Abstract
Eosinophilia, both peripheral and in cutaneous tissue, is not a typical finding in mycosis fungoides; in fact, when faced with a lymphoeosinophilic infiltrate, mycosis fungoides is often not part of initial differential considerations. However, eosinophilia has been described in certain subtypes of mycosis fungoides, namely, in folliculotropic mycosis fungoides. We describe three challenging cases of folliculotropic mycosis fungoides presenting with varied clinical morphologies and a dense lymphoeosinophilic infiltrate and/or severe hypereosinophilia that obscured the final diagnosis for years. Only after treatment of the eosinophilia were the underlying atypical lymphocytes more apparent on histology and a correct diagnosis made. Thus, when characteristic features of mycosis fungoides are subtle, eosinophils can act as a red herring in terms of clinico-pathologic correlation and may prevent early and accurate diagnosis of mycosis fungoides. We suggest that further studies are needed to evaluate whether treatments to reduce eosinophilia, once other causes have been excluded, may help clear the confounding reactive inflammatory infiltrate and facilitate the diagnosis of mycosis fungoides.
Keywords
Introduction
Lymphoeosinophilic infiltrates are found in a number of disorders including subtypes of dermatitis, insect bite reactions, autoimmune bullous diseases and drug reactions. 1 Eosinophilia, both peripheral and in cutaneous tissue, is not a common finding in mycosis fungoides (MF); however, it has been described in association with folliculotropic mycosis fungoides (FMF) and tumor stage MF. We describe three cases of FMF recently referred to our institution that were a diagnostic challenge given the concomitant presence of eosinophils in tissue, blood or both. Although clinical suspicion was high and repeat biopsies performed and reviewed, the diagnoses were only confirmed after the patients were treated with systemic agents reducing the confounding inflammatory infiltrate.
Case 1
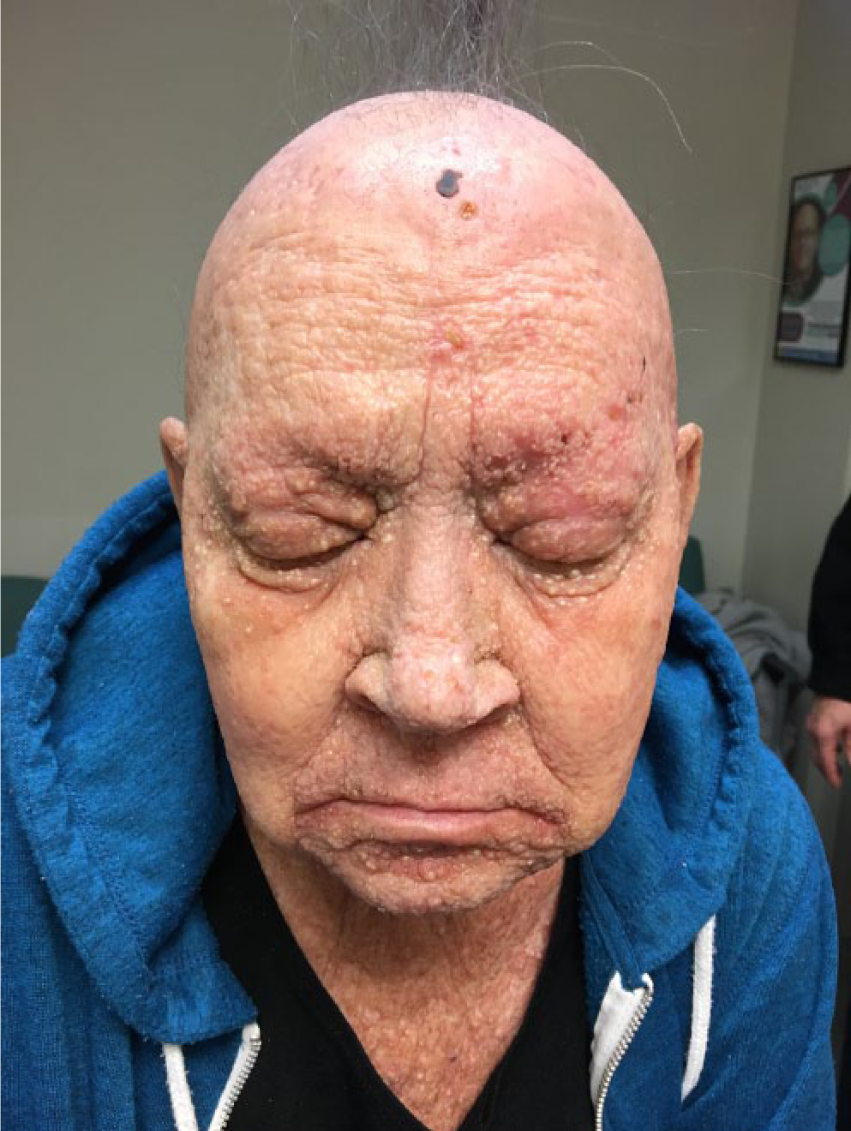
A healthy 56-year-old female was referred for a 5-year history of progressive skin rash. On examination, she presented generalized infiltrated erythematous-violaceous plaques and diffuse facial tumoral thickening with cystic areas, giving an overall leonine appearance (Figure 1). Suspecting an underlying lymphoproliferative etiology, biopsies including immunophenotyping and gene rearrangement studies were done and revealed a dense heavy dermal lymphoeosinophilic infiltrate, with no evidence of atypical lymphocytes (Figure 2). Flow cytometry and full blood workup were also negative, with no peripheral eosinophilia. To control her disfiguring disease, the patient was treated with oral prednisone up to 1 mg/kg daily, but due to persistent suspicion of lymphoma, repeat biopsies were performed. Only 4 months later while on continuous prednisone therapy did a biopsy show a greatly diminished eosinophilic infiltrate with underlying findings finally consistent with FMF (Figure 3). The patient is currently improving on low dose interferon, alitretinoin and local radiotherapy.

Case 1: Diffuse tumoral thickening of the face with cystic areas.
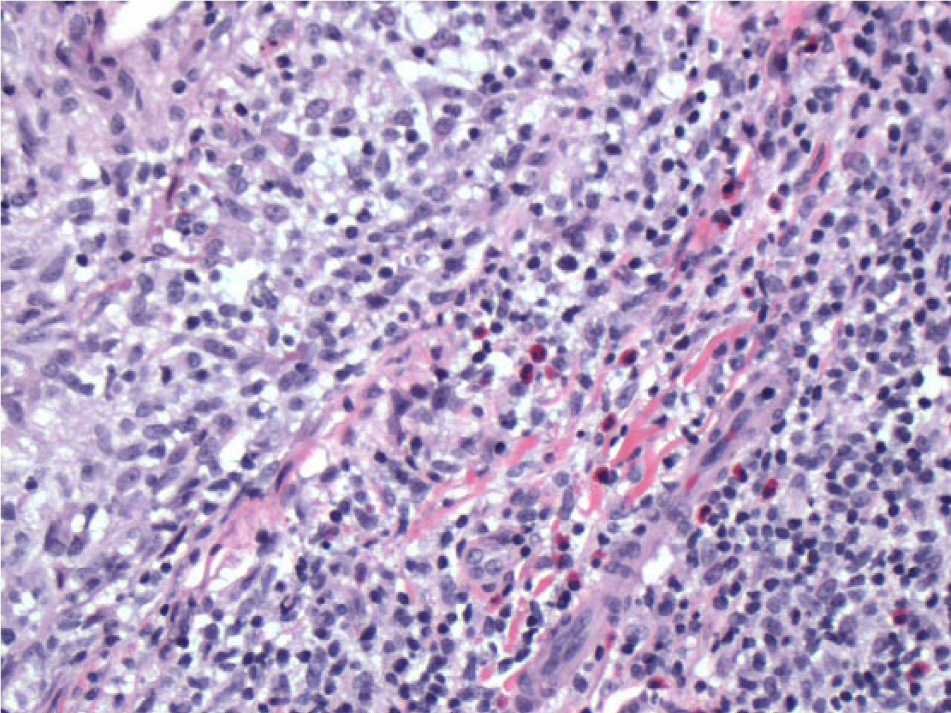
Case 1: Microscopic examination (200× magnification) of the initial biopsy demonstrates a prominent mixed lymphohistiocytic and eosinophilic, perifollicular and dermal, inflammatory infiltrate. The infiltrate is polymorphous and dominated by reactive cells obscuring the atypical lymphoid population.

Case 1: Microscopic examination (200× magnification) of the follow-up (post-treatment) biopsy demonstrates more moderate inflammation with less reactive background cells including fewer eosinophils. The atypical folliculotropic infiltrate is more conspicuous and permitted more reliable ancillary pathological testing.
Case 2
A 69-year-old male was referred for assessment of a 10-year refractory pruritic nummular rash with persistent eosinophilia around 1.00 × 109/L, for which extensive workup in hematology failed to reveal an underlying cause. Although clinically suspicious for MF, numerous biopsies over the course of his 5-year follow-up showed only spongiotic psoriasiform dermatitis with a lymphoeosinophilic infiltrate, without any atypical lymphocytes or epidermotropism. Gene rearrangement studies and direct immunofluorescence were unremarkable. With a presumed diagnosis of atypical psoriasiform dermatitis and no histologic evidence of MF, the patient was treated with corticosteroids, phototherapy and trials of various systemic agents. Following an acute flare unresponsive to over 3 months of daily prednisone and leflunomide, a 12th repeat biopsy finally revealed lymphoid atypia with prominent epidermotropism and folliculotropism with positive clonality, consistent with MF. He is now successfully undergoing treatment with low dose oral chlorambucil.
Case 3
A 72 year-old female was referred to our clinic for a 6-year history of widespread comedogenic rash (Figure 4). She was found to have hypereosinophilia up to 32.00 × 109/L; extensive assessment was unremarkable, including BCR-ABL, PDGFRA and B, JAK2, GBFB/MYH11, AML1/ETO, bone marrow biopsy, blood flow cytometry, and computed tomographic (CT) scan of the chest and abdomen. Although FMF was suspected, multiple skin biopsies over the course of 3 years only revealed follicular hyperkeratosis and dense mixed perifollicular inflammation with marked eosinophils. She was eventually treated with hydroxyurea, imatinib and interferon-alpha for presumed hypereosinophilic syndrome. Her eosinophilia improved 1 year later, and a repeat cutaneous biopsy at that time finally confirmed atypical lymphocytic infiltrate suspicious for MF. As her skin had not improved on interferon, oral alitretinoin and ultraviolet light therapy were added to her treatment regimen.
Case 3: Multiple comedo-like papules of the face with alopecia of the scalp and eyebrows.
Discussion
FMF is often considered an aggressive variant of MF with treatment frequently requiring systemic agents as for tumor stage disease. 2 As demonstrated by our three cases, the clinical presentation of FMF is extremely variable and includes follicle-based patches, plaques, tumors, keratosis pilaris–like lesions, alopecia and acneiform lesions. Unlike conventional MF, 3 eosinophils are sometimes reported in FMF and tumor stage MF. 4 Our case series highlights the importance of considering FMF in the differential diagnosis of dense lymphoeosinophilic infiltrates, especially if the clinical picture is suggestive as was the case in our patients.
The early diagnosis of MF is often challenging; clinical and histologic features can be subtle and overlap with that of many other inflammatory dermatoses. Diagnosis is often missed on initial biopsies and only confirmed later when the lymphocyte atypia becomes more obvious. In our three cases, although the high clinical suspicion was communicated to the pathologist, and all biopsies were interpreted at our center, which is recognized for its expertise in cutaneous lymphoma, biopsies remained un-diagnostic.
The presence of peripheral eosinophilia in MF, although reported in a few patients in the past,5 –7 is an uncommon finding, especially at such marked levels as in Cases 2 and 3. As the eosinophilia was not initially thought to be solely a reactive process to the underlying neoplasia in our patients, this presented a confounding factor that led to a diagnostic delay in confirming MF. A proposed explanation for the presence of eosinophils in MF is a shift from TH1 in patch stage to TH2 in tumor stage and/or Sezary syndrome. 8 This TH2 response leads to increased cytokines such as interleukin (IL)-4, IL-5 and IL-13, thus leading to eosinophilia. Elevated levels of eosinophil-derived eotaxin-3 and CC chemokine receptor 3 (CCR3) messenger RNA have also been described in advanced stages of MF. 9
There has been only one other report of a difficult to diagnose case of FMF presenting with eosinophilia in which prednisone decreased the eosinophil count before a diagnosis could be confirmed. We hypothesize that treatment of the eosinophilia with systemic corticosteroids in Cases 1 and 2, or with hydroxyurea in Case 3, ultimately enabled the histopathologic diagnosis by lessening the associated reactive infiltrate and allowing better visualization of the atypical lymphocytic population. Although eosinophil numbers were not quantitatively compared in the non-diagnostic and diagnostic biopsies, the three cases were ultimately reviewed by a single pathologist to eliminate potential confounders and discussed jointly with the local team of dermatologists and oncologists also specializing in cutaneous lymphomas. We believe the consideration of treating eosinophilia for the purpose of reducing the inflammatory distraction may clarify the correct diagnosis. More studies are necessary before coming to a conclusion, as there exists a theoretical concern that ablating the inflammatory response to the tumor cells may increase the risk of disease progression, although this was not observed in our series.
In conclusion, in patients with skin lesions clinically suspicious of MF but histologically presenting only a dense lymphoeosinophilic infiltrate, specific attention should be given to potential underlying atypical lymphocytes. Although an infrequent finding, severe peripheral eosinophilia can also be seen in MF, particularly FMF, and this should not exclude the diagnosis. Once other causes of peripheral eosinophilia and organ involvement have been ruled out, the use of systemic corticosteroids or other eosinophil-targeted therapies may help with the patients’ symptoms and lessen the confounding reactive infiltrate from the tissue. Studies should be done to better quantify the inflammatory infiltrate pre- and post-treatment and evaluate the impact of such treatment on potential progression of disease. Finally, it is important to emphasize that if there is a high clinical suspicion of MF, sometimes the only option is to stay persistent, repeat biopsies, and remain aware that eosinophils can be a red herring masking lymphoma.
Footnotes
Acknowledgements
A.J. and A.M. contributed equally as co-first authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained for all patients.
