Abstract
Anti-neutrophil cytoplasmic antibody–associated crescentic glomerulonephritis commonly presents as a renal emergency requiring timely care to reduce risk of kidney failure and related mortality. Milder forms of disease are less common, and it requires a high index of suspicion to detect them. Herein, we report a case of focal proliferative and necrotizing glomerulonephritis in a 21-year-old patient presenting with a new-onset microscopic haematuria and minimal proteinuria associated with normal renal function.
Keywords
Background
Globally, anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis is the most common cause of rapidly progressive glomerulonephritis (RPGN). Renal involvement in ANCA-associated vasculitis represents the most aggressive form of acute glomerulonephritis with the majority of patients presenting with an acute nephritic picture and rapid kidney function loss.
ANCA-associated vasculitis may be isolated to the glomerulus (renal-limited) or can be multi-systemic (occurrence in association with a systemic disease, like granulomatosis with polyangiitis or microscopic polyangiitis), and generally occurs in older adults with a peak incidence in the fourth to sixth decades of life.
We report a case of ANCA-associated glomerulonephritis in a young patient presenting with mild urinary abnormalities and normal function leveraging a standard framework for reporting of case reports. 1
Case presentation
This is a 21-year-old Caucasian male with a history of recurrent episodes of generalized joint pains (polyarthralgia) over a 2-year period before the current presentation. These recurred multiple times and resolved spontaneously with no specific rheumatologic diagnosis (he was HLA B27 negative). The only pertinent findings of note on physical examination were diffuse cervical lymphadenopathy, inflamed tonsils and splenomegaly.
Medical history
The patient had been experiencing polyarthritis for the previous 2 years. These arthralgias and arthritis recurred multiple times, however would resolve spontaneously without any treatment. No specific diagnosis made under Rheumatology Care. Due to increased dysphagia and potential airway compromise, he underwent a tonsillectomy at the beginning of August 2015. At the same time, he was also noted to have diffuse lymphadenopathy as well as splenomegaly. Computed tomography (CT) scan showed bilateral tonsillar hypertrophy which was dealt with surgically. For anaemia, he had upper and lower gastrointestinal scopes which showed no lesions, a subsequent capsule endoscopy showed ulceration in terminal ileum. This was followed by a double balloon push enteroscopy for tissue diagnosis.
Family history
He lives with his parents and played competitive Ice Hockey.
Social history
He was a non-smoker, occasional alcohol user and not a recreational drug user. He worked mainly at construction sites.
Medications
Escitalopram 20 mg daily May and June 2014,
Pregabalin 50 mg three times daily from July to December 2014,
Duloxetine 30 mg daily from December 2014 to June 2015.
Investigations
Patient underwent tonsillectomy for dysphagia and potential airway compromise in the community before his referral to nephrology service. The tonsillar tissue did not show any evidence of lymphoproliferative disorder on histopathology. Further workup showed a positive cANCA with PR3 positivity (a titre of 881 MFU (Normal < 120 MFU)). Chest x-ray showed vague pulmonary infiltrates, and the patient tested negative for tuberculosis (Figure 1). CT chest showed pulmonary parenchymal findings in keeping with an infectious process (Figure 1).
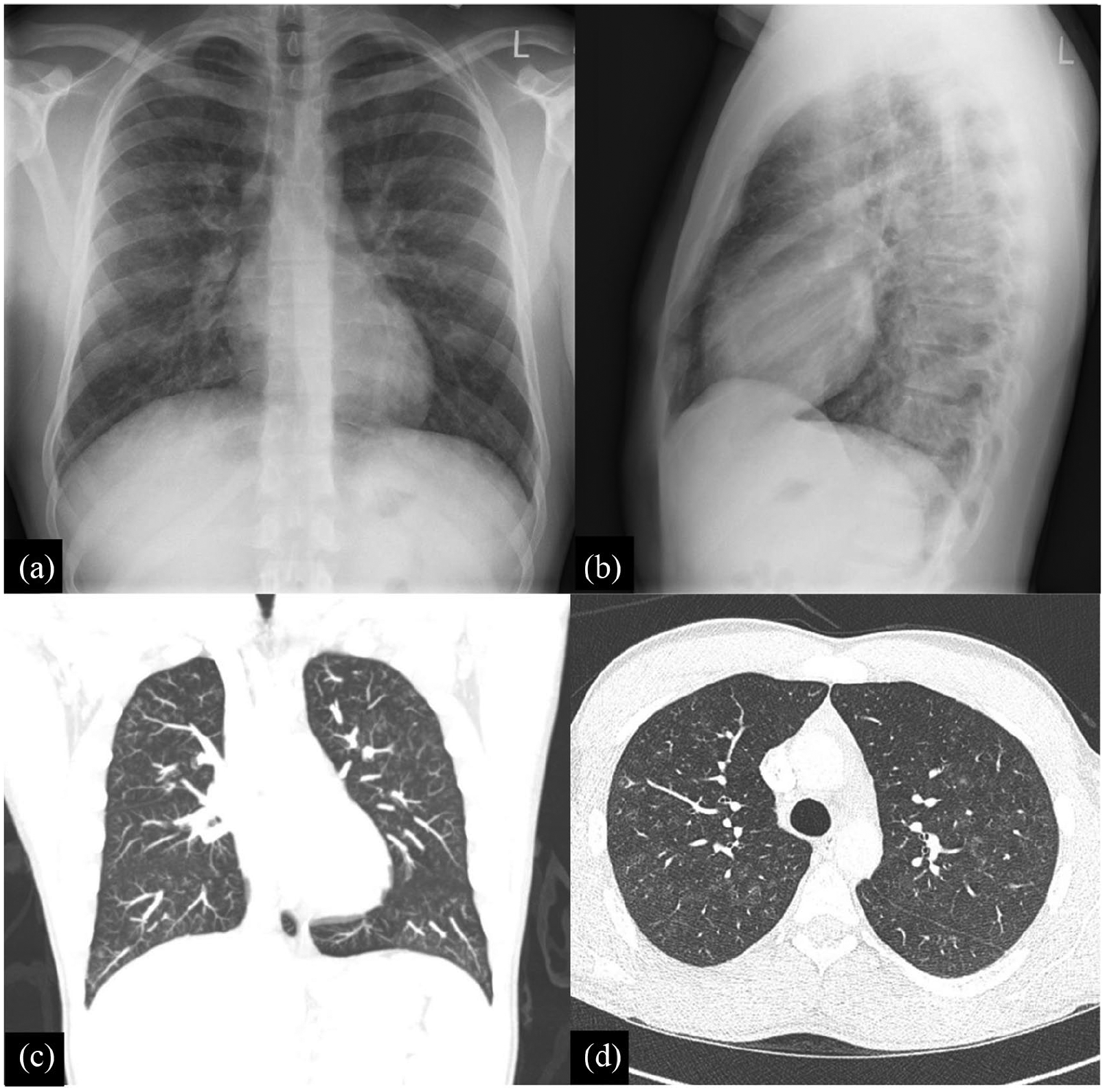
Imaging studies: (a) chest x-ray anteroposterior view, (b) chest x-ray lateral view, (c) chest CT scan coronal section and (d) chest CT scan sagittal section.
He had evidence of significant anaemia and underwent an endoscopy for gastrointestinal symptoms and anaemia (Haemoglobin 63 g/L (Normal 135–175 g/L)) that showed only ulceration in terminal ileum. The biopsy of the intestinal tissue did not show any evidence of vasculitis.
The kidney function was normal at presentation and remained stable (within the normal range) throughout the follow-up period (Figure 2). At 2 months of follow-up, he was noted to have new onset of microscopic haematuria and low-grade proteinuria. Urinalysis showed 1+ haemoglobin and trace protein, and urine protein: creatinine ratio of 36.9 mg/mmol (Normal < 13 mg/mmol) (Figure 2). Urine Microscopy for sediment showed dysmorphic red blood cells. A urinalysis completed 3 months prior to presentation was completely bland (we could not trace any urinalysis after this till Nephrology was consulted). Relevant serology results were all negative except for the ANCA PR3 positivity. The serology workup completed was as follows, and were all negative or within a normal range:
Antinuclear antibody (ANA)
Rheumatoid factor <10 (20 KU/L)
Anti-cyclic citrullinated peptide (anti-CCP) < 8 (0–16 U/ml)
Anti-double stranded DNA (anti-dsDNA) 14(<120 MFU)
Extractable nuclear antigens (ENA)
Hepatitis B surface antigen (HBsAG)
Hepatitis B surface antibody (HBsAb) 4.98 IU/L
Hepatitis C antibody
Parvovirus IgM
Serum complements [C3 1.1.(0.80–2.1 g/L) and C4 0.38(0 > 15–0.50 g/L)]
Immunoglobulins: IgG 12.6 (6.94–16.18 g/L) IgA 2.51 (0.70–4 g/L) IgM 0.87 (0.60–3.00 g/L)

Laboratory changes in renal function.
Based on the previous features of a systemic illness and new findings on urinalysis and urine microscopy, the patient underwent a kidney biopsy, and the tissue was analysed and reported by our pathology service (Figure 3). The diagnosis was in keeping with ANCA-associated vasculitis (focal proliferative and necrotizing glomerulonephritis). In summary, there were 11 glomeruli present in representative sections, and two contained segmental proliferative foci. The remaining glomeruli showed no conspicuous changes on light microscopy. In the plastic embedded material prepared for electron microscopy, the light microscopy revealed seven additional glomeruli of which one showed segmental sclerosis and another showed segmental proliferative focus with crescentic formation. There was mild interstitial edema and patchy mild interstitial fibrosis and tubular atrophy without significant interstitial inflammation. In consultation with our pathologists, the lesion was classified as focal based on Berden’s classification. 2

Kidney biopsy: light microscopy – (a) PAS staining and (b) H&E – the glomerulus exhibits a cellular crescent with focal breach in the Bowman’s capsule; no fibrinoid necrosis is seen; there were 11 glomeruli in the representative section, two contain segmental proliferative foci and remaining glomeruli show no conspicuous changes; mild intestinal edema and patchy mild interstitial fibrosis and tubular atrophy without segmental interstitial fibrosis; and vessels show no conspicuous changes. (c) Electron microscopy (EM) – the EM photomicrograph shows mild effacement of podocyte foot processes and a near break in the capillary basement membrane which is the cause of crescent; no immune complex deposition; and peritubular capillaries have normal structure.
We did evaluate the case using the Birmingham Vasculitis Score (BVAS), and overall score was eight. 3
Differential diagnosis
Other forms of acute glomerulonephritis were excluded based on relevant biochemical and pathologic criteria. Differential considered were IgA nephritis (new-onset haematuria and proteinuria in the setting of tonsillar inflammation), post-infectious GN was a consideration and there was no evidence of infection in the tonsillar biopsy and the complement C3 was normal. Unfortunately, immunofluorescence was not performed due to paucity of tissue. There was no mesangial expansion and/or hypercellularity to suggest IgA nephritis on the light microscopy. Furthermore, the fact that the serum complements were normal and the electron microscopy did not show any sub-epithelial deposits argue against an immune complex nephritis.
Also, patient’s blood pressure and renal function remained stable as well pointing against this type of diagnosis.
Treatment
He received the following treatments:
(a) Induction therapy with steroids as Methyl-Prednisolone 500 mg daily for 3 days, followed by Oral Prednisone 60 mg daily for 4 weeks, tapered to 50 mg daily, followed by a decrement of 10 mg every week to 5 mg daily by the third month of treatment as per our local protocol (tapering doses over a period of 6 months), and rituximab (1000 mg ×2 dose 2 weeks apart) on day 1 and day 15.
(b) Maintenance therapy included Azathioprine 2 mg/kg for 18 months.
Outcome and follow-up
The patient made a full recovery following the induction immunosuppressive treatment received. Two years later on follow-up, there has been no recurrence of symptoms or urine abnormalities and the patient remains well.
Discussion
The occurrence of microscopic haematuria is an important sign of an underlying renal parenchymal disease. Concomitant proteinuria is also common in glomerular causes of haematuria. Although serological testing for ANCA has proven value in the setting of RPGN, histopathological diagnosis remains the gold standard criterion for a specific diagnosis. 4 The advantage of kidney biopsy on serology is the ability to detect crescents (morphological diagnosis of an extra-capillary proliferation of cells), the hallmark of all RPGN. Renal disease can also present as subacute or chronic nephritis. 5 Renal involvement occurs in 18% of patients initially and later over 77% patients go onto develop glomerulonephritis. 6
This kind of presentation involving multi-organ systems obviously raised concerns about a vasculitic process. The cANCA and anti-proteinase (PR3) positivity correlated with renal and lung involvement suggestive of an ANCA-associated glomerulonephritis and vasculitis. 7 The new onset of persistent haematuria and proteinuria was reflective of a renal pathology which was then proven by a kidney biopsy. This picture is more in keeping with other forms of glomerulonephritis’, such as lupus nephritis where 30% of patients would have a diagnosis of stage III or IV lupus nephritis with relatively preserved renal function. However, this unique presentation has not been extensively described in ANCA-associated vasculitis setting, where a biopsy is usually performed in an RPGN like clinical setting. 8 Moreover, it may not be fully appreciated that even in RPGN haematuria and proteinuria appear and persist with normal serum creatinine well before the onset of renal impairment. Indeed 50% of renal function may be lost before serum creatinine is elevated and therefore a relatively normal serum creatinine does not exclude RPGN. Findings of proteinuria and haematuria are not specific for RPGN but are more sensitive than serum creatinine in early stages of the disease 9 This comment is emphasized from the point of view of General Practitioners who may not pay much attention to urinalysis if the serum creatinine is normal. Also, renal involvement in early evolution of ANCA-related vasculitis is rather sparse at 18% 6 and appropriate attention may not be given to this aspect.
The key message here is that renal biopsy should be considered in patients with mild urinary abnormalities when a diagnosis of vasculitis is sought even when renal function (serum creatinine) is normal. In addition, a biopsy of extra renal tissue has a rather low yield for a definitive diagnosis of ANCA-related vasculitis, and in many cases, it is necessary to repeat biopsy and make repeated attempts to establish the diagnosis. 10
Learning points
ANCA-associated vasculitis may present in the context of completely normal renal function (normal serum creatinine and estimated glomerular filtration rate).
Practitioners should have high index of suspicion for glomerulonephritis in patients with asymptomatic urinary abnormalities and systemic features.
In young individuals, isolated microscopic haematuria in the setting of multisystem disease may signal RPGN.
Footnotes
Acknowledgements
I thank the pathologists Drs Mudassar Hussain and Kim Solez for help with the pathology slides, radiologist Dr Sean Winters for help with the imaging slides, and Dr Mohamad Osman for the write up and coordination.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
