Abstract
An elderly woman admitted in our geriatric inpatient unit suffered from disturbing outbursts of crying and, less frequently, episodes of laughing. The patient was diagnosed with pseudobulbar affect related to a mixed neurodegenerative disorder. This condition is often underdiagnosed and undertreated, despite being relatively frequent in patients with neurodegenerative disorders. This case report describes the treatment of pseudobulbar affect in this patient. The only available treatment in Canada for this condition, antidepressants, was not effective for our patient. Dextromethorphan/quinidine is a good accepted alternative, but the combination is not marketed in Canada. To manage this problem, we used compounded quinidine capsules and dextromethorphan cough syrup. The crying of our patient improved significantly and rapidly after the initiation of this treatment. This case will help professionals to review their central role in treating this complex and disabling condition.
Keywords
Introduction
Pseudobulbar affect (PBA) is a condition associated with excessive crying or laughing in situations that would normally not trigger this type of reaction. The reaction is either disproportionate or incongruent to the situation.1 –3 This condition is relatively frequent in neurological disorders, 4 but, according to a panel consensus, it is often underdiagnosed and undertreated. 5 PBA can also be misdiagnosed with a psychiatric condition because of the presentation. 6 One accepted treatment of PBA, dextromethorphan/quinidine (DM/Q) combination, is not available in Canada. This case report describes the treatment with compounded quinidine capsules and dextromethorphan cough syrup in a patient diagnosed with PBA related to a mixed neurodegenerative disorder. The role of the pharmacist was important in this case because it supported the medical team in having access to DM/Q and monitoring its efficacy and security. This case will help professionals review their central role in treating this complex and disabling condition.
Case presentation
An 86-year-old woman admitted in our geriatric inpatient unit exhibited frequent episodes of involuntary, uncontrolled, particularly disturbing outbursts of crying and, less frequently, uncontrolled episodes of laughing. She also had episodes of wandering at night. The situation was becoming untenable and could not be managed by her husband anymore. Past medical history was positive for hypertension, osteoporosis, gait abnormality, nocturnal urinary incontinence, and Alzheimer’s disease (AD) with related behavioral problems. Her medications included amlodipine (2.5 mg daily), venlafaxine (75 mg daily), quetiapine (25 mg at bedtime), and calcium/vitamin D (500 mg/400 units daily). Parkinsonism was present on physical examination.
The initial workup included biochemical and laboratory investigation to exclude metabolic, inflammatory, hormonal, and toxic causes for dementia. All results were within normal limits. This patient had an obvious adverse impact on activities of daily living and, as she had a progressive and severe multi-domain cognitive decline, she could not perform an MMSE (Mini-Mental State Examination). Brain computerized tomography (CT) scan performed 4 years earlier showed mild cerebral atrophy and leukoaraiosis in the white matter regions of the brain (Figure 1). An electromyogram was performed to rule out motoneuron disease, which was negative. Magnetic resonance imaging (MRI) revealed nonspecific subcortical signal abnormalities of the white matter associated with subcortical ischemic changes and a left-sided cerebellar lacuna (Figure 2). Brain positron emission tomography (PET) with fluoro-2-deoxy-

CT brain showing mild cerebral atrophy and leukoaraiosis in the white matter regions of the brain.

MRI brain showing (a) nonspecific subcortical signal abnormalities of the white matter associated with subcortical ischemic changes and (b) a left-sided cerebellar lacuna.
Venlafaxine was stopped to try different medications. Table 1 describes the medication trials received by the patient during her hospitalization in order to reduce her crying and laughing symptoms. DM/Q was determined to be the most effective medication. A mild improvement was observed by the husband following 3 days of treatment. After 1 week, the effect was more apparent, as crying episodes were less frequent and of shorter duration. After 2 weeks, the crying episodes were reduced by 50% and wandering at night was less disturbing. At that point, the patient was discharged home with DM/Q and then 1 year later moved to a nursing home, where she died a few weeks after admission.
Medication trials during hospitalization.

PBA: pseudobulbar affect; DM/Q: dextromethorphan/quinidine.
Discussion
The pathophysiology of PBA is not well established; the best hypothesis remains a disruption of the cortico–pontine–cerebellar circuit, resulting in a lack of emotional control.1,6 Several neurotransmitters are implicated in PBA, mostly serotonin and glutamate. 1 This condition is associated with different neurological disorders such as stroke, amyotrophic lateral sclerosis (ALS), multiple sclerosis (MS), traumatic brain injury (TBI), AD or Parkinson’s disease (PD). 4 In one study, among several neurological disorders, the mean prevalence of PBA was 36.7%, and TBI was the most prevalent etiology at 52.4%. 4 In our case, PBA was secondary to a mixed neurodegenerative disorder (possibly AD or/and DLB), which correlates with the scientific literature. Patients affected by PBA have an increased risk of social and relationship problems as well as psychiatric symptoms.1,6 The medical team initially considered relocating the patient to a nursing home because her husband was overwhelmed by the situation. Eventually, the DM/Q combination greatly helped in reducing her symptoms and allowed her to return home.
The most recent diagnostic criteria for PBA published by Miller et al. 1 are listed in Table 2. The Center for Neurologic Study–Lability Scale (CNS-LS) is also helpful for PBA screening. Table 3 shows the self-report items of the CNS-LS developed by Moore et al. 7 Each item must be evaluated on a scale of 1 to 5: 1 (never), 2 (rarely), 3 (occasionally), 4 (frequently), and 5 (most of the time). 7 The CNS-LS is strictly validated for screening PBA in ALS and MS. 2 Therefore, using this scale for neurological disorders other than ALS and MS, as was the case in our patient, requires caution. The CNS-LS cutoff point to detect PBA is 13 or more for ALS and 17 or more for MS. 2 The case description and the neurologist evaluation confirmed that our patient had the four essential criteria seen in Table 2 to diagnose PBA.
Diagnostic criteria for PBA proposed by Miller et al. 1
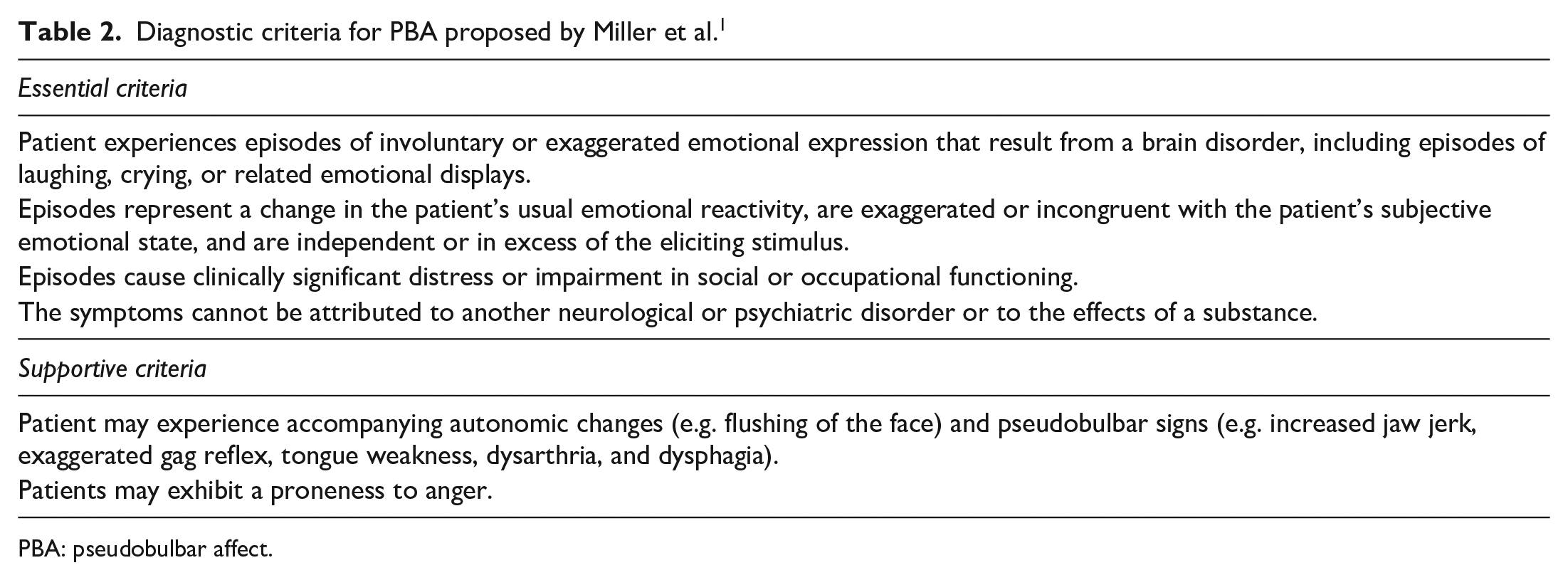
PBA: pseudobulbar affect.
Self-report items of the CNS-LS developed by Moore et al. 7

CNS-LS: Center for Neurologic Study–Lability Scale.
Table 4 summarizes the differential diagnosis of PBA. PBA can be misdiagnosed with psychiatric conditions, contributing to potentially unnecessary treatment and a risk of increasing adverse drug reactions or drug interactions. 6 Two of the main features that help differentiate PBA from a psychiatric condition are the duration and the congruence of emotion.1,6 In PBA, the distress episode lasts from 1 s to a few minutes, 8 and it is either disproportionate or incongruent to the situation.1,6 In contrast, the mood changes observed in a psychiatric condition persist in time and are usually congruent to the situation.1,6 The treatment of PBA is challenging as a depressive episode can coexist as frequently as 50% in patients with PBA. 6 The psychiatric evaluation of our patient was helpful to eliminate comorbid psychiatric conditions.
PBA: pseudobulbar affect; MS: multiple sclerosis.
Treatment of PBA is described in Table 5. Published studies assessing the benefits of antidepressants in PBA are small, difficult to compare, and included patients where the underlying disorder was usually stroke.
1
Response to antidepressants in PBA seems faster and occurs at lower doses than in psychiatric conditions.
2
The only approved treatment of PBA by the Food and Drug Administration (FDA) and the European Union (EU) is DM/Q.
9
The active drug dextromethorphan is an N-methyl-
Double-blind studies in the treatment of PBA. 2
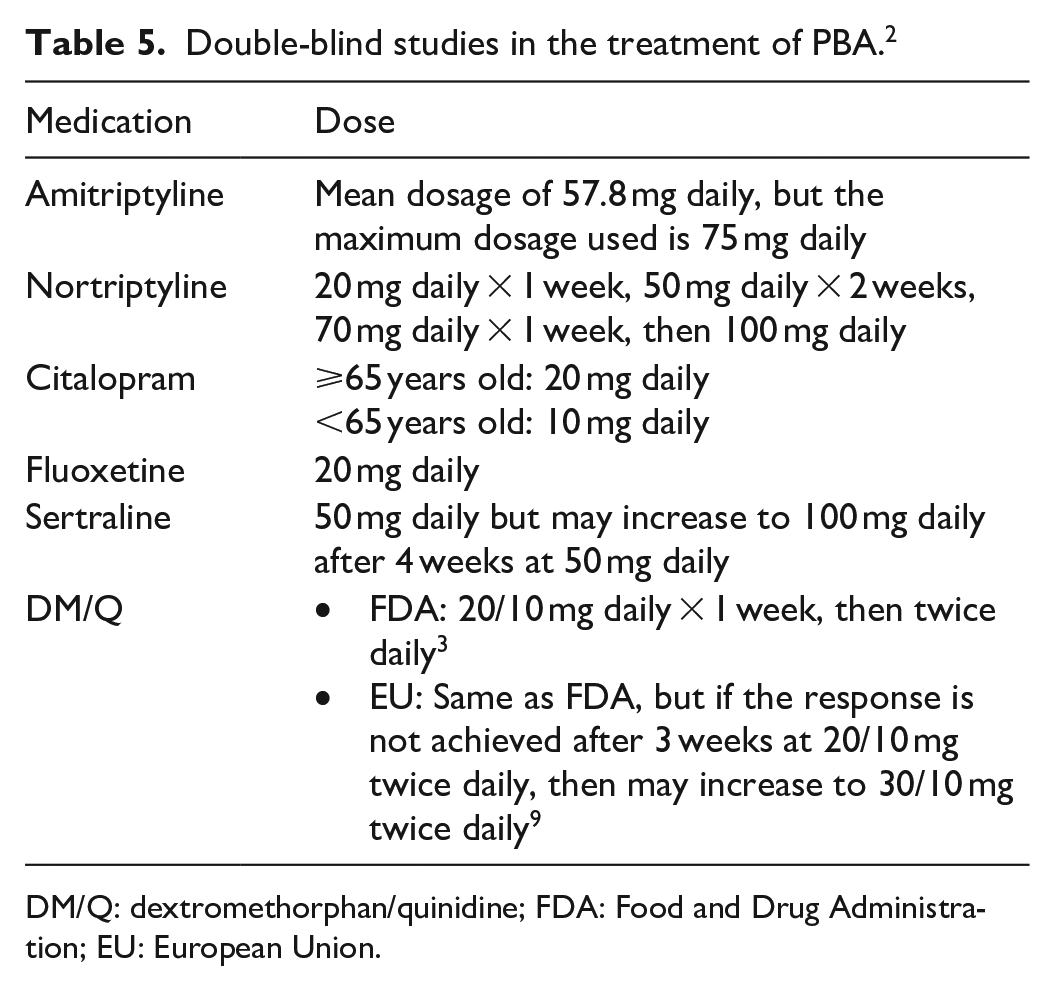
DM/Q: dextromethorphan/quinidine; FDA: Food and Drug Administration; EU: European Union.
The most frequent adverse drug reactions from short-term trial of DM/Q are diarrhea, dizziness, cough, vomiting, asthenia, peripheral edema, urinary tract infection, influenza, elevated gamma-glutamyltransferase, and flatulence. 3 An electrocardiogram (ECG) is recommended after the first dose of DM/Q to monitor the QT interval, 3 even though, according to the European Medicines Agency, there is a low risk of QT prolongation for patients without cardiac disease. 9 The QT interval of our patient was normal after the first dose of DM/Q. Recognizing quinidine is a CYP2D6 inhibitor, drug interactions must be considered, especially if the interaction involves a drug that prolongs QT interval.3,9 Moreover, quinidine is metabolized by CYP3A4; CYP3A4 inducers or CYP3A4 inhibitors must therefore be avoided. 9 CYP3A4 inhibitors could increase the risk of QT prolongation. 9 The use of an antidepressant with DM/Q should be approached with caution due to a possible risk of pharmacokinetic or pharmacodynamic interactions (QT prolongation, serotonin syndrome), depending on the antidepressant. 10 Finally, quinidine is a P-glycoprotein inhibitor.3,9 The sertraline could have been reassessed for our patient when we noticed the effectiveness of DM/Q, but the medical team decided to keep it as they did not want to risk any deterioration. Indeed, the goal of care was to provide comfort, which was much improved compared to her state upon admission. We considered the risk of interaction low, and the combination was well tolerated.
Since DM/Q is not available in Canada, we had to find an alternative for our patient. First, we used the expertise of a pharmaceutical laboratory to make compounded 10 mg quinidine capsules. As we did not know whether the patient would need 20 or 30 mg of dextromethorphan, we determined that a dextromethorphan cough syrup was an easy available option to facilitate the titration. However, we stopped at 20 mg, as it achieved a good effectiveness. Interestingly, we found two case reports with positive response for PBA in which DM/Q was changed to dextromethorphan cough syrup plus fluoxetine, considering fluoxetine is also a CYP2D6 inhibitor.11,12 This treatment combination was not considered for our patient as she had presented akathisia induced by fluoxetine.
Conclusion
PBA is a common finding in neurological disorders and it must not be confused with a psychiatric condition. Good patient care is essential to minimize the disruptive consequences of PBA on the patient and his or her family. A subset of patients will not respond to antidepressants for treating PBA; in this case, DM/Q is a good alternative. This case report could help pharmacists and other professionals considering compounded quinidine capsules and dextromethorphan cough syrup as an alternative to the marketed DM/Q combination when it is not available. The pharmacist can play an important role in supporting the medical team in the treatment and monitoring of patients with this condition.
Footnotes
Acknowledgements
The authors thank Audrey Attia for her support with the literature search, Theresa Feeser for editing the manuscript, and Dr David Landry for selecting CT/MRI images. All of the authors approved the final version and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from a legally authorized representative for anonymized patient information to be published in this article.

