose mechanism of initiation is nowadays unclear, although a hypothesis that this haematoma may be due to the rupture of the vasa vasorum has been formulated by some authors.7,8 Symptoms are similar to chest pain that occurs during aortic dissection. The evolution of the IMH is to evolve in 28%–47% of cases true the classic B-type dissection with true and false lumen, while another 20%–45% evolve in the formation of a pseudoaneurisma and in a rupture. Only a small proportion of patients evolve towards reabsorption of haematoma with only antihypertensive medical therapy. 6 The favourable prognostic factors are the young age of the patient, the absence of a thoracic aortic aneurysm (diameter <4.0–4.5 cm) and the haematoma thickness <1.0 cm. Unfavourable prognostic factors are the persistent and refractory thoracic pain in medical therapy and the appearance of the computed tomography (CT) angiography of a penetrating ulcer of the aorta. The different localization of the haematoma, in ascending thoracic aorta or bow and descending thoracic aorta, imposes different clinical decisions. The presence of an ascending aortic haematoma imposes an aggressive intervention of immediate repair of the aorta, while an IMH in the aortic arch or in the descending thoracic aorta allows a less aggressive intervention and short re-evaluation with radiological examination after stabilization of the symptomatology.
The PAU is characterized by the appearance of an ulceration of an atherosclerotic aortic plaque that penetrates through the intima and deepens into the average tunic of the aortic wall. 9 The incidence of PAU among AASs is between 2.3% and 7.6%. 10 It usually affects patients with extensive atherosclerotic pathology around the 7th decade. In addition, it usually affects the descending thoracic aorta at the mid-distal 1/3 level and rarely the ascending aorta. In these rare cases, urgent surgical intervention is essential.4,11 The evolution of a PAU includes the development of a localized IMH due to the erosion of the vasa vasorum of the aortic part by the ulcer, or the formation of a pseudoaneurisma, or progression of dissection or aortic rupture up to 40% of patients.4,12,13 In this article, we reported four cases of IMH of the descending thoracic aorta treated at the Department of Cardiovascular Diseases of the Gemelli Molise Hospital in Campobasso, Italy.
Materials and methods
From January 2015 to December 2017, four patients affected by IMH of the descending thoracic aorta were treated at the Department of Cardiovascular Diseases of the Gemelli Molise Hospital in Campobasso, Italy. Informed consent has been obtained from the patients for publication of the case report and accompanying images.
Case 1: A 55-year-old patient, hypertensive, transferred from the emergency room of another hospital due to the appearance of epigastric pain and associated with left thorax pain. The patient underwent thoracic and abdominal computed tomography angiography (angio-CT) reporting the presence of IMH (from the origin of the subclavian to the superior mesenteric artery). The patient underwent a drug therapy to maintain a controlled hypotension – 100/70 mm Hg for 7 days. A new CT revaluation documented an increase in the thickness of the IMH and a more hypodense appearance. Then, a thoracic aorta endoprosthesis (GORE TAG 343415) was placed from the emergence of the subclavian left artery.
Case 2: A 71-year-old patient, hypertensive, transferred from the emergency room of another hospital for the appearance of epigastric pain and associated with pain in the left hemithorax. The patient underwent an angio-CT of thoracic and abdominal aorta reporting the presence of IMH (from the origin of the subclavian to the superior mesenteric artery). The patient underwent a drug therapy to maintain a controlled hypotension – 100/60 mm Hg for 7 days. A new CT evaluation reported the appearance of a penetrating ulcer within the haematoma. Then, a thoracic aorta endoprosthesis (GORE TAG TGE 404015) was placed in correspondence of the penetrating lesion.
Case 3: A 82-year-old patient, woman, hypertensive, transferred from another hospital for the appearance of epigastric pain and associated with left thorax pain. The patient underwent an angio-CT of thoracic and abdominal aorta reporting the presence of IMH (from the origin of the subclavian to the superior mesenteric artery). The patient underwent a drug therapy to maintain a controlled hypotension – 100/70 mm Hg for 7 days. A new CT revaluation documented an increase in the thickness of the IMH and the appearance of several penetrating ulcers throughout the thoracic aorta from the origin of the subclavian to the superior mesenteric artery. Two endoprostheses (GORE TAG TGE 373720 and 373715) were positioned in order to exclude the thoracic aorta tract due to the presence of the penetrating ulcerations.
Case 4: A 81-year-old patient, hypertensive, transferred from the emergency room of another hospital due to the appearance of bar-irradiated epigastric pain and associated with left sore thorax pain. The patient underwent an angio-CT of thoracic and abdominal aorta reporting the presence of IMH from the origin of the subclavian with a left pleural effusion that extended to the superior mesenteric artery. The symptomatology completely disappeared after 2 days. However, the hyperdense appearance of the IMH and the simultaneous presence of pleural effusion suggested an urgent treatment intervention. Haematoma was excluded by placing a Valiant Captivia endoprosthesis (Medtronic 3434200) starting from the origin of subclavian to about 2 cm before the celiac trunk, in order to avoid the risk of spinal ischaemia due to the reduced thickness of haematoma at this level.
All patients underwent a placement of cerebrospinal fluid catheter and drainage before treatment.
In all four cases, the evolved aorta was not covered completely in order to avoid the risk of Adamkiewicz artery occlusion.
Results
Patients’ characteristics are shown in Table 1.
Population characteristics.

IMH: intramural haematoma; PAU: penetrating ulcer of the aortic wall; TGU (GORE): conformable GORE TAG thoracic endoprosthesis.
All patients were treated with endovascular intervention with 100% technical success and absence of migration or retrograde type A dissection. There were no complications related to femoral surgical access or access routes, as shown in Table 2. Perioperative mortality was null. No patient had paraplegia and in all patients a cerebrospinal fluid catheter and drainage was placed for 48 h. No strokes or transient ischemic attack (TIA) or perioperative myocardial infarction were observed, as shown in Table 3. The average hospitalization was 5 days. After 3 months, angio-CT reported for all patients a complete reabsorption of the IMH and a complete exclusion of the PAU present at the time of the intervention. There have been no cases of distant thoracic aortic tears.
Surgery characteristics.

Mortality and postoperative morbidity.
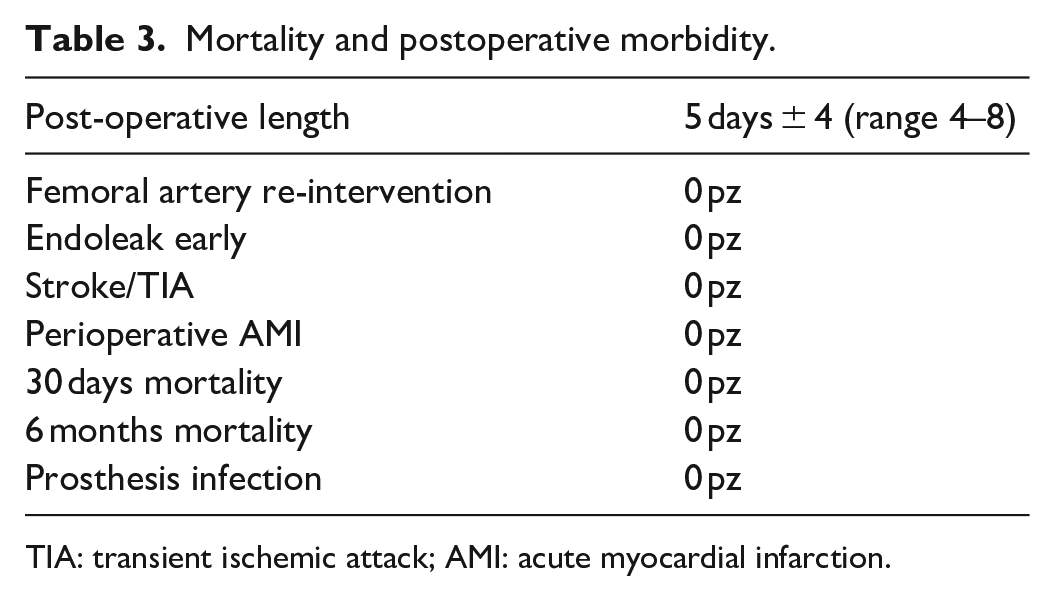
TIA: transient ischemic attack; AMI: acute myocardial infarction.
As example, Figure 1 shows the temporal evolution of IMH for case 1, from the first imaging after thoracic pain (a), after 10 days medical therapy (b), control after 30 days thoracic endovascular aortic repair (TEVAR) (c) to control after 12 months TEVAR (d).
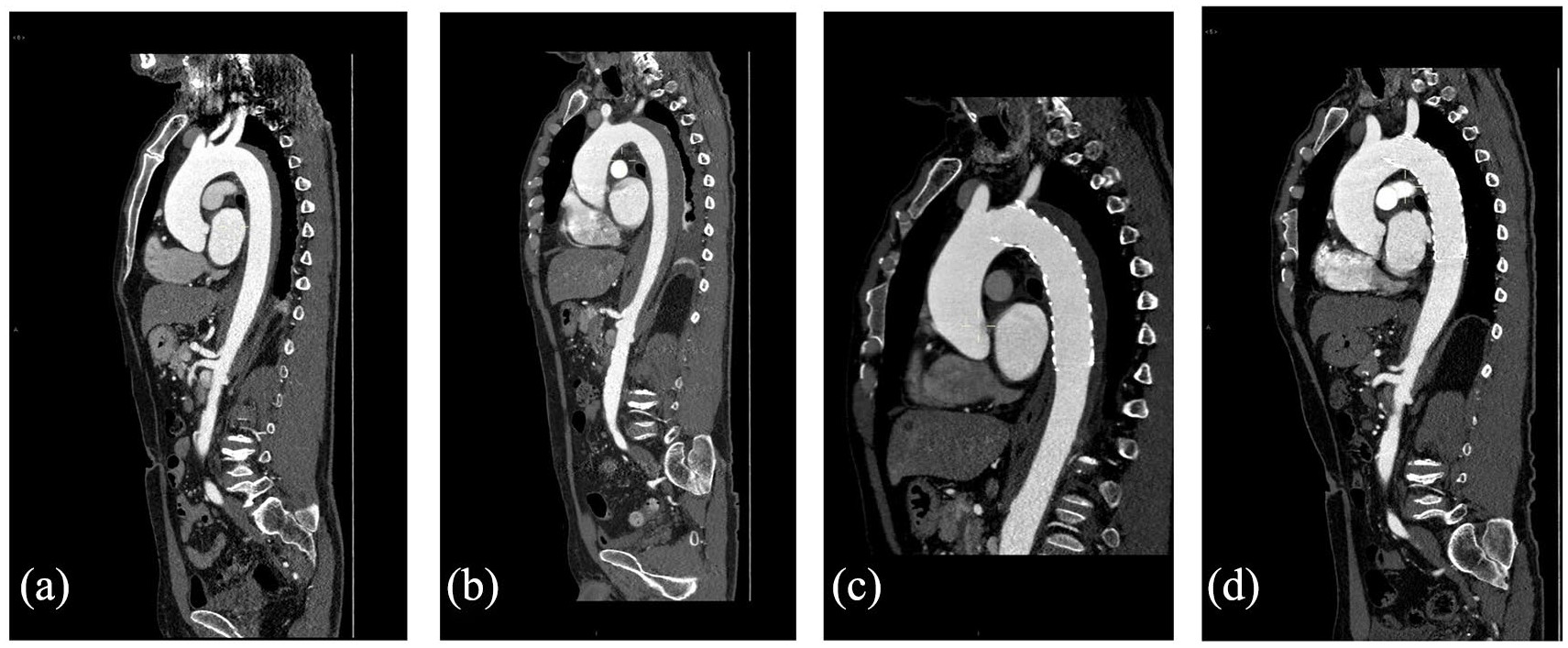
Intramural haematoma temporal evolution for case 1: (a) first imaging after thoracic pain, (b) after 10 days medical therapy, (c) control after 30 days TEVAR and (d) control after 12 months TEVAR.
Discussion
The definition of AAS includes the presence of five types of dissection ranging from classical aortic dissection, to the IMH of the descending thoracic aorta, to the penetrating ulcer of the aortic wall, to the post-traumatic dissection of the thoracic aorta. While for the classical dissection of the thoracic aorta, the management and treatment of aortic pathology are now codified, the indications to a possible treatment are still not clear for IMH and penetrating ulcer. Shennan 14 was the first to describe in 1934 a case of a penetrating ulcer of the aortic wall. The pathophysiological mechanism that was described was an erosion by the atherosclerotic plaque of the inner elastic lamina towards the middle tunic with the formation of a penetrating ulcer. The natural history of this pathology is still not known and the treatment intervention has been very controversial. Stanson et al. 15 in 1986 argued that it was necessary to have an aggressive approach towards this pathology, since it was necessary to avoid the transmural perforation of the vessel and therefore the haemorrhage. Other authors16,17 asserted the need to have a more conservative approach trying to perform a clinical and instrumental observation even if 30% of these patients could require a delayed surgery. Ganaha et al. 18 argued that in the presence of IMH of the descending thoracic aorta, the continued persistence of pain despite antihypertensive therapy, or the appearance of the following radiological signs to the chest CT scan, should prompt the surgeon to perform surgery because the risk of breakage could be high. In fact, the presence of a pleural effusion, the increase in the thickness of the IMH with a thickness of more than 12 mm or the appearance of a PAU within the haematoma pose the suspicion that the aorta wall can be strongly weakened and therefore malicious. 19 In our four cases, patients underwent a non-invasive monitoring of vital parameters (blood pressure, heart rate, diuresis and saturation) with important antihypertensive therapy in order to maintain a controlled hypotension (100/60 mm Hg) for at least 7 days. Then, the thoracic aorta CT-angiography was repeated. These patients were treated because of the persistence of a dull chest pain and the presence of one of aforementioned radiological signs present at the control CT at 7 days.
Patients presenting an IMH of the thoracic aorta or a penetrating ulcer of the thoracic aortic wall present a range of co-morbidities such as hypertension, chronic lower limb arterial disease and coronary artery disease. Therefore, an open surgical operation on the descending thoracic aorta would be burdened by a mortality and morbidity risk of up to 20%. In fact, this approach would include an extended thoracotomy, a possible cardiopulmonary bypass, aortic clamping with relative medullary ischaemia and prolonged pulmonary ventilation for the longer duration of the operation.
The endovascular approach to aortic pathology has allowed us to treat these diseases with lower mortality and morbidity. Dake et al. 20 for the first time in 1992 treated a thoracic aortic aneurysm by placing an endoprosthesis in the descending thoracic aorta. The endovascular treatment allows with a simple femoral access to locate the endoprosthesis with a much shorter surgical time, avoiding aortic clamping and cardiopulmonary assistance.
The incidence of endovascular treatment complications is also very low. In fact, the incidence of cerebrovascular complications, such as stroke or TIA, is 1%–2% and the onset of perioperative myocardial infarction is 0%. The other complication may be the appearance of a retrograde type A dissection whose incidence is 0.5%. Paraplegia in endovascular treatment is 1%–2% and may increase up to 6% in patients who have already performed ‘endovascular’ or ‘open’ surgery at the abdominal aorta level for aneurysmal pathology where the lumbar arteries, which vascularize the marrow, were surgically linked or endovascularly closed. Another important factor in order to avoid paraplegia is to extend the length of the endoprosthesis as little as possible, in order to preserve a greater number of intercostal arteries. In fact, in patients with IMH of the thoracic aorta or with a penetrating ulcer of the aortic wall, it is sufficient to use a single endoprosthetic piece in order to reduce the risk of paraplegia. 21 Moreover, the use of the cerebrospinal fluid catheter and drainage is fundamental in these patients in order to deliquorate them already during the intervention in the case in which an increase in the endorachoid pressure appears above 15 mm Hg.
In our small experience, all the patients were subjected to catheter placement and in Case 3, a drainage was performed for the positioning of two endoprosthesis for the vast extension of PAUs at the aortic level and also in the case 2 where the single endoprosthesis was implanted at the level of the thoracic aorta flush with the celiac tripod. In all other cases a single endoprosthesis was placed and there was no need for cerebrospinal fluid catheter and drainage. Also in our series, there were no cerebrovascular complications nor paraplegia nor retrograde dissection of Type A, as shown in Table 3.
In addition, it must be highlighted that all patients had a complete reabsorption of the IMH. One hypothesis is that this favourable outcome could be due to the positioning of the endoprosthesis that would reduce the arterial supply at the haematoma level. In particular, we believe that the placement of the stent graft causes an increase in pressure on the vessel wall and therefore it helps to reduce haemorrhage within the medial and adventitia tunica of the aorta.
We also preferred to use the GORE endoprosthesis, since it has a greater ability to adapt to the anatomy and it allows a reduced oversizing with respect to other endoprostheses.
An open question is still debated in order to decide optimal time for the endovascular treatment of IMH of the descending thoracic aorta, whether early or after stabilization of the clinical picture. In our experience, we tried to stabilize blood pressure and control chest pain in the first three patients; after 7 days, we repeated angio-CT in order to evaluate the evolution of the haematoma, considering the increase in extension, diameter and appearance of PAU. This approach allowed the positioning of a thoracic endoprosthesis in order to completely cover that part of the thoracic aorta where the haematoma is located. Only for the fourth patient, we adopted a more aggressive approach because the patient had a left pleural effusion which presented an impregnation with contrast medium to the chest CT scan. The timing of endovascular treatment is very important for the prognosis and survival of the patients.
Conclusion
The IMH of the thoracic aorta and the penetrating ulcer of the aortic wall can be considered as two forms of AASs and are a variant of the aortic dissection. The pathophysiological mechanisms are not distinct but seem to overlap with a form evolving in the other. Since the angio-CT shows that the penetrating ulcer of the aortic wall is formed in the same area where there was an IMH, endovascular treatment represents the gold standard for the treatment of aortic disease. The surgery timing should be performed after a period of careful hemodynamic monitoring unless there are signs of haemodynamic instability or the presence of a left pleural effusion to the chest CT at the time of diagnosis. The key point in treatment is the choice of the most appropriate timing to perform the endovascular procedure.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
