Abstract
Objectives:
Duplication of the inferior vena cava (IVC) complicates interventional procedures. This case report aims to shed light on this unusual anomaly and the preoperative considerations necessary when treatment of venous thromboembolism is undertaken.
Methods:
An IRB approved case report of a 58 year old woman presented emergently with right lower extremity phlegmasia due to extensive thrombosis of her right iliofemoral and infrarenal portion of her duplicated IVC.
Results:
The patient underwent IVC filter placement and rheolytic thrombectomy with thrombolysis using the Angiojet device followed by venoplasty and stenting of the iliofemoral system and right IVC. Complete symptomatic and radiographic resolution on duplex imaging was achieved at 1 year follow up.
Conclusions:
With adequate preoperative awareness of IVC anomalies and treatment options available satisfactory results can be achieved and complications minimized for this unique patient population.
Introduction
Congenital duplication of the inferior vena cava (IVC) is reported in 0.2%–3% of the population1 –4 and is most frequently encountered incidentally due to widespread use of cross-sectional imaging in asymptomatic patients. However, anomalies of the IVC are increasingly recognized as a risk factor for venous thrombotic events.5,6 If venous thrombosis occurs, management strategy involves treatment of the early and late consequences of venous thrombosis, and protection from pulmonary embolism. Strategies to decrease the risk of venous thromboembolism (VTE) in this unique population include anticoagulation alone, 7 anticoagulation with IVC filter placement,8 –11 and steel coil embolization of duplicated IVC.8,12
Most recently, pharmacomechanical thrombectomy with thrombolysis has been used to treat thrombosis of a duplicated IVC, but only short-term follow-up is available. 13 We describe a patient who presented with extensive right iliofemoral thrombosis extending into her right duplicated IVC, who was successfully treated with Angiojet pharmacomechanical thrombectomy with thrombolysis (Angiojet, Possis Medical, Minneapolis, Minnesota, USA). This was followed by venoplasty and stenting of her right-sided IVC. At 1-year follow-up, she had complete symptomatic resolution of her deep vein thrombosis (DVT). Institutional review board (IRB) approval and informed consent were obtained from the patient.
Case report
A 58-year-old woman presented to the emergency room with acute swelling of the right lower extremity with progressive discoloration and severe pain. Venous duplex performed demonstrated extensive thrombosis of her right leg veins including the iliofemoral veins. The patient was diagnosed with phlegmasia, and venous recanalization was indicated to relieve her of her limb threat. She was not diagnosed with a duplicated IVC preoperatively. The patient was taken to the hybrid suite, and under fluoroscopic guidance, a Bard Eclipse IVC filter (Bard peripheral vascular, Tempe AZ, USA) was inserted into her infrarenal IVC via a right jugular vein approach. This IVC was noted to course to the patient’s left, which suggested aberrant anatomy. Next, via cannulation of the right lesser saphenous vein, venography was performed demonstrating near complete occlusion of the right leg venous system with extensive clot burden throughout (Figure 1). In addition, a duplicated right IVC was found which was distinct from the vessel in which the IVC filter was previously placed. The diagnosis of a duplicated IVC was thus made (Figure 2). Thrombolysis of the occluded right IVC, iliofemoral, and popliteal veins was performed with 10 mg of tissue plasminogen activator (TPA) delivered via an Angiojet DVX catheter (Angiojet, Possis Medical, Minneapolis, Minnesota, USA). Rheolytic thrombectomy was then performed with the same catheter. Following the procedure, residual stenosis in the duplicated right IVC and iliac veins were treated with serial venoplasty using a Dorado 10 × 150 mm balloon (Bard peripheral vascular, Tempe AZ, USA). Persistent stenosis after angioplasty was treated via stent placement. Four separate wallstents measuring 18 × 90, 18 × 60, 18 × 40, and 16 × 90 mm2 (Boston scientific/Meditech, Natick MA, USA) were deployed to cover the entire length of the duplicated right infrarenal IVC and right iliac veins. Completion venography now showed wide patency of the right iliofemoral vessels with restoration of flow through the duplicated right IVC into the suprarenal IVC (Figures 3 and 4). Postoperative review of the patient’s non-contrast computed tomography (CT) scan done 5 years prior had misidentified her left-sided IVC as an enlarged gonadal vein (Figure 5). The patient noted immediate resolution of limb edema, discoloration, and pain and was discharged on post-operative Day 2 with therapeutic anticoagulation and external compression. She subsequently underwent successful removal of her IVC filter that was placed in her duplicated left IVC. Nine months later, her follow-up duplex showed no residual venous thrombus with wide patency of the duplicated right IVC and iliofemoral veins. She remains asymptomatic at 1 year, and so her anticoagulation was stopped, and she is managed with lifelong antiplatelet therapy.
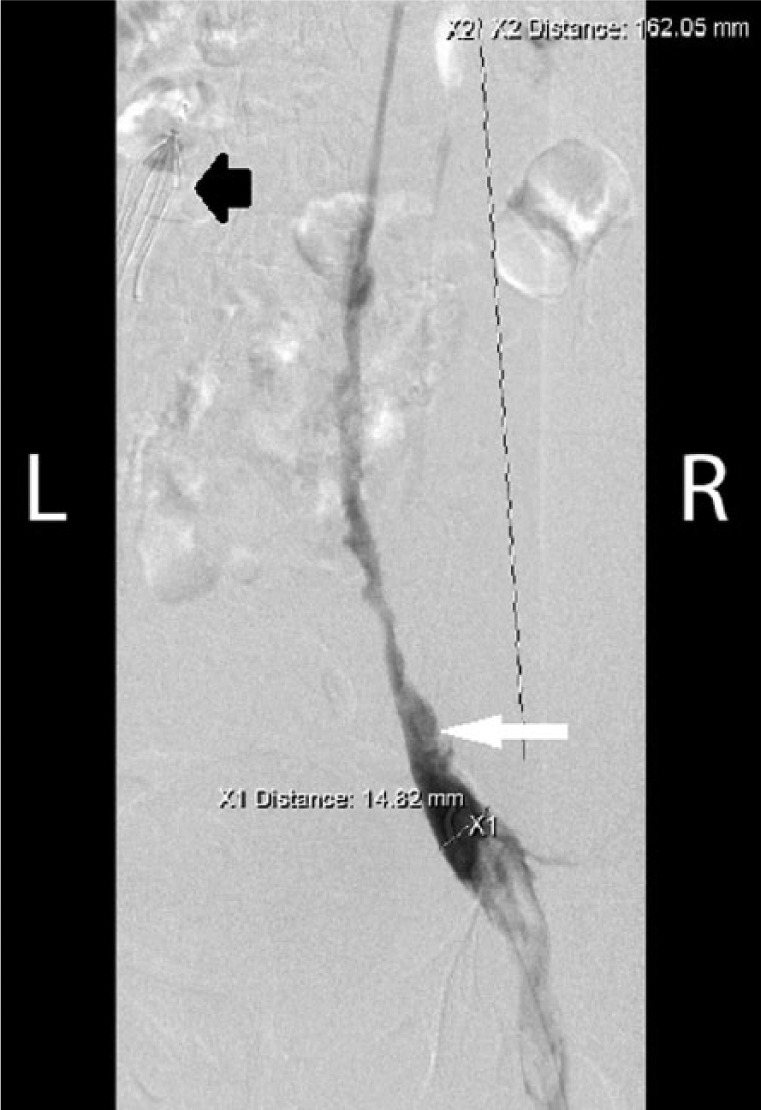
In the prone position, digital subtraction venography shows the patient’s right external iliac vein and right caval system almost completely occluded with thrombus (white arrow). An IVC filter has been deployed in the duplicated left caval system (short black arrow).

Digital subtraction images depicting patient’s true anatomy: venogram shows left-sided IVC with filter (black arrow), pre-aortic trunk (white arrow), and a wallstent (star) deployed in right infrarenal IVC.
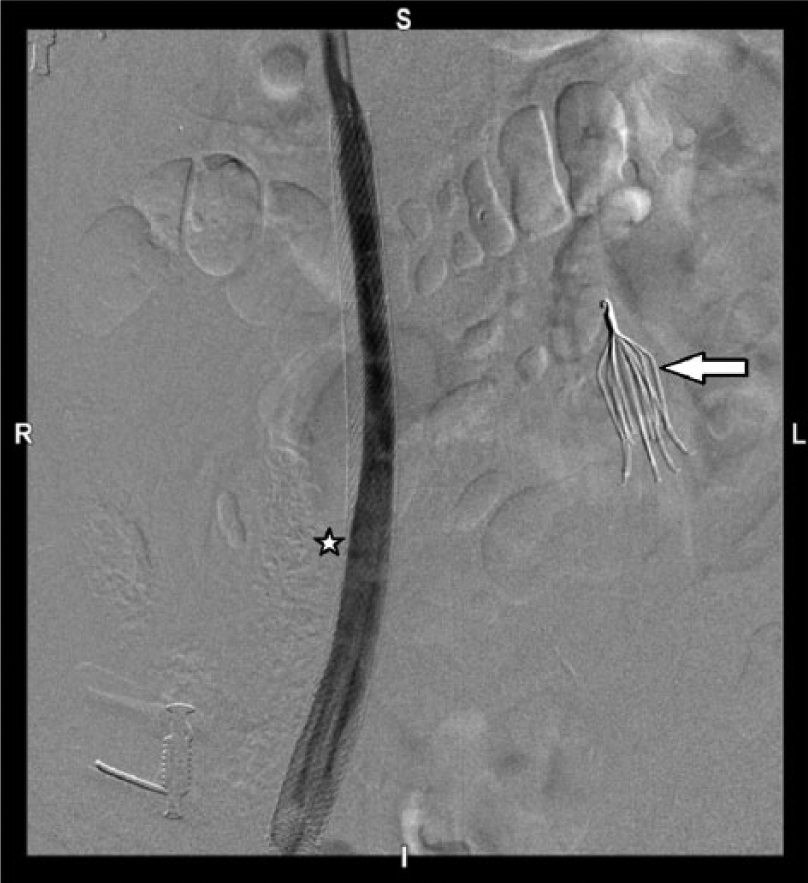
Fluoroscopic image showing Bard Eclipse IVC filter placed in the infrarenal portion of left IVC (white arrow). Venogram of the right-sided duplicated IVC after angioplasty and stenting shows wide patency (star).
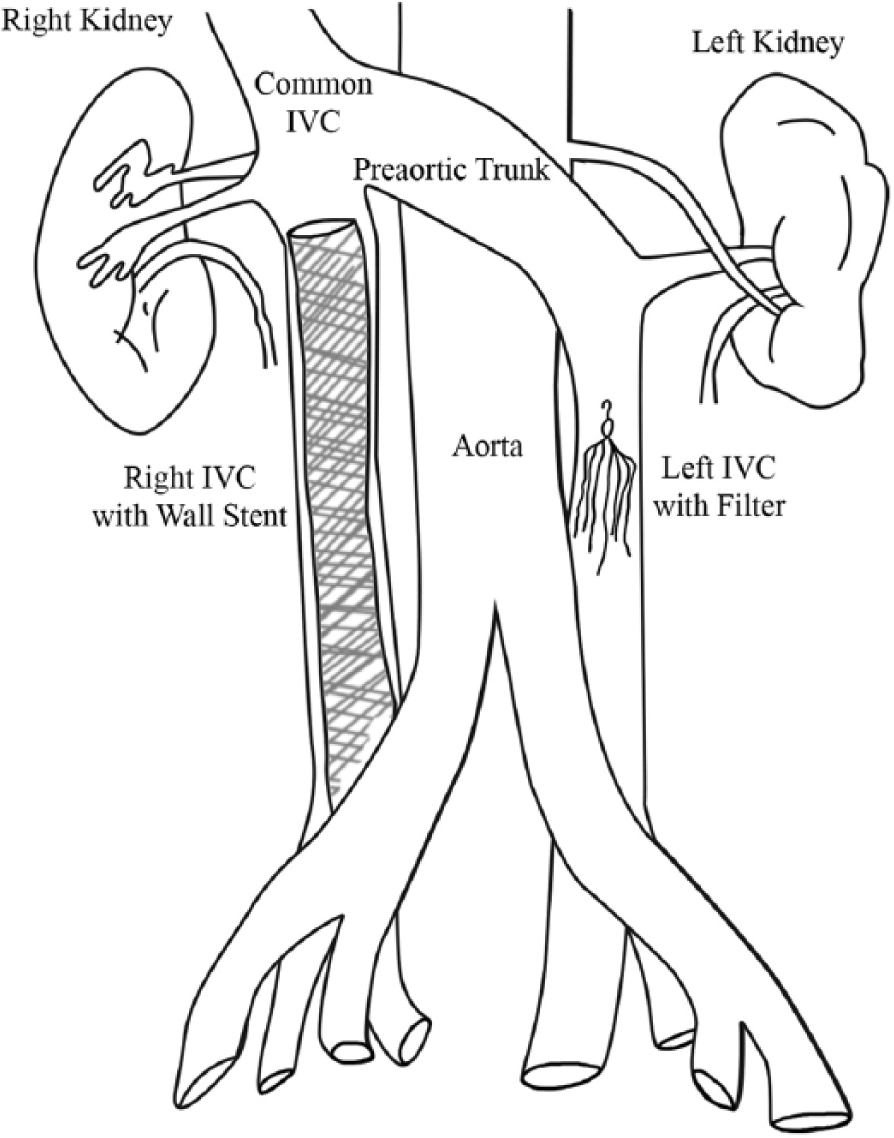
Cartoon depiction of anomalous anatomy shows pre-aortic trunk, stent placement in the infrarenal portion of the right IVC, and filter placed in the infrarenal portion of the left duplicated IVC.
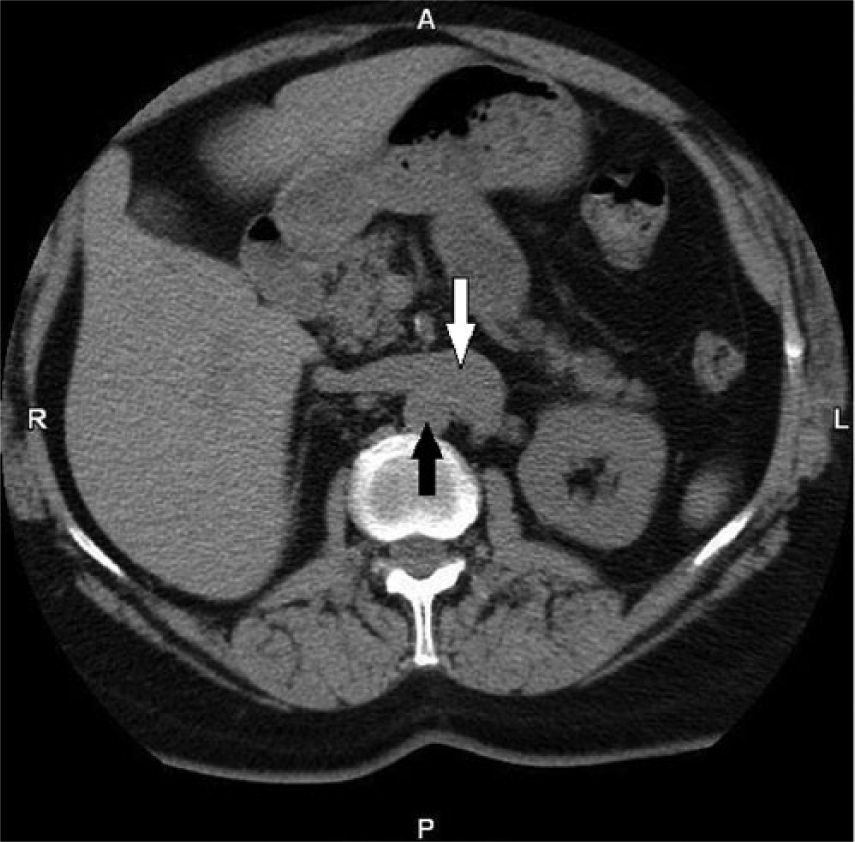
A non-contrast enhanced CT scan done 3 years prior to the misidentification of the duplicated IVC as an enlarged left gonadal vein. Pre-aortic trunk (white arrow) and aorta (black arrow).
Discussion
The presence of aberrant IVC anatomy should be suspected in patients where venography fails to demonstrate confluence of the common iliac veins, shows an aberrant course of the IVC, or shows unusually narrow central veins. Preoperatively, in such patients, the anomalous veins are frequently misdiagnosed on radiographic imaging.14,15 The majority of the literature regarding management of DVT in the presence of aberrant IVC anatomy focuses on decreasing the risk of pulmonary embolism with caval interruption.8 –11 In addition, the importance of preoperative cross-sectional imaging in the planning of open abdominal aortic aneurysm repair or retroperitoneal dissections cannot be understated. The presence of retro-aortic renal veins has been cited in the literature to complicate retroperitoneal dissections and aortic aneurysm repairs sometimes leading to significant hemorrhage.4,16 –18 Currently, there still remains, however, a paucity of data regarding advanced endovascular treatment of DVT in patients with aberrant venous anatomy, and the long-term patency after revascularization is currently unknown. A recent study has been published demonstrating short-term success with the use of the Trellis device for DVT in a patient with a duplicated IVC. 13
Similarly, the patient described in this report maintained venous patency with continued resolution of venous thrombosis at 1 year. At operation, it was unclear whether the patient had a duplicated IVC or a left-sided IVC; therefore, the IVC filter was placed in what was visualized to be the only patent infrarenal central vein. After correctly identifying the patient’s duplicated IVC, the option of suprarenal cava filter placement was considered. However, the risk of embolization was felt to be minimal, so thrombolysis and recanalization were performed without distal embolic protection.
Conclusion
This case sheds light on the treatment options available with evolving endovascular therapies for this uncommon subgroup of patients and highlights the importance of accurate intraoperative identification of venous anatomy. Recognizing aberrant venous anatomy ensures optimal management.
Footnotes
Acknowledgements
This work received the Second Place award at the Connecticut Chapter of the American College of Surgeons Resident Paper Competition 2013.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
