Abstract
The presence of retroperitoneal fibrosis after an aortic graft replacement is a marker of poor prognosis following aortic graft replacement. Herein we report the case of a 39-year-old man with retroperitoneal fibrosis that had been causing ureteral obstruction. The man had undergone repeated aortic graft replacement due to bacteremia and aortic graft–small intestinal fistula that occurred 4 years after initial aortic grafting for an inflammatory aortic aneurysm. The patient was discharged after 4 weeks of intravenous antibiotic therapy following the latest aortic graft replacement.
Introduction
Infection of aortic prosthetic grafts is an uncommon complication; 1 however, when it occurs, it may lead to life-threatening sequelae, with a mortality rate ranging from 25% to 88%. 2 Previous studies have shown that hydronephrosis, which is caused by retroperitoneal fibrosis or compression of the ureter by native and graft vessels, is associated with a high frequency of graft complications, such as anastomotic aneurysm, graft thrombosis, and amputation, and with a graft infection rate of 14%–48%. Hydronephrosis may be present preoperatively and persist after the surgery, or it may develop as a late complication after vascular reconstruction.3–6 We herein describe a patient who experienced various postoperative complications including hydronephrosis and graft thrombosis after the surgery of inflammatory aortic aneurysm.
Case report
A 35-year-old Japanese man presented with complaints of lumbar discomfort. Computed tomography (CT) showed the presence of an abdominal aortic aneurysm, and he was referred to our hospital. The CT image showed prominent periaortic tissue thickening (Figure 1(a) and (b)); thus, under the diagnosis of inflammatory aortic aneurysm, resection of the aortic aneurysm was performed by using a bifurcated knitted Dacron graft (Gelsoft Plus 16 × 8 mm; Terumo, Tokyo, Japan). The right graft limb was sewn directly onto the transected right external iliac artery with reconstruction of the right internal iliac artery using a separate graft leg piece. On the left side, the iliac graft limb was anastomosed to the side of the distal external iliac artery with oversewing of the distal common iliac artery. Histopathologic findings of the resected specimen showed a moderate degree of fibrous thickening in the intimal layer, fragmented elastic tissue in the medial layer, and dense fibrous tissue in the adventitial layer (Figure 2(a)). In the adventitial layer, infiltration of inflammatory lymphocytic cells, which were negative for IgG4 staining, was observed in the perivascular regions of the connective tissue (Figure 2(b) and (c)). Microbiological analysis of the resected aortic tissue was not performed. A duration of 22 days after the operation, the patient felt dysesthesia and hypesthesia of the lower limbs suggestive of graft failure, and CT demonstrated thrombotic occlusion of the graft (Figure 3(a)). As a result, removal of an intraluminal thrombus by Fogarty catheter was performed, and the implanted stent graft was replaced with a new one (Gelsoft Plus; Terumo).
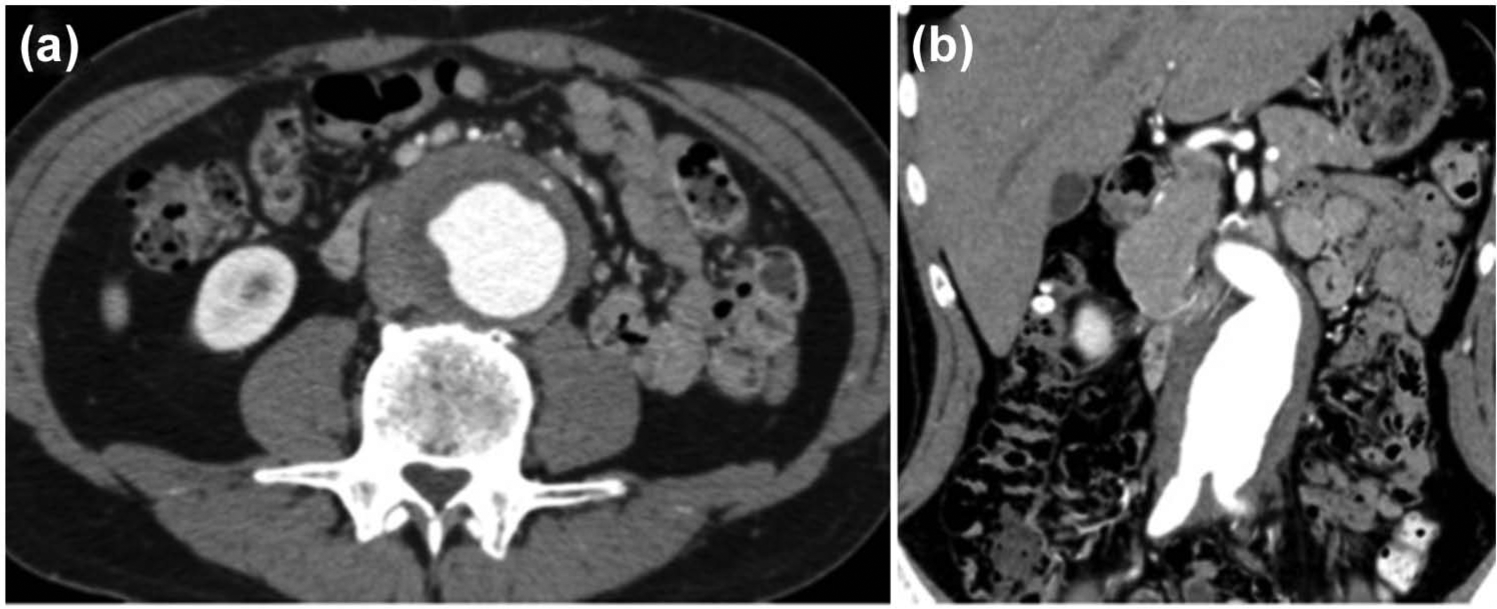
Computed tomography (CT) images and IgG4 staining of the tissue sample: (a) transverse and (b) coronal sections of the CT images before surgery are shown. Dense periaortic soft tissue was found to be surrounding the abdominal aorta and bilateral common iliac arteries. (c) IgG4 staining for the surgically resected sample.
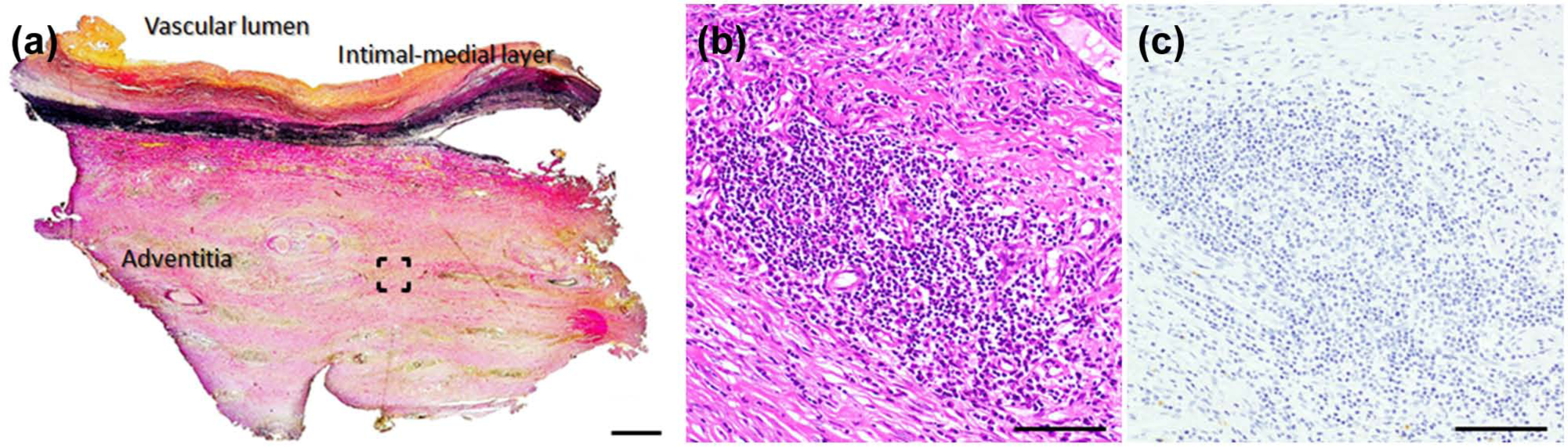
Pathological images for the surgically resected samples: (a) macroscopic image (elastica van Gieson’s staining) and (b and c) microscopic images of hematoxylin–eosin staining and IgG4 staining, respectively, of the bracketed region in (a). Original magnification, 200×. Scale bars indicate (a) 1 mm and (b and c) 100 µm.

Computed tomography and 18F-fluorodeoxyglucose-positron emission tomography (18F-FDG-PET) images after the initial graft surgery: (a) thrombotic graft occlusion occurring 12 days after the first graft surgery, (b) subphrenic abscess (arrows) was observed after ureteral stent implantation, (c) 18F-FDG-PET 29 months after the first surgery, and (d) abscess formation in the right gastrocnemius muscle 39 months after the first surgery. Ring enhancing lesions (arrows) are seen.
The patient experienced an intermittent low-grade fever starting from 3 months after the initial operation. Three months later, CT showed bilateral hydronephrosis, for which ureteral stent implantation was required, and a subphrenic abscess (Figure 3(b), arrows). Following antibiotics treatment, corticosteroid therapy was started at an initial dose of 30 mg/day; however, ureteral obstruction did not improve. Therefore, the ureteral stent could not be removed, which led to the necessity of ureteral stent replacement every 3 months. Twenty-nine months after these initial admission, pyuria and intermittent high fever developed, and 18F-fluorodeoxyglucose-positron emission tomography/CT showed abnormal uptake at the bilateral ureters and periaortic tissues (Figure 3(c)). After treatment with antibiotics (sulbactam/cefoperazone), C-reactive protein (CRP) levels were decreased (from 14.3 to 0.3 mg/dL) and the patient was discharged.
Ten months later (i.e. 39 months after the initial admission), the patient again complained of lower limb pain which was accompanied by rubefaction and swelling. A blood test showed an increased white blood cell count (13,470 cells/µL) and CRP levels (13.5 mg/dL). CT demonstrated abscess formation in the right gastrocnemius muscle (Figure 3(d)). Abscess drainage was performed and the patient received intravenous antibiotics (panipenem/betamipron 2 g/day) for 20 days. Four months later, the patient felt hypesthesia and dysesthesia of the right lower limb and was again admitted to our hospital. At this latest admission (44 months after the initial admission), thrombotic occlusion of the implanted graft at the level of the right iliac artery was found. Despite antithrombotic therapy for 6 days, the symptoms of the patient did no change. In addition, CT showed the presence of perigraft air (Figure 4). At this time, Gram-positive bacilli were identified in the blood culture, and

Computed tomography images during the latest admission. Computer tomography images 44 months after the initial admission: (a) transverse, (b) coronal, and (c) sagittal sections are shown. Perigraft air (arrows) was observed, which suggested graft infection.
During the operation, an aortic graft–small intestinal fistula was found (Figure 5). After partial resection of the small intestine, the contaminated graft was replaced with a new rifampin-soaked graft (Gelsoft Plus; Terumo), which was covered by the major omentum. Removal of ureters from the periaortic tissue was impossible owing to severe adhesion. Microbiology analyses of the explanted graft showed a positive culture for

Aortic graft–small intestinal fistula: (a) adhesion of the prosthetic graft and small intestine was observed and (b) after the separation of graft, perforation of the small intestine became apparent (arrow).
Discussion
In the current study, we have described a patient who developed multiple complications after aortic graft implantation for inflammatory aortic aneurysm; these include graft thrombosis, gastrocnemius muscle abscess formation, graft infection, and aortoenteric fistula. Although corticosteroid therapy was continued, it did not ameliorate the ureteral entrapment induced by retroperitoneal fibrosis; thus, repetitive replacement of ureteral stents was required. On the latest admission, open surgical repair was mandatory due to the graft infection and intestinal fistula.
Inflammatory aortic aneurysm is a distinct clinical entity from atherosclerotic aortic aneurysm that was first described in 1972, 7 and it is defined by the presence of a thickened aneurysm wall, extensive perianeurysmal and retroperitoneal fibrosis, and dense adhesions of adjacent abdominal organs. 8 In the present case, the uretal obstruction was not apparent before the operation. We note that inflammatory aortic aneurysm is generally diagnosed in the absence of ureteral obstruction, whereas it may be called perianeurysmal retroperitoneal fibrosis when a ureteral stricture coexists. 9
Endovascular aortic repair (EVAR) is a newer option for the treatment of aortic aneurysm with fair feasibility and durability. 10 Several previous studies have shown that the perianeurysmal mantle may shrink, albeit not always, after both open surgery and EVAR; 11 furthermore, recent studies showed that periaortic inflammation regressed in more than half of the patients after EVAR.12,13 The ureteral obstruction may worsen postoperatively, as in our case. For example, Von Fritschen et al. 14 demonstrated that after surgery for inflammatory aortic aneurysm, hydronephrosis that was not present at the time of the operation was found to have developed in 19% of patients, despite improving or stable inflammatory lesions. In addition, Vallabhaneni et al. 15 advocated that it may not be a therapeutic option when perianeurysmal fibrosis is present preoperatively, and EVAR may induce perianeurysmal fibrosis in approximately one-sixth of patients without evidence of this condition.
On the contrary, several previous studies have shown that EVAR may also improve hydronephrosis, although the rate of the improvement, which might be dependent on the graft that is used, 16 may be lower than that of open surgery.12,17,18 Ureteral entrapment, when it persists postoperatively, may develop into renal atrophy, and therefore, corticosteroid or other immunosuppressive therapy, as well as ureteral stenting, may be considered empirically. However, whether the persistent inflammatory response can be modified pharmacologically remains debatable. 19 In fact, despite the amelioration of periaortic cuffing with corticosteroid therapy, bilateral hydronephrosis persisted in the present case.
Postoperative retroperitoneal fibrosis may be not only difficult to manage but also associated with high incidence of graft complications, which might occur a few years after the surgery as in our case.3,6 In a retrospective analysis of 33 years of experience, Wright et al. 6 reported that among 3580 patients who had underwent aortoiliac reconstruction, ureteral complications developed postoperatively in 44 patients, among whom, 24 experienced graft complications (anastomotic aneurysm, infection, thrombosis of the graft) at an average of 3.3 years after operation.
At the latest admission, CT showed perigraft air, suggestive of graft infection. Perigraft air may be more likely to be seen with an aortoenteric fistula; however, it may be present with severe graft infection alone.5,20 A combination of other diagnostic modalities, such as ultrasonography, endoscopy, and gallium scintigraphy, may improve the accuracy for the diagnosis of graft infection with or without aortoenteric fistula. 5 Although the direct extravasation of contrast from the aorta into a bowel loop is a specific sign of fistula formation, such findings may be quite difficult to identify on CT. 21 In our case, however, in addition to sustained inflammation, aortoenteric fistula, which may be highly life-threatening, was suspected; therefore, other diagnostic tests, including nuclear medical tests, could not be performed at that time.
Our patient experienced intragraft thrombosis in addition to aortoenteric fistula. It should be questioned whether the EVAR approach, when possible, is superior in suppressing these multiple complications as compared with open surgery. On the contrary, late, as well as early, graft thrombosis may occur after the stent graft implantation.22,23 Considering that intragraft thrombosis may also be a consequence of graft-related infection, 24 controlling the infection may be more essential than deciding which strategies to select.
In summary, we herein have described a patient who developed aortic graft infection 3.5 years after open surgical repair for the treatment of inflammatory abdominal aortic aneurysm. The patient was also treated for ureteral obstruction that was caused by worsening retroperitoneal fibrosis with ureteral stent implantation and corticosteroid therapy. The patient experienced other graft-related complications, namely, intragraft thrombosis and aortoenteric fistula, and partial resection of the small intestine and replacement of the contaminated graft with a new one were required. As both persisting and de novo periaortic fibrosis may lead to serious sequelae, such as chronic renal failure, sepsis, and other graft infection-related complications, a careful long-term follow-up is mandatory.
Footnotes
Declaration of conflicting interests
The authors declare no conflict of interest in preparing this article.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
