Abstract
The sequential occurrence of Langerhans cell histiocytosis and acute leukemia in only one individual has been reported previously; however, it is rarely observed that Langerhans cell histiocytosis can transform into acute lymphoblastic leukemia, and the underlying mechanisms remain unclear. In this report, we have analyzed a case of acute lymphoblastic leukemia converted from Langerhans cell histiocytosis using high-throughput sequencing method, and found that mitogen-activated protein kinase gene mutation, which can act as a marker for poor prognosis, might be involved in disease transformation. This is the first description about acute lymphoblastic leukemia B-cell type after Langerhans cell histiocytosis diagnosis and therapy in China.
Keywords
Introduction
Langerhans cell histiocytosis (LCH) and acute leukemia are two independent diseases commonly observed among children. A number of previous studies have reported that T-cell-type acute lymphocytic leukemia (T-ALL) can subsequently develop into LCH although the incidence rate is very low,1,2 and the mechanism might be related to the RAS (RAS proto-oncogene, GTPase) mutation and homozygous deletion of CDKN2A (cyclin-dependent kinase inhibitor 2A). 3 However, it is even rarer that LCH can potentially develop into ALL. LCH possesses the characteristics of both inflammatory disease and malignancies, and the latest classification has refined the disease as inflammatory myeloid neoplasm, 4 which indicates the possibility of malignant change of Langerhans cells. In addition, previously, only 27 such cases have been reported, and the development of lymphomas, solid tumors, as well as other hematological malignancies have been associated with the application of chemotherapy for LCH management, although most patients primarily develop AML after LCH treatment. 1 The exact mechanism of the disease origin is still unclear, whereas chemotherapeutic drugs, such as VP16, have been speculated to be the main reason behind evolution of the secondary malignancies. However, exact mechanisms besides the involvement of chemo drugs remain unclear and need further investigation. We encountered such a special case during our clinical work, in which LCH was found to have transformed into B-ALL using high-throughput sequencing method, and we thus aimed in this study to find its possible pathogenic gene and transformation mechanism.
Case presentation
A 3-year-old girl was hospitalized for pain in her right leg. She had fever and bone pain, active bone lesion of the right femur was detected by radionuclide bone scan, magnetic resonance imaging (MRI) of the abdomen and pituitary was normal, and she was then diagnosed as LCH by biopsy of the right femur (Figure 1(a)–(c)). She received treatment with the following drugs, vinblastine, prednisone, as well as 6-mercaptopurine, and her clinical symptoms disappeared after the treatment. One year later, her symptoms such as bone pain recurred again, along with fever and pallor. Complete blood count indicated anemia, and bone marrow (BM) smear consisted of both lymphoblastic and langerhans cells, with the percentage being 46% and 35%, respectively (Figure 2). Thereafter, acute B lymphoblastic leukemia was diagnosed by flow cytometry subsequently (positive for CD19, CD10, CD9, CD34, CD38, HLADR, CD22, CD123, CD20, TDT, and cytoCD19a). She received childhood GD2008 protocol (a treatment protocol for childhood acute lymphoblastic leukemia in the Guangdong Province of China) and complete remission when the induction treatment was completed. However, she has suffered a relapse 2 years later.

Biopsy of the right femur: (a) HE staining of the biopsy, massive distribution of the Langerhans cells with eosinophil and lymphocyte infiltration. (b) Immunohistochemistry showed the Langerhans cells were positive for CD1a. (c) Immunohistochemistry showed the Langerhans cells were positive for Langerin.
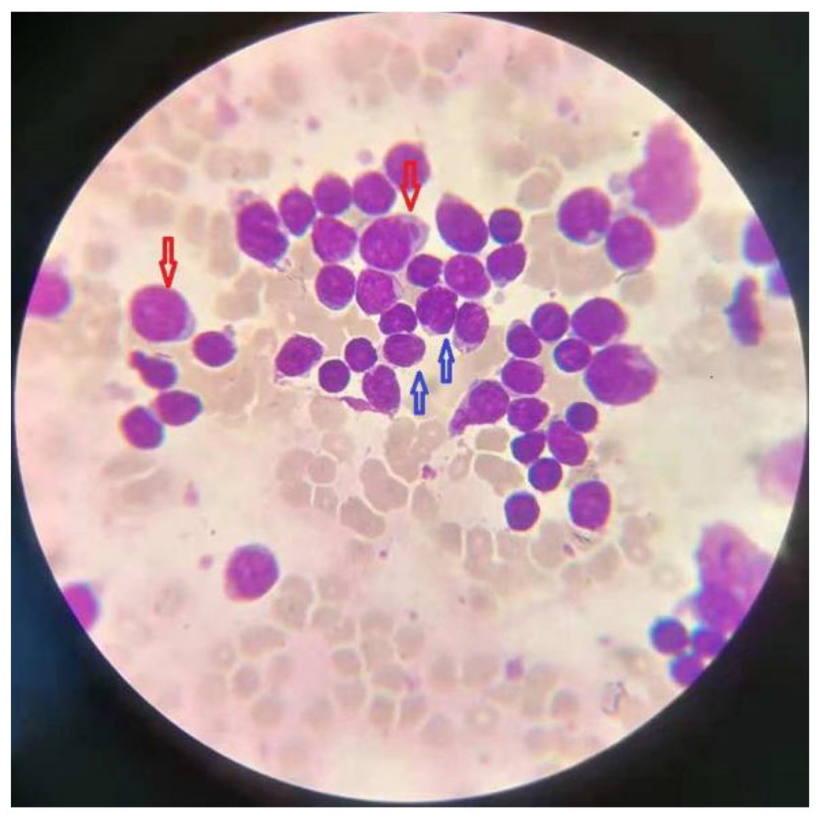
Bone marrow smears of the disease. The cell was observed by Wright staining and oil lens of OLYMPUS microscope (100×). Langerhans cell histiocytosis concurrent acute lymphoblastic leukemia: red arrow indicates Langerhans cells and blue arrow indicates lymphoblastic cells.
Genomic analysis was approved by the ethics board of the Guangdong Provincial People’s Hospital, and informed consent was obtained from the patient’s guardians. We conducted whole exome sequencing (WES) of the sample; DNA was then extracted from the obtained BM sample and whole exome capture was accomplished using SureSelect Human All Exon kit V5 (Agilent Technology, USA). The captured targets were subjected to sequencing with HiSeq 2000 (Illumina, USA). We found gene mutation MAPK (T > G; MAP3K15: NM_001001671: exon22: c.A3085C: p.N1029H) present in the DNA samples obtained from both LCH and ALL stages.
Discussion
LCH is one of the common histiocytosis in children and is often characterized by inflammatory lesions containing abundant CD1a + CD207 + histiocytes. The clinical symptoms of LCH are heterogeneous and often involve skin, bone, liver, spleen as well as BM. A number of previous reports have suggested the occurrence of multi-organ lesions that can potentially predict poor prognosis. 5 The characteristics of LCH include both inflammation and malignancy; hence, it is called inflammatory myeloid neoplasm, 4 which indicates the common clone of Langerhans cells and leukemia blasts. Although the chance remains very low; however, the malignancies that developed after the onset of LCH have been repeatedly reported, and acute leukemia is the most common malignancy associated with LCH. 1 However, there are still very few studies to establish the underlying causes behind the transformation mechanism of this disease.
We collected the BM sample of a patient who was diagnosed as B-ALL transformed from LCH. We found that MAPK might be possibly involved in the process of the disease transformation based on the research of the gene alterations by using the whole exons sequencing analysis. MAPK (mitogen-activated protein kinase) is a member of the MAP kinase family. MAP kinases, also known as extracellular signal-regulated kinases (ERKs), act as an integration point for multiple biochemical signals and have been reported to be involved in a wide variety of cellular processes, such as proliferation, differentiation, transcription regulation, and development. 6
The genetic mechanism about LCH is still unclear, and the frequent gene mutations found are that of MAPK and BRAF. 7 For instance, Choiet al. 8 found changes in TP53 gene might have contributed to the malignant features of Langerhans cell sarcoma (LCS). Taki et al. 9 found T-ALL and LCH cells were derived from a common precursor with TCR rearrangement and a single NOTCH1 mutation. Collin and Milne 10 reported that distinct TGF-β signaling pathways were involved in the development of Langerhans cells and maintenance of their homeostasis. In addition, previous studies have reported that RAS mutation and homozygous deletion of CDKN2A were detected in ALL cases, which subsequently developed into LCH. 3 However, we did not find these reported changes, and this might be because the conversion mechanism of ALL developed to LCH can be different from that of LCH developed to ALL. Interestingly, mutations in MAPK gene have been reported to be related to both LCH and leukemia,11–13 so we speculated that MAPK might serve as a potential biomarker for the disease conversion. However, further investigations are needed because there is no evidence or studies about the relationship of those various gene mutations and disease. In our case, the girl suffered from B-ALL following LCH without being administered chemo drug VP16, which has never been reported in China so far. We speculated that chemotherapy might be the relevant factor of disease occurrence; in addition, common precursors of Langerhans cells as well as leukemia blasts might also play an important role in the disease progression; however, B-ALL type is not the common type of transforming leukemia, and therefore, the exact mechanisms needs further detailed analysis.
Conclusion
In this peculiar case, the girl suffered from B-ALL following LCH without being administered chemo drug VP16, which has never been reported in China so far, and WES analysis indicated that MAPK gene might be a potential biomarker for the disease conversion.
Footnotes
Acknowledgements
The authors are very grateful to the patient’s family for their support of this study.
Contributors’ statements
J.Z. designed the study, drafted the initial manuscript, and approved the final manuscript as submitted. S.Z. carried out the initial analyses, revised the manuscript, and approved the final manuscript as submitted. B.L. carried out the biopsy results and provided the biopsy pictures and approved the final manuscript as submitted. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval to report this case was obtained from * Ethics Committee Board of the Guangdong Provincial People’s Hospital (Guangdong General Hospital) and Guangdong Academy of Medical Science (approval no. GDREC2018389H[R1]).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by the Guangdong Basic and Applied Basic Research Foundation (grant No. 2018A030313524).
Informed consent
Written informed consent was obtained from the patient’s guardians (parents) for the publication of this case report.
