Abstract
We describe here the clinical outcome of four women with epilepsy with eyelid myoclonia (aged 21–53 years). All patients had an uneventful early history, normal physical growth and appearance and no comorbid sensory or motor disability and normal brain magnetic resonance imaging finding. Two women were moderately and one mildly intellectually disabled and one showed a low-average intelligence. The overall well-being of the patients was hampered by psychiatric or various somatic comorbidities and related psychosocial problems. The three women with an intellectual disability had been treated with narrow-spectrum antiepileptic drugs and one also with vigabatrin during childhood and adolescence. The patient with a low-average intelligence had been on broad-spectrum antiepileptic medication (i.e. valproate and ethosuximide) since the epilepsy diagnosis but she has had compliance problems. Based on these cases, the cognitive deficits in patients with epilepsy with eyelid myoclonia may occur more commonly than what has been thought hitherto. We discuss the role of narrow-spectrum antiepileptic drugs as a contributing factor to poor seizure control and an impaired intelligence.
Keywords
Introduction
Epilepsy with eyelid myoclonias (EEM; Jeavons syndrome) was first described as a separate form of photosensitive epilepsy in 1977. 1 EEM is a genetically determined syndrome with a female predominance and it is characterized by eyelid myoclonia, eye closure–induced electroencephalogram (EEG) paroxysms or absence seizures and photosensitivity. The seizure onset usually occurs between ages 2 and 14. Many patients have a positive family history with epilepsy. The patients’ early history, as well as appearance and neurological examination, is usually normal.2,3
Childhood-onset epilepsy syndromes, in general, show highly variable clinical outcome with regard to seizure remission and patient’s cognitive development and adaptive skills. 4 Neurocognition in EEM patients is considered normal but is in fact poorly known. To our knowledge, there are only two cognition-related studies. Fournier-Goodnight et al. 5 have defined neurocognitive functioning in a detailed manner in six young patients, whose global IQ ranged from low-average to borderline impaired. Capovilla et al. 6 have suggested that there may be an EEM subtype that is associated with an impaired cognition.
Here, we describe the clinical outcome of four women with EEM. All patients were followed by the first author (M.A.) and occasionally by other specialists in an epilepsy clinic at a public central hospital.
Each study subject had a decisional capacity to provide an informed consent.
Cases
All four women had an uneventful early history, normal physical growth and appearance, but no motor or sensory disability. Brain magnetic resonance imaging (MRI) showed normal finding in patients 2, 3 and 4; the oldest woman, patient 1, had a slight cortical atrophy. EEM was identified in adulthood on all four women. By reviewing their medical records and numerous EEG recordings retrospectively, we found that the diagnostic criteria for EEM had been reached already at the onset of seizures. Women had similar interictal EEG findings, that is, normal background but generalized 3–6 Hz spike/polyspike waves. Eye closure (fixation off sensitivity) and intermittent photic stimulation activated the EEG and often elicited eyelid myoclonias with or without absences. A typical EEG abnormality was facilitated also by hyperventilation in all but patient 4, who did not hyperventilate during registration. Gene panel of epilepsy including CHD2 epileptic encephalopathy was not done to any of the study subjects.
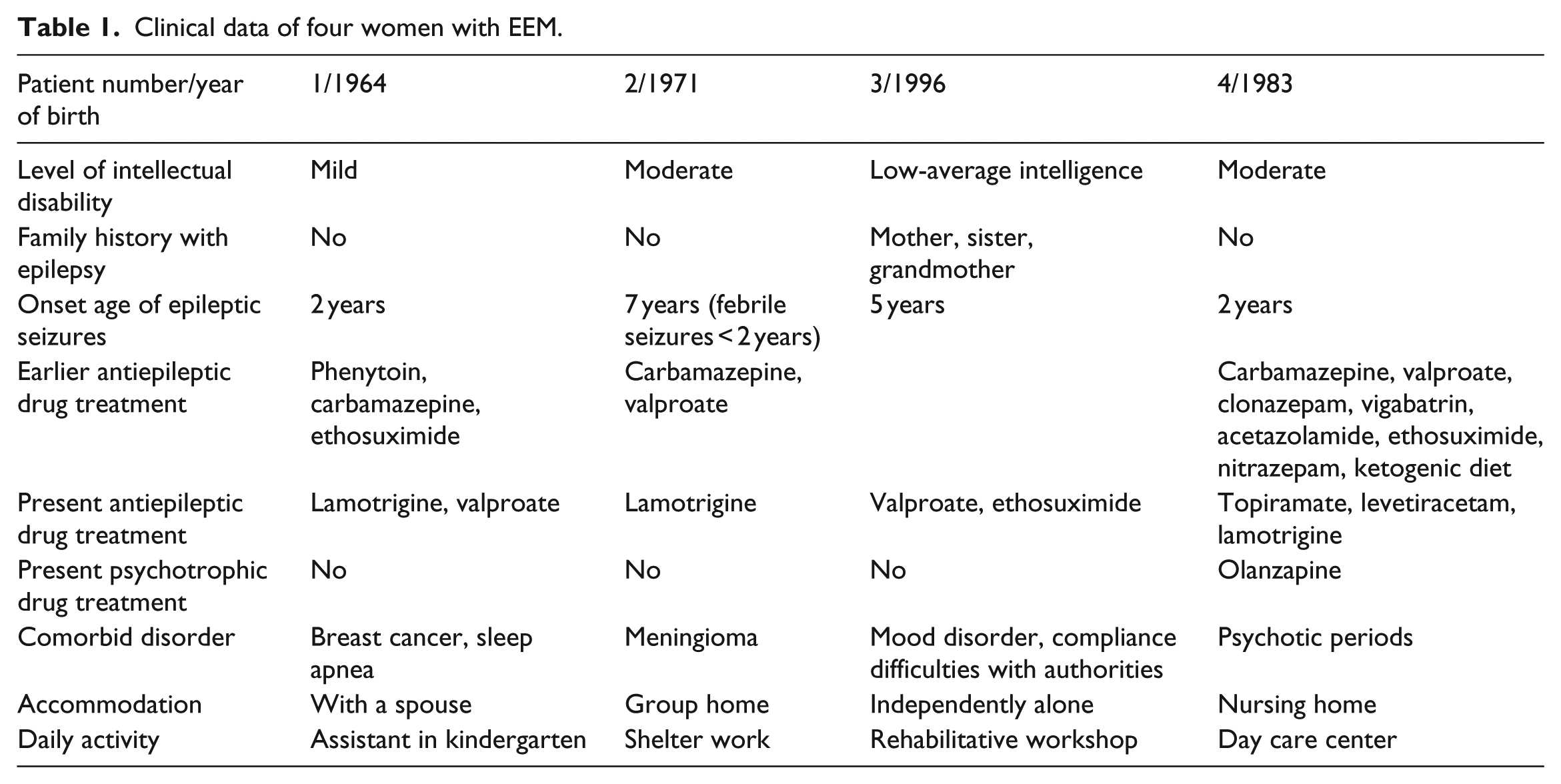
Clinical data including family history with epilepsy, onset age of epilepsy, earlier and present epilepsy drug medication and comorbidities of the study subjects are presented in the table.
Patient 1
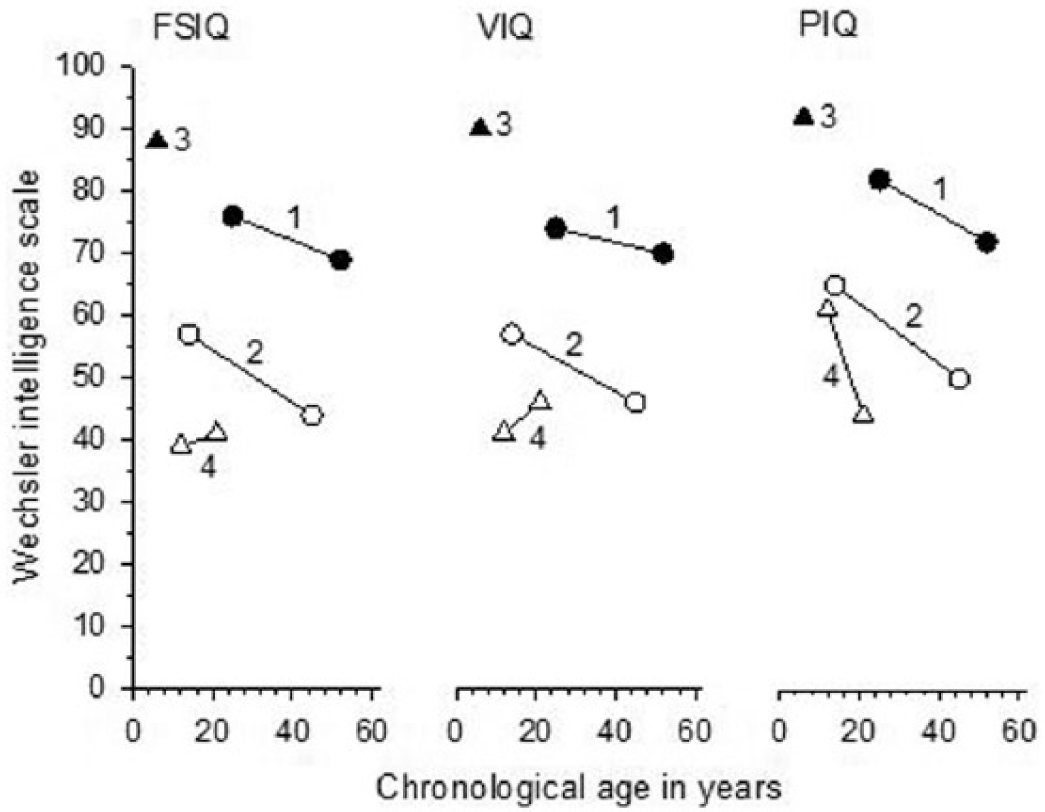
The initial diagnosis of patient 1 (a 54-year-old woman) was childhood absence epilepsy and later on epilepsy with myoclonic astatic seizures (Doose syndrome). During childhood and adolescence, she was treated with phenytoin and carbamazepine. She suffered one or two generalized seizures on a monthly basis but since the initiation of lamotrigine and identification of EEM at the age of 45, she has had only a single absence seizure per year (apart from daily eyelid myoclonias). Her intelligence test results done during childhood and adolescence were not available. At the age of 25, she showed subnormal intelligence (Wechsler Adult Intelligence Scale (WAIS), 7), and at the age of 53, a mild intellectual disability (ID; WAIS-III, 8) (Figure 1). In adulthood, she has suffered from somatic comorbidities (Table 1) but has good social skills and she lives with a spouse and works as an assistant in kindergarten.
Clinical data of four women with EEM.
Patient 2
Patient 2 (a 46-year-old woman) had experienced febrile seizures before epilepsy diagnosis was made at the age of 7. Her initial diagnosis, before identifying EEM at the age of 43, was childhood absence epilepsy. During childhood and adolescence, she was treated with carbamazepine with poor response. After the initiation of valproate and later of lamotrigine (because of obesity caused by valproate), at the age of 35 she had only eyelid myoclonias until she reached the age of 45 and experienced a prolonged generalized seizure. Soon after this, a brain scanning revealed a meningioma necessitating a brain surgery. Mild ID (Wechsler Intelligence Scale for Children (WISC), 9) was diagnosed at the age of 14, and at the age of 46, she acted on a level of moderate ID (WAIS-III, 8) (Figure 1). She lives quite independently in a group facility and attends shelter work.
Patient 3
Patient 3 (a 21-year-old woman) has a positive family history of epilepsy (Table 1). Her initial diagnosis was childhood absence epilepsy before EEM was recognized at the age of 19. She has been on valproate and ethosuximide medication since the epilepsy diagnosis, but she suffers from daily eyelid myoclonias and occasionally from prolonged epileptic seizures due to an irregular use of antiepileptic medication. She has repeatedly refused a psychiatric assessment. At the age of 6, she showed a low-average intelligence (Wechsler Preschool and Primary Scale of Intelligence—Revised (WPPSI-R), 10) (Figure 1). She has passed the normal curriculum at comprehensive school. She lives alone and attends a rehabilitative workshop.
Patient 4
The initial diagnosis for patient 4 (a 34-year-old woman) was epilepsy with myoclonic astatic seizures (Doose syndrome). She has been unresponsive to ketogenic diet and to several antiepileptic drugs (Table 1) except to topiramate, after initiation of which (from the age of 19) she has been seizure free (except daily eyelid myoclonias). At the ages of 12 and 22, she showed moderate ID (WPPSI-R, 10; Wechsler Adult Intelligence Scale—Revised (WAIS-R), 11). She is in psychiatric follow-up because of her aggressive behavior and intermittent psychic symptoms and she lives in a 24-h nursing home. The clinical geneticist has examined her and ordered wide metabolic and molecular genetic analyses which did not reveal any specific etiology.
Global intellectual ability
The figure presents the full-scale, verbal and performance IQs of the four patients. In patients 1, 2 and 4, the performance IQ was somewhat better than the verbal IQ. They all had difficulties at precise understanding and naming and in higher order verbal reasoning. All had quite good perception of visual details but difficulties in visuomotor and/or visuoconstructive functions. The same three patients (1, 2 and 4) had problems in executive functions which have most likely affected their memory and their auditive working memory which turned out to be narrow. On the other hand, all three showed slow verbal and nonverbal learning but quite good long-term recalling. Patients 2 and 4 were found to have prominent problems in attention, especially in flexibility.
Discussion
To our knowledge, this is the only long-term study in which cognitive function has been followed in EEM patients. The intelligence is generally thought to be normal., ID was diagnosed in as many as three out of the four patients. The observed frequency seems high but the possibility of recruitment bias cannot be totally excluded as these four cases may not necessarily represent all EEM patients living in the district (with a population of 202,000).
Two groups have earlier evaluated retrospectively the cognition in patients with EEM. Fournier-Goodnight et al. 5 defined neurocognitive functioning using the same Wechsler test (as we) in six young patients but none of them can be considered intellectually disabled based on their performance on higher order tasks including nonverbal reasoning. The IQ figure of our youngest patient 3 (i.e. the only one without a confirmed ID diagnosis), who was assessed at the age of 6 was in line with the study patients of Fournier-Goodnight et al. In another retrospective study by Capovilla et al., 6 no details are given either of the used test methods or the age at which the diagnosis of ID was established, which makes a reliable comparison of the cognitive status between their 14 intellectually disabled cases and our patients impossible.
The underlying cause for an impaired cognition in our three oldest patients may have been their poor epilepsy control or unidentified CHD2 epileptic encephalopathy 12 apart from possible genetic susceptibility. These patients had been treated with narrow-spectrum antiepileptic medicines and one with vigabatrin in their childhood. The prescribed medication can also have contributed to the impaired cognition taking into account that carbamazepine and phenytoin are contraindicated in photosensitive epilepsies because they may aggravate various types of seizures and because vigabatrin is known to provoke absence and myoclonic seizures.13,14 Interestingly, patient 3 with the best cognition had been treated only with valproate and ethosuximide, that is, broad-spectrum anticonvulsants that are nowadays generally recommended for EEM. In this particular case, however, the problem was a poor adherence to the antiepileptic medication. Thus, it might be too speculative to claim that her better cognitive capacity compared to the three other cases could have been due to the right medication prescribed at the time when her epilepsy was initially diagnosed. In addition, the differential diagnostics may have been insufficient because gene panel of epilepsy was not carried out.
Pharmacotherapy should and is ideally based on an as accurate epilepsy diagnosis as possible, but the identification of epilepsy or epilepsy syndrome in a clinical realm is often time-consuming15,16 as also in these four cases. This leaves us with an open question: Would the cognitive outcome have been any better if the EEM diagnosis had not been delayed and if the patients had been treated according to the current guidelines from the beginning of their epilepsy diagnosis?
Conclusion
EEM does not belong to a group of epileptic encephalopathies in which epileptogenesis forms a causative or contributing factor for ID. Consequently, this epilepsy syndrome is not usually associated with ID. However, the three out of our four EEM patients turned out to be intellectually disabled. The right diagnosis was delayed in all cases and the initially prescribed narrow-spectrum antiepileptic drugs or vigabatrin could have impaired their epilepsy control (provided that none of the patients had CHD2 epileptic encephalopathy). All this may have had a negative impact on their cognition. These clinical observations emphasize the importance of the right epilepsy diagnosis and the initiation of an appropriate medication at as early stage as possible. Epilepsy gene panel that was unfortunately not available for our cases can be expected to help in choosing the right antiepileptic medicine in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patients for their anonymized information to be published in this article.
