Abstract
In our case report, we discuss a 1-day-old boy presenting with blueberry muffin syndrome diagnosed with Langerhans cell histiocytosis. The diagnosis complicated by an initial difficult-to-interpret biopsy showing only a hint of perifollicular CD1a-positive cells; however, given our team’s strong clinical suspicion of Langerhans cell histiocytosis, a second biopsy of a more mature lesion was done and showed typical histopathology. This case introduces the possibility of perifollicular Langerhans cells early in this condition, demonstrates the importance of appropriate biopsy site selection, and highlights the importance of maintaining a high degree of suspicion when there is poor clinicopathologic correlation. Our case report contains a comprehensive table which reviews the systemic and cutaneous clinical features, as well as the laboratory, pathology, and imaging findings for the differential diagnoses of blueberry muffin baby.
Keywords
Introduction
Langerhans cell histiocytosis (LCH) is a disorder characterized by proliferation and infiltration of clonal Langerhans cells into tissue(s) that is thought to occur through both inflammatory and neoplastic mechanisms.1,2 The BRAF V600E mutation has been implicated and may promote expansion of myeloid dendritic cell precursors.1,3,4 Historically, the disorder was divided into four subtypes (Letterer–Siwe disease, Hand–Schüller–Christian disease, eosinophilic granuloma, and Hashimoto–Pritzker disease), but is now considered a clinical spectrum that ranges from single system to extensive multi-systemic disease (MSD).5–8 Herein, we report a case of LCH that was nearly missed due to atypical histopathology on initial skin biopsy, which suggests a potential novel origin of clonal cells early in the disease.
Case report
A 1-day-old term male presented to hospital with multiple necrotic skin lesions. His mother was healthy; however, the antenatal course was complicated by presumed viral gastroenteritis in the first trimester, three viral upper respiratory infections in the second trimester, and a two-day history of diarrhea and nausea 2 weeks prior to delivery. The pregnancy was otherwise unremarkable with protective serologies, negative group B streptococcus screening, and normal ultrasounds. Family history was unremarkable and there was no history of travel. The family had dogs, chickens, turkey, and sheep. Labor was precipitous, with the patient born via spontaneous vaginal delivery to paramedics at term. Apgars were 10 and 10, and the placenta was delivered intact. A midwife arrived shortly after delivery and recommended urgent evaluation for his skin lesions.
Examination revealed approximately 20 cutaneous lesions ranging from brown macules to purple-blue-black necrotic and hemorrhagic-crusted papulonodules (Image 1). The two largest lesions were each 0.5 cm in diameter, located on the midline back and shoulder (Image 2). In addition to full skin examination, a thorough full physical examination verified by multiple pediatricians and pediatric specialists was normal.

In this photo, several brown macules and purple-blue-black necrotic and hemorrhagic-crusted papulonodules are visible on the right forehead, and abdomen.
Larger lesion 0.5 cm purple-blue-black necrotic and hemorrhagic-crusted papulonodule on the patient’s upper back.
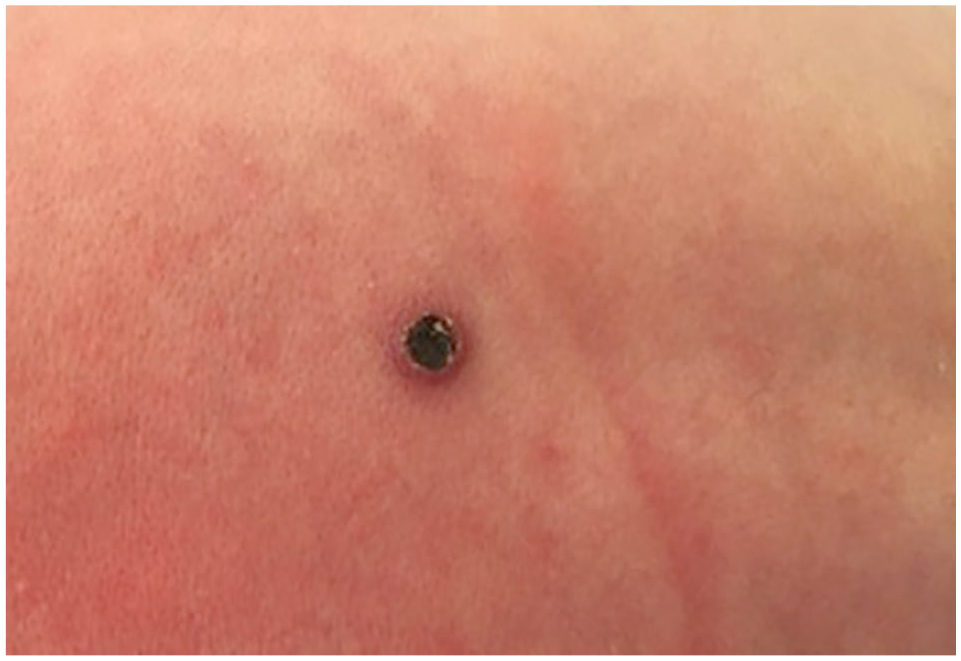
A punch biopsy was performed on a subtly violaceous crusted macule on the right forearm (Image 3). Pathology demonstrated an isolated focus of dense perifollicular histiocytic and dendritic cellular infiltrate sparing the epidermis (Image 4(a)). Eosinophils were not identified. Immunohistochemistry for CD1a (Image 4(b)), CD163, S100, and Cyclin-D1 were strongly positive in the lesional cells and confirmed them to be Langerhans cells. A repeat biopsy (Image 5(a)) of a fully developed crusted necrotic blue-black papule was pursued given the poor clinicopathologic correlation. This second sample revealed extensive epidermal ulceration and dense infiltrates of eosinophils and CD1a-positive Langerhans cells with vesicular nuclei with prominent grooves and ample eosinophilic cytoplasm in the dermis and subcutaneous tissue (Image 5(b)). BRAF mutation testing was negative.

Smaller 0.3 cm violaceous crusted macule on the patient’s arm.
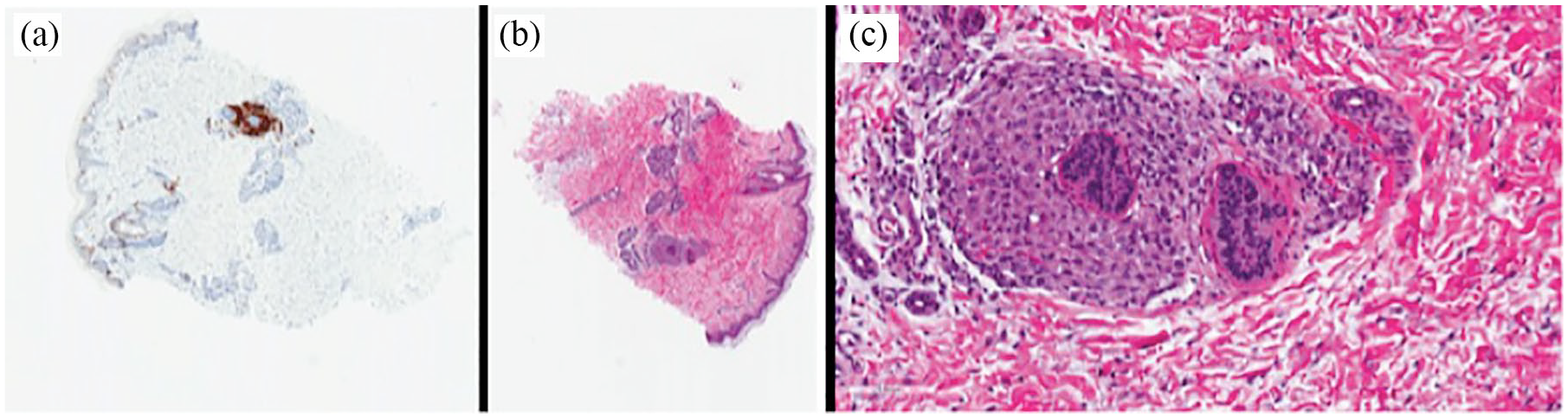
Immunophenotypic features of the initial punch biopsy. (a) Langerhans cells highlighted by CD1a (IHC CD1a 40×). (b) and (c) Langerhans cells showing perifollicular cellular infiltrate. Note spared intact epidermis and no notable eosinophils were present (H&E 40× and H&E 200×).

Immunophenotypic features of the follow-up punch biopsy. (a) Langerhans cells highlighted by CD1a (IHC CD1a 40×). (b) Langerhans cells showing diffuse cellular infiltrate extending to fascia. Note epidermal ulceration. (c) High power view showing Langerhans cells admixed with abundant eosinophils (H&E 200×).
Laboratory and imaging investigations following the recommendations of the Histiocyte Society confirmed the diagnosis (for details, see LCH in Table 1). Infectious serologies, including a TORCH screen, were negative. Complete blood count, liver transaminases, electrolytes, bilirubin, urea, and creatinine were all within normal limits. Skeletal survey, chest radiograph and abdominal and head ultrasounds of our patient revealed no evidence of systemic involvement. Our patient was reassessed by both pediatric dermatology and pediatric oncology at 2 weeks of age, and then regularly every 3 months until the age of 2. All LCH lesions had resolved by 18 months of age. He continues to be followed every 6 months and has no evidence of internal organ involvement on repeat bloodwork or imaging.
Source: Adapted from American Academy of Pediatrics, 12 see Table 2.
ALT: alanine transaminase; TSH: thyroid-stimulating hormone measurement; CT: computed tomography; CBC: complete blood count, including hemoglobin, white blood cell, differential count, and platelet count; CNS: central nervous system; CSF: cerebrospinal fluid; VDRL: venereal disease research laboratory test; VZV: varicella-zoster virus; IgG: immunoglobulin G; PCR: polymerase chain reaction; HSV: herpes simplex virus; WBC: white blood cell count; IgA: immunoglobulin A; IgM: immunoglobulin M; DFA: direct fluorescent antibody; APTT/PTT: activated partial thromboplastin time/partial thromboplastin time; AST: aspartate transaminase; γGT: gamma-glutamyltransferase; INR/PT: international normalized ratio/prothrombin time; ESR: erythrocyte sedimentation rate.
Discussion
Blueberry muffin syndrome refers to red-to-violaceous indurated macules, papules, or nodules present within 2 days of life. 9 The differential diagnosis is extensive and includes infections, cancers, and hematologic disease. Distinguishing clinical and laboratory features of potential causes are summarized from up-to-date publications in Table 1.10–20 In all cases, these unique lesions occur due to the presence of extramedullary erythropoiesis in the dermis.
In our case, the diagnosis of LCH was confirmed by histopathology and exclusion of other causes. The first biopsy revealed a sparse, unusual pattern with an isolated focus of deep perifollicular infiltrate consisting exclusively of CD1a-positive Langerhans cells without the characteristic eosinophils or epidermotropic infiltrate. The histopathology of LCH can be variable, but typically consists of a heavy infiltrate of Langerhans cells with reniform nuclei invading the basal layer of the epidermis with accompanying lymphocytes and eosinophils.2,21 Given our strong suspicion for LCH, a second biopsy of a more mature lesion was pursued and clinched the diagnosis. This case demonstrates the importance of clinicopathologic correlation and repeat biopsy when the two do not fit, as well as the importance of obtaining a mature lesion for biopsy to ensure typical histopathology.
LCH is believed to have an inflammatory and/or neoplastic etiology, although the mechanism of tissue infiltration remains unknown. 1 Interestingly, the first biopsy in our case revealed Langerhans cells clustered around the base of a hair follicle which suggests that perifollicular infiltration of the clonal cells may occur early in the disease. One previous case report noted a similar perifollicular infiltration of Langerhans cells on histopathology. 22 Normal resident Langerhans cells of the epidermis may not be the origin of LCH, instead LCH may originate from deep periadnexal precursors, highlighting that the classic epidermotropism associated with LCH is a progressive infiltration that becomes established only in well-developed lesions. How the infiltrate expands from perifollicular to densely dermal with epidermotropism remains to be elucidated and warrants further investigation. This possible early perifollicular histology of LCH is important for pathologists to be aware of in order to avoid missed diagnoses when faced with a biopsy of new LCH lesions. LCH can be single site (monostotic bone involvement, isolated skin involvement, and solitary lymph node involvement) or multiple site disease (polyostotic bone involvement, multifocal bone disease affecting two or more different bones, and multiple lymph involvement). 23 Our patient has single site histiocytosis, historically called “congenital self-healing reticulohistiocytosis,” characterized by skin lesions that rapidly involute and eventually self-resolve, typically over a number of weeks to months. 2 Distinguishing self-limited cutaneous disease from MSD is imperative to direct management and appropriately counsel patients’ families. Unfortunately, the initial presentations of both diseases can be clinically and histopathologically indistinguishable, and there is little available evidence to definitively conclude that limited cutaneous involvement at presentation will not evolve into MSD. Negative imaging at diagnosis does not rule out the possibility of systemic involvement at a later time. Features associated with a self-healing, limited cutaneous form of this disease include early disease onset of isolated skin involvement, quick resolution of skin lesions, and lack of BRAF mutation. 24
Footnotes
Authors’ note
The views expressed in the submitted article are that of the authors and not an official position of the institution.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from the patient’s parent for the use of non-identifying photos of his lesions for academic purposes.

