Abstract
Basal cell carcinoma metastases are rare. We report a case of an 83 year-old male with recurrent cutaneous infiltrative basal cell carcinoma on the right vertex with tumor aggregates of infiltrative basal cell carcinoma in the lymphatic vessels of the superficial dermis strongly suspected on pathology slide evaluation during Mohs micrographic surgery. D2-40 immunohistochemical stain allows the detection of lymphatic invasion by highlighting endothelial cells. Further histologic review and D2-40 stains performed on the paraffin-embedded sections did not reveal an endothelial cell lining in favor of lymphatic invasion. What was identified as lymphatic invasion was the result of a peculiar basal cell carcinoma stroma. Correct identification of basal cell carcinoma stroma as a mimicker of lymphatic invasion would prevent potentially invasive investigation as well as associated costs. This is of great importance as the prognostic and therapeutic implications can be of significant value for the patient.
Introduction
Basal cell carcinoma (BCC) metastases are very uncommon, with estimated risk ranging from 0.0028% to 0.005% of tumors. 1 Metastasis can occur via hematologic or subcutaneous spread; however, lymphatic spread accounts for 70% of cases. 1 Lymphatic invasion is a feature considered by some to be associated with increased risk of metastasis. 2 We report a case of a recurrent cutaneous infiltrative BCC on the right vertex with lymphatic invasion strongly suspected during Mohs micrographic surgery (MMS). Further histologic review and immunohistochemical stains did not show lymphatic invasion and revealed BCC tumor stroma to be a mimicker of lymphatic invasion. To our knowledge, no histologic mimickers of lymphatic invasion have been identified.
Case report
An 83-year-old immunocompetent male presented to the dermatology clinic with a 6-month history of a pearly pink plaque on his right vertex measuring 1.5 × 1.0 cm2. The lesion appeared at the site of a previous nodular BCC treated with MMS by another surgeon 5 years prior and reconstructed using a full-thickness skin graft. The patient had no systemic complaints and physical examination did not reveal lymphadenopathy.
Considering the recurrent nature and the location of the lesion, a second MMS was indicated. Five stages were required to reach pathological clearance. Infiltrative BCC was the main pathological subtype identified in the MMS stages. Stage 2 pathology slide evaluation revealed probable tumor aggregates of infiltrative BCC in the lymphatic vessels of the superficial dermis (Figure 1).

Presumed infiltrative BCC tumor aggregates in the lymphatic vessels of the superficial dermis (hematoxylin and eosin, original magnification, 10×).
Further histologic review and D2-40 immunohistochemical stains performed on the paraffin-embedded sections did not reveal an endothelial cell lining in favor of lymphatic invasion (Figure 2). In addition, there was no evidence of vascular or perineural invasion.
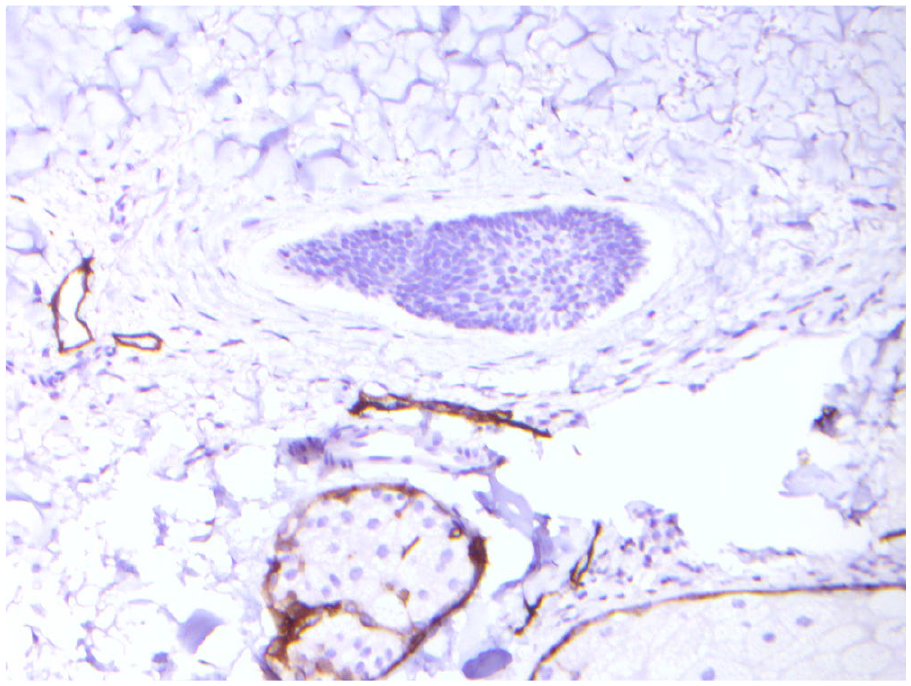
Immunohistochemical stains performed on the specimen did not reveal an endothelial cell lining compatible with lymphatic invasion revealing BCC stroma as a mimicker of lymphatic invasion (D2-40, original magnification, 20×).
The Mohs defect measuring 8.0 × 5.0 cm2 was reconstructed with a split-thickness skin graft. Considering the size, pathological subtype and recurrent nature of the BCC, the patient was referred to a radiation oncologist for adjuvant treatment. Further imaging of his head and neck with computed tomography (CT) scan did not show any bone invasion or cervical adenopathy.
Discussion
This is a rare case of tumor stroma as a mimicker of lymphatic invasion. Infiltrative BCCs are usually composed of small islands of basaloid cells surrounded by a myxoid and mucinous stroma admixed with inflammatory cells. In our case, the basaloid cells were surrounded by a cleft larger than usual, a phenomenon possibly enhanced by the frozen section artifact. Clefting between the stroma and the tumor is often less distinctive in the infiltrative subtype. 3 Myxoid or mucinous stroma was not present in our case, but there was a concentric condensation of collagen mimicking a vessel wall. D2-40 immunohistochemical stain allows detection of lymphatic invasion by highlighting endothelial cells. 4 In our case, the D2-40 immunohistochemical stain was negative around the basaloid cells. Therefore, what was identified as lymphatic invasion was the result of a peculiar BCC stroma. Other features particular to the stroma of infiltrative BCC include thicker stroma and smaller tumor radii with a correspondingly smaller tumor-to-stroma ratio. 5
The clinical relevance and therapeutic implication of lymphatic invasion are not well documented due to its rarity. 2 In addition, the likelihood of lymph node metastasis associated with lymphatic invasion remains unknown. 2 Sentinel node biopsy and positron emission tomography (PET) scan are often used to detect metastatic disease; however, their utility is not well documented because of low incidence of metastatic BCCs. 2 Milam et al. 6 recommend sentinel lymph node biopsy and/or PET scan, with high-risk BCCs demonstrating vascular or lymphatic invasion. The decision to pursue further testing is based on clinical judgment and on the risk of recurrence or metastasis. 6 Correct identification of BCC stroma as a mimicker of lymphatic invasion would therefore prevent additional potentially invasive investigation as well as associated costs.
As this case demonstrates, the stroma of infiltrative BCC may mimic lymphatic invasion. The recognition of this mimicker is of great importance as the prognostic and therapeutic implications can be of significant value for the patient.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Patient consent was received to publish this case report.
