Abstract
Objective:
Ventricular assist device is one of the treatment options for heart failure patients. Therefore, the purpose of this review is to aid in clinical decision-making of exchanging previous older ventricular assist device models to the newest one, HM3.
Methods:
The search was conducted across several databases until February 25, 2023, and was registered with the ID of CRD42023405367. Risk of bias was performed using Cochrane Risk of Bias 2.0 and the Newcastle Ottawa Scale. In order to rank and evaluate the pooled odds ratios and mean differences with 95% confidence intervals, we employed conventional and Bayesian network meta-analysis converted to surface under the cumulative ranking.
Results:
A total of 49 studies with 31,105 patients were included in this review. HM3 is the best device exchange choice that causes the lowest risk of mortality (HM3 (99.98) > HM2 (32.43) > HVAD (17.58)), cerebrovascular accidents (HM3 (99.99) > HM2 (42.41) > HVAD (7.60)), other neurologic events beside cerebrovascular accident (HM3 (91.45) > HM2 (54.16) > HVAD (4.39)), pump thrombosis (HM3 (100.00) > HM2 (39.20) > HVAD (10.80)), and bleeding (HM3 (97.12) > HM2 (47.60) > HVAD (5.28)). HM3 is also better than HM2 in hospital admissions (OR: 1.90 (95% CI: 1.15–3.12)). When complications were present, HM2 or Heartware ventricular assist devices exchange to HM3 lowered the mortality rate compared to exchanging it to the same device type.
Conclusion:
HM3 is the best device for all six outcomes. Exchange from Heartware ventricular assist devices or HM2 to HM3 rather than the same ventricular assist device type is recommended only if a complication is present.
Background
Out of all ventricular assist devices (VADs), HeartMate 3 (HM3), which belongs to the newest generation of VADs and has received Food and Drug Administration (FDA) approval, is the most widely used and the only available VAD in the market today. Many trials and observational studies claimed that HM3 is associated with lower complication risks.1–12 Despite HM3’s superiority and sole availability, a large number of patients ultimately used HeartMate 2 (HM2) or Heartware ventricular assist devices (HVAD) since HM3 had not been widely distributed previously and was still in trials. What is even more concerning for these patients is that the FDA has instructed HVAD distribution to stop as of 2021. 13 In such cases, the issue becomes, “Is device exchange to HM3 beneficial?” Hence, our study will evaluate three types of VADs using a large sample pool in order to provide greater significance to investigate the need for device exchange.
Method
Materials and Methods
Our review was conducted following the guidelines outlined in the Cochrane Handbook for Systematic Reviews of Interventions (6.2). Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) were used to report our results. In addition, we ensured compliance with international research ethics regulations by registering this study in the international prospective register of systematic reviews (PROSPERO) under the distinct identification number CRD42023405367.
Search strategy
Until February 25, 2023, four independent reviewers reviewed multiple databases, including Wiley, EBSCOHost, Scopus, PubMed, and ProQuest. Four independent reviewers searched multiple electronic databases, such as Wiley, EBSCOHost, Scopus, PubMed, and ProQuest, up to February 25, 2023. HeartMate 2, axial flow pump, HeartMate 3, centrifugal flow pump, HeartWare VAD, and exchange were the keywords utilized for the research process. Advanced search methods to refine the search were used whenever applicable and accessible.
Study eligibility criteria
The inclusion and exclusion criteria were utilized to screen the studies. Inclusion criteria: (1) an observational study or randomized control trial; (2) comparison between HM2 versus HM3 versus HVAD or comparing HM2 or HVAD exchange to HM3 or the same type of VAD; (3) heart failure patients, but without any coexisting conditions that can influence survival rate; and (4) qualitative and quantitative outcomes. Exclusion criteria: (1) unsuitable study design, including reviews, commentaries, letters to the editor, or preclinical studies; (2) studies with qualitative outcome data only. All steps are visually depicted in Figure 1.
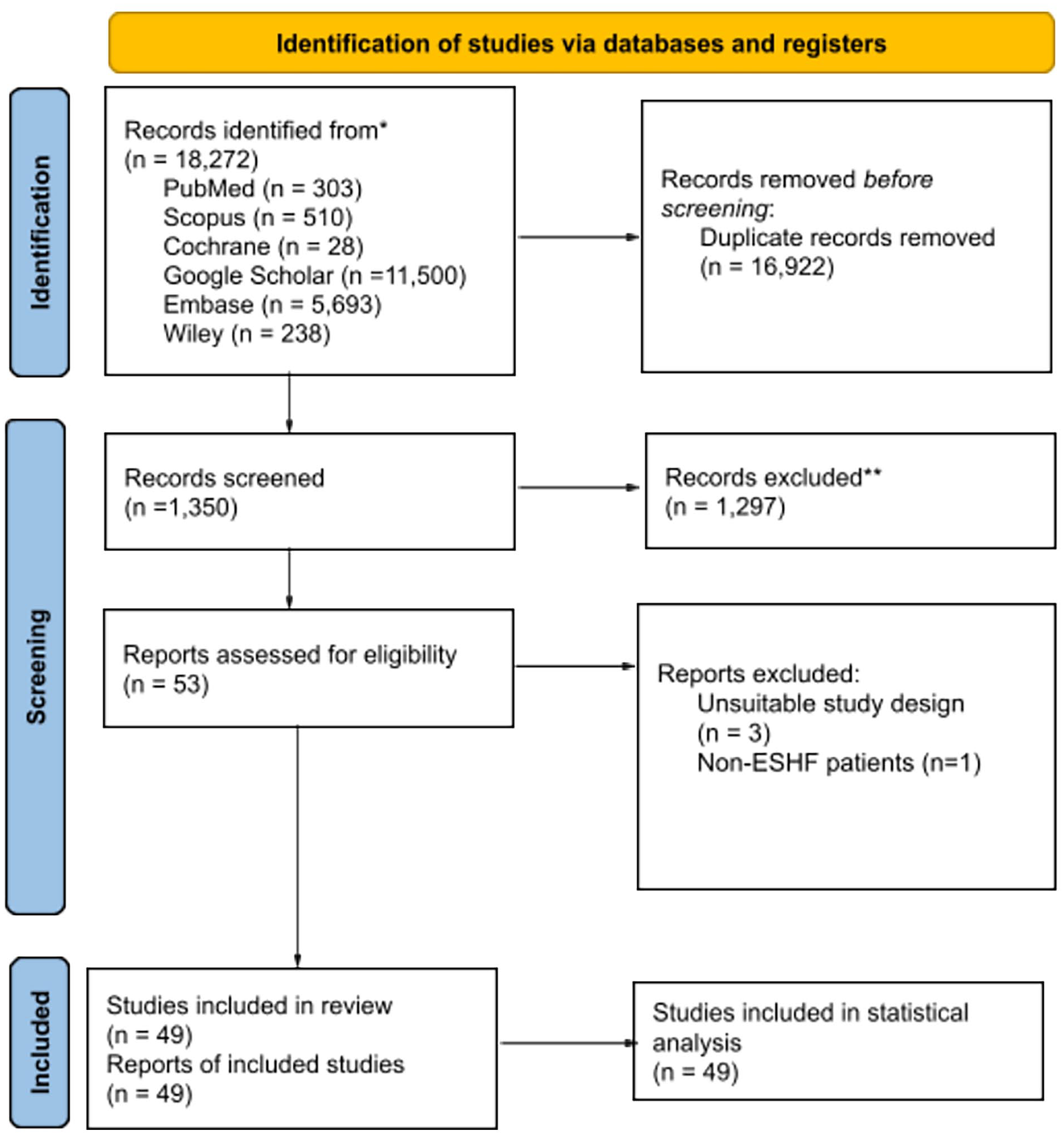
PRISMA flowchart.
Data extraction
Our extraction data consist of (1) study ID; (2) study attributes, including the method and place of study; (3) samples, including the size, age, and device types; and (4) study outcomes. Study results are divided into two main sections: hospital stay and admissions, as well as postimplantation outcomes for the first implantation and after the exchange. The hospital stay and admissions section consists of first hospital admissions, length of stay (LOS) in the intensive care unit (ICU), and total LOS. Meanwhile, the postimplantation outcomes include infection related to VAD, sepsis, cerebrovascular accidents (CVAs), other neurologic events, device malfunction and/or replacement, right ventricular failure, pump thrombosis, bleeding, cardiac arrhythmias, respiratory failure, renal dysfunction, hepatic dysfunction, reoperation due to any complications, and all-cause mortality. During the process of data extraction, all authors obtained and recorded both qualitative and quantitative characteristics. The accuracy of the extracted data was then further verified during the statistical analysis phase.
Risk of bias in assessment
For observational studies, a thorough appraisal of the included studies’ bias was performed using the Newcastle–Ottawa quality assessment scale. 14 The obtained scores were subsequently converted to the Agency for Healthcare Research and Quality (AHRQ) standards, where scores above 6 were classified as good, scores ranging from 4 to 6 were deemed fair, and scores below 4 were categorized as poor. 15 On the other hand, the randomized control trials were evaluated using the Cochrane Risk of Bias 2.0 (Cochrane Methods, 2021), which evaluates five domains of possible bias sources which are randomization bias, bias due to deviations from intended interventions, missing outcome data, outcome measurement, and bias in reporting results. The first domain, the randomization bias, is further divided into the randomization process and participant selection. 16 The results will be compacted and illustrated with the RobVis tool. Three reviewers independently conducted the quality assessment without knowledge of each other’s scores. Subsequently, they engaged in discussions until a consensus was achieved.
Statistical analysis
Review Manager ver. 5.4 (The Nordic Cochrane Center, The Cochrane Collaboration, Copenhagen) was used to perform statistical analysis. The odds ratio was obtained from studies, and then the pooled effects were interpreted. The DerSimonian–Laird random effects model provided by Riley et al. was applied due to the possibility of indecipherable heterogeneity from studies. Heterogeneity was then calculated using estimated effect (I 2 ) statistics based on the Cochrane threshold (heterogeneity cut-off limits: 0% as insignificant, 25% as low, 50% as moderate, and 75% as high). 17
We carried out a Bayesian network meta-analysis using MetaInsight V4.0.1 Beta for outcome(s) with more than two groups that yielded a p-value <0.05 in the conventional meta-analysis. Forest plots of pooled effect compared each treatment and ranking panel that is included in this study using surface under the cumulative ranking (SUCRA) that was used for the intervention measures, with higher values indicating lower complications. However, the inconsistency assessment of the network cannot be performed due to the open loop of the network. Consequentially, the heterogeneity was reported by means of direct assessment from the forest plot random effects model. 18
Results
Study selection and characteristics
Initially, a total of 18,272 studies were discovered through 6 database searches (specific details can be found in Supplemental Appendix A.1). After removing duplicate and irrelevant studies, 63 records were evaluated based on the eligibility criteria. Ultimately, 49 studies were deemed suitable for both qualitative and quantitative analysis, as depicted in Figure 1. The characteristics of these included studies can be found in Supplemental Appendix B.1 (pertaining to the first implantation) and Supplemental Appendix B.4 (concerning device exchange after the initial implantation). Additionally, Supplemental Appendix B.1 also provides information on the patients’ INTERMACS scores, which is a scoring system utilized for evaluating clinical profiles and predicting unfavorable outcomes following VAD implantation.1–12,19–52
This systematic review encompassed a total of 31,105 patients across all included studies. The majority of these studies were conducted in the United States or Europe. VAD is a commonly employed treatment for patients with advanced heart failure, serving as either a definitive therapy or a bridge to transplant. The study considered three types of VADs: HM2, HM3, and HVAD. Out of the 49 studies, only 5 directly compared these 3 intervention techniques. Specifically, 11 studies compared HM2 with HM3, 16 studies compared HM2 with HVAD, 13 studies compared HM3 with HVAD, and 4 studies focused on outcomes related to VAD exchange to HM3.
Quality assessment
All randomized controlled trials (RCTs) included in this study have a low risk of bias based on Cochrane risk of bias 2.0 (Figure 2 and Supplemental Appendix C.1). The observational study included in our study was assessed in terms of selection, comparability, and outcome using the Newcastle–Ottawa Scale (Supplemental Appendix C.2–C.7). Overall, these observational studies were rated to be of high quality with a low risk of bias. According to the AHRQ, only six studies were deemed to be of “poor quality” (Supplemental Appendix C.5).1–12,19–52

Visualization of risk of bias 2.0.
Postimplantation outcomes
Fourteen aspects of postoperative outcomes were assessed, such as infection, neurological adverse events, postoperative bleeding, mortality (Supplemental Appendix B.2).1–12,19–52 The device showed the highest score in the SUCRA that indicates the lowest risk of adverse events is HM3. This VAD is correlated with a significantly lower risk of complications which is overall mortality (HM3 (99.98) > HM2 (32.43) > HVAD (17.58)), CVA (HM3 (99.99) > HM2 (42.41) > HVAD (7.60)), other neurologic events beside CVA (HM3 (91.45) > HM2 (54.16) > HVAD (4.39)), pump thrombosis (HM3 (100.00) > HM2 (39.20) > HVAD (10.80)), and bleeding (HM3 (97.12) > HM2 (47.60) > HVAD (5.28)). An exception was found in terms of bleeding that showed HM3 did not differ significantly from HVAD. All of these results presented a heterogeneity ranging from insignificant to high. Meanwhile, for other outcomes such as infection, cardiac arrhythmia, and reoperation, as well as right ventricular, respiratory, and renal failure, these three types of VADs have similar results (Supplemental Figure 1).
Hospital admissions and stay
Five included studies that reported patients’ first hospital admissions after VAD implantation (Supplemental Appendix B.3).4,5,10,11,25,38,45,48 HM3 is proven to be superior to HM2 with lower hospital admissions (OR: 1.90 (95% CI: 1.15–3.12)) with range of admission timing from 1 until 2 years. However, comparison between HM3 and HVAD could not be made because only one study reported this outcome and within a short follow-up term (1-month) (Supplemental Figure 2). While another included studies that reported ICU, and overall, LOS were unable to be analyzed because of different outcome measurements.
VAD exchanges
Device exchange in most included studies is usually performed when device malfunction is present due to pump thrombosis, other mechanical problems, or other VAD-related problems such as infection. An exchange that was performed due to complications showed that the mortality rate is lower if the HM2 or HVAD is replaced with HM3 rather than replacing it with the same device type. However, if the exchange is performed electively (early exchange to prevent complication risks), HVAD exchange to HM3 showed a higher mortality rate compared to continuing HVAD support (Supplemental Appendix B.4).53–56
Discussion
VADs are technological supports used as a lifesaving treatment option for patients with severe decompensated HF and an ejection fraction lower than 25%. These devices, which are implanted into the left chambers of the heart, provide an alternative pathway for oxygenated blood flow toward the aorta.1,3 The evolution of VADs has marked significant technological advancements, particularly in the transition from second- to third-generation pumps. Second-generation VADs, exemplified by the HeartMate II (Abbott Laboratories, Illinois, USA) and Jarvik 2000, are characterized by axial flow, directing blood flow parallel to the axis of rotation. These devices employ mechanical or contactless bearings to support the impeller. 57
In contrast, third-generation VADs, such as the HVAD and HM3, utilize centrifugal flow, where blood flow is directed perpendicular to the axis of rotation. The HVAD incorporates hybrid magnetic/hydrodynamic impeller suspension technology, while the HM3 features fully magnetic levitation. This innovation in the HM3 significantly reduces heat generation, friction, and shear stress, which in turn minimizes the risk of thrombus formation within the pump. 57
The HM3 received approval for commercialization in Europe in October 2015 and in the United States in August 2017. The HVAD, initially approved in Europe in 2009 and in the United States in 2012 as a bridge to transplantation, faced discontinuation in June 2021 due to technical issues and the superior clinical outcomes demonstrated by the HM3. The HM3’s design has shown a marked reduction in thrombotic events and ischemic stroke rates compared to its predecessors, solidifying its status as the only third-generation VAD currently approved by the FDA.58–60
Despite showing development compared to its prior generations, implantation of the VAD has not been completely free from adverse events. Moreover, reviews that discuss postimplantation outcomes are still severely limited, and none address VAD-related hospital stays or admissions. 56 Our systematic review and meta-analysis included 31,105 patients across 6 RCTs and 43 observational studies. All outcomes were categorized into two big sections: postimplantation outcomes as well as hospital admissions and stay.
For postoperative outcomes, our study reported that HM3 is the best device with the lowest mortality rate (HM3 (99.98) > HM2 (32.43) > HVAD (17.58)). However, a meta-analysis by Cavarretta et al. 61 that compared several devices across different VAD generations with a total of 1,141 patients stated that HM2 is the best device, whilethe HM3 and HVAD have similar overall mortality rates and do not show any statistically significant differences in mortality. A meta-analysis by Kerdany et al. 62 showed there is no discernible difference between HM2 and HM3 in terms of mortality. The results of both of these meta-analyses are in contrast with our study. However, our results are proven to be more valid because other meta-analyses that only include limited amount of samples, and their included studies are dominated with indirect comparison with single-arm trial studies, further decreasing the significance of the results.
HM3 also causes the lowest risk of other adverse events such as CVA (HM3 (99.99) > HM2 (42.41) > HVAD (7.60)), other neurologic events besides CVA (HM3 (91.45) > HM2 (54.16) > HVAD (4.39)), pump thrombosis (HM3 (100.00) > HM2 (39.20) > HVAD (10.80)). A network meta-analysis by Cavarette et al. 61 reported that HM3 is the best device that has a lower risk of device thrombosis, and stroke, thus strengthening our study results. Mehra et al.’s 1 study also strengthens our finding with their statement that showed HM3 might become an answer to survival free of disabling stroke or reoperation (HM2 (21.2%) versus HM3 (10.7%); OR: 0.368 (0.273–0.497)). Furthermore, as aforementioned, the HM3 lower flow disruptions, leading to a lower number of pump thrombosis (HM2 (89 (17.6) versus HM3 (11 (2.1)); 0.090 (0.049–0.163)). Therefore, supported by a study by Heathley et al., 63 lower pump thrombosis correlates with lower reoperation. The free-from-reoperation rate of HM3 at 6 months reaches 87% due to fewer pump replacements in case of thrombus and drive-line problems, and at 24 months, the success rate becomes 55%. With optimized fluid dynamics and wider blood-flow passages, HM3 is surely a choice for lowering both neurological problems and pump thrombosis events.1,61,62
Further discussing the comparison with HVAD, the VAD that is also in the newest generation group as HM3 was discontinued due to pump thrombosis and malfunction, as it was also proven by our study’s results. These adverse events were caused by different rotors because as previously stated HM3 utilized magnetically levitated ones, while HVAD used hydrodynamic bearings, which, while reducing frictions, still involve some degree of mechanical contact. This led to higher wear and tear over time, thus contributing to pump thrombosis and malfunctions that became the basis of our study’s results. Furthermore, HVAD also caused higher CVAs, and this is mainly proposed due to lower effectivity of mimicking heart pulsality, which potentially led to higher shear stress on blood component, increasing risk of clot formation and CVAs. HM3 is designed to create a more physiologic pulsatile flow despite being a continuous flow-device, which can improve hemocompability, reducing the risk of clot and CVAs, as opposite to HVAD mechanism. Future VADs are suggested to have a rotor with low even nonexistent flow disruption as well as effective pulsatile flow to prevent major complications such as CVA, pump thrombosis, and malfunction, these major complications are the one that causes HVAD, as part of the new generation to be discontinued.1,24,61
In terms of other adverse events, network meta-analysis by Cavaretta et al. also reported that HM3 is the best device that has a lower risk of bleeding, thus strengthening our study results. However, in contrast to Cavaretta et al., our study showed that bleeding in HM3 (OR: 1.35 (95% CI: 0.92–2.03)) did not differ significantly from HVAD. In addition, the Cavaretta et al.’s study also stated that HVAD is better than HM2 in terms of bleeding and device thrombosis outcomes, while our study stated otherwise (HM3 (97.49) > HM2 (42.02) > HVAD (10.49)). 57 Nevertheless, another meta-analysis by Kerdany et al. stated that bleeding and pump thrombosis between HM2 and HM3 have no significant differences, which contradict ours and Caveretta’s study results. This is mainly due to more indirect comparison with single-arm trial studies included in Kerdany et al.’s and Cavaretta et al.’s study.61,62
According to Cavaretta et al.’s study 61 HM2 is also claimed to be the best device for driveline infection, neurologic, and renal dysfunctions, as well as respiratory and right ventricular failures. These results align with a meta-analysis by Kerdany et al. 62 that stated VAD infection is lower in HM2 compared to HVAD, although there is no direct comparison between these three devices. Our study, on the other hand, reported that there is no significant difference of infection, renal, and right ventricular failure between the three devices in all of these outcomes. This is mainly due to more indirect comparative studies included in Kerdany et al.’ and Cavaretta et al.’s studies. Hence, showing more reliability in our study results provided a more direct comparison if compared to the previous two studies.
Regarding the infection that become the major concern for VAD-supported patients, our study as previously stated showed comparable results. This is mainly because these VADs utilized similar percutaneous drivelines that cause similar infection rates. Regarding the percutaneous placement, Wert el al.’s study 64 stated that double tunneling or placement through rectus muscle, then to subcutaneous led to significantly lower infection rates. Based on this, placement in other layers such as muscles will lower infection rates. Besides the placement method, future VADs could develop drivelines with antimicrobial coatings or materials in the driveline to reduce the risk of bacterial adherence. 65 Aside from the antimicrobial materials, the diameter size and stiffness of the driveline also matter; Imamura et al.’s study 66 stated that VAD with larger outer diameter and higher stiffness might lower infection rate as proposed in the comparison between HM2, EVAHeart, and DuraHeart. In addition, wireless power transmission for VADs aims to eliminate the need for percutaneous drivelines, eliminating driveline-related infection risks. Campi et al.’s study demonstrated the potential of near-field wireless power transfer systems using magnetic resonant coupling, capable of efficiently powering implanted devices like VADs. 67
For the hospital admissions, our study showed there are lower hospital admissions in HM3 compared to HM2 (OR: 1.90 (95% CI: 1.15–3.12)). Our study results are also strengthened by Pagani et al., that stated HM3 patients have overall lower readmissions events. 4 Patients with VAD are usually rehospitalized due to a few common reasons, including bleeding, pump thrombosis, and neurologic events. 21 In our analysis, HM3 was also found to cause the lowest risk for CVA (HM3 (99.99) > HM2 (42.41) > HVAD (7.60)), other neurologic events besides CVA (HM3 (91.45) > HM2 (54.16) > HVAD (4.39)), pump thrombosis (HM3 (100.00) > HM2 (39.20) > HVAD (10.80)), and bleeding (HM3 (97.12) > HM2 (47.60) > HVAD (5.28)). Lower adverse events in all of these outcomes that become common reasons for patients to be hospitalized could cause lower admissions in HM3 patients compared to HM2. 11 Meanwhile, hospital admissions for HM2 and HVAD did not differ significantly due to lots of similarities in most complications’ risks between these two devices. 61
Few studies have reported on the ICU stay and overall LOS after VAD implantation, but no explanations are given about these results. In our study regarding this group, we were unable to perform statistical analysis due to incomparable measurement parameters. All of our included studies have shown that there are a high number of variations in the length of ICU and LOS. Therefore, there is no definite conclusion about which device is best for lowering ICU stay and LOS. Still, further studies with the same measurement parameters are needed to make a definitive conclusion about the overall hospital and ICU stay.68,69
VADs exchange
In line with the findings of our study that proved HM3 as the newest and best VAD in regards to postimplantation outcomes (specifically in six outcomes), an exchange to HM3 is considered. Our study results showed that HM2 or HVAD exchange to HM3 caused a lower mortality rate than exchange to the same device type as the previous one. The exchange that was performed due to device problems, such as pump thrombosis and other VAD-related problems shows greater benefits for the patients rather than staying on support with the same device type. For elective exchange, a study by Cogswell et al. stated that HVAD to HM3 exchange was associated with a higher mortality rate (26.7%) compared to continuation of HVAD support (21%). Coyle et al. also stated that HVAD exchange to HM3 is correlated with higher mortality and complications rates compared to HM2 exchange to HM3 (29%–43% versus 0%). Furthermore, it is also correlated with higher postoperative complications including right ventricular failure requiring right VAD support (71% versus 0%), prolonged ventilator support days (1 versus 11), and increased need for reoperation (14% versus 100%). This leads to a suggestion that HM2 to HM3 exchange is more recommended than HVAD to HM3, although if there is a life-threatening complication, exchanging to HM3 is preferred over exchanging with the same VAD type. Furthermore, from these studies, there are no enough evidences that support elective exchange of HM2 or HVAD to HM3.53–56
Strength and limitations
Some of our study outcomes differed from those of the previous studies, but to be noted is the fact that huge limitations existed in other studies which might affect the results. In the meta-analysis performed by Cavaretta et al., most of the p-values are above 0.05, proving their results are insignificant. 58 Besides, a meta-analysis by Kerdany et al. that compared HM2, HM3, and HVAD only provided limited direct network comparison between the three devices and a limited amount of samples. 59 Superior to all these studies is the fact that our study provided significant results in six outcomes than other studies that provided direct comparisons. Furthermore, we also included newer and more studies with larger patient populations for VAD outcomes. We also provided outcomes about VADs exchange.
Despite the previously demonstrated superiority, this study acknowledges several limitations. These limitations encompass the heterogeneity of the data, which may stem from variations in outcome parameters, sample sizes, and study designs (including both RCTs and observational studies). We still decided to include both study designs because the RCTs performed gave various outcomes which differ between studies. Furthermore, the incomparability of study periods and timeframes could potentially result in the omission of specific study developments, and studies primarily focused on the USA or Europe might have been inadvertently overlooked. Additionally, the meta-analysis results concerning device exchange outcomes are constrained by the limited number of studies available, but more likely in the current evidence trend, device exchange to HM3 is only suggested if there are life-threatening complications.
Conclusion
HM3 has proven to be better than its previous generation, HM2, and another third generation, HVAD as initial VAD choice. HM3 provides better outcomes by lowering overall mortality, neurological problems (CVA and others), pump thrombosis, bleeding, and hospital admissions, but it did not differ significantly from HVAD in terms of bleeding and hospitalization. In addition, HM2 has been proven to be superior to HVAD in terms of bleeding and CVA. Other than the aforementioned, all three devices have similar outcomes. Postimplantation device exchange from HM2 or HVAD to HM3 rather than the same VAD type is recommended only if a life-threatening complication is present. Future suggestion about the VAD technological and material advancement as well as surgical technique will contribute to lowering all adverse events aforementioned.
Supplemental Material
sj-doc-6-smo-10.1177_20503121241278226 – Supplemental material for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis
Supplemental material, sj-doc-6-smo-10.1177_20503121241278226 for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis by Dudy Arman Hanafy, Theresia Feline Husen, Ruth Angelica, Ilona Nathania, Widya Trianita Suwatri, Pasati Lintangella, Wahyu Prima Erdianto and Prisillia Prasetyo in SAGE Open Medicine
Supplemental Material
sj-docx-1-smo-10.1177_20503121241278226 – Supplemental material for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis
Supplemental material, sj-docx-1-smo-10.1177_20503121241278226 for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis by Dudy Arman Hanafy, Theresia Feline Husen, Ruth Angelica, Ilona Nathania, Widya Trianita Suwatri, Pasati Lintangella, Wahyu Prima Erdianto and Prisillia Prasetyo in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241278226 – Supplemental material for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis
Supplemental material, sj-docx-2-smo-10.1177_20503121241278226 for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis by Dudy Arman Hanafy, Theresia Feline Husen, Ruth Angelica, Ilona Nathania, Widya Trianita Suwatri, Pasati Lintangella, Wahyu Prima Erdianto and Prisillia Prasetyo in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121241278226 – Supplemental material for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis
Supplemental material, sj-docx-3-smo-10.1177_20503121241278226 for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis by Dudy Arman Hanafy, Theresia Feline Husen, Ruth Angelica, Ilona Nathania, Widya Trianita Suwatri, Pasati Lintangella, Wahyu Prima Erdianto and Prisillia Prasetyo in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121241278226 – Supplemental material for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis
Supplemental material, sj-docx-4-smo-10.1177_20503121241278226 for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis by Dudy Arman Hanafy, Theresia Feline Husen, Ruth Angelica, Ilona Nathania, Widya Trianita Suwatri, Pasati Lintangella, Wahyu Prima Erdianto and Prisillia Prasetyo in SAGE Open Medicine
Supplemental Material
sj-docx-5-smo-10.1177_20503121241278226 – Supplemental material for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis
Supplemental material, sj-docx-5-smo-10.1177_20503121241278226 for Heartware ventricular assist device versus HeartMate II versus HeartMate III in advanced heart failure patients: A systematic review and meta-analysis by Dudy Arman Hanafy, Theresia Feline Husen, Ruth Angelica, Ilona Nathania, Widya Trianita Suwatri, Pasati Lintangella, Wahyu Prima Erdianto and Prisillia Prasetyo in SAGE Open Medicine
Footnotes
Acknowledgements
All authors have nothing to declare.
Author Contributions
Conceptualization, Dudy Arman Hanafy and Theresia Feline Husen; methodology, Theresia Feline Husen; software, Theresia Feline Husen, Ilona Nathania, and Ruth Angelica,; validation, Dudy Arman Hanafy and Widya Trianita Suwatri, formal analysis, Theresia Feline Husen, Wahyu Prima Erdianto, Ilona Nathania, Ruth Angelica; investigation, Dudy Arman Hanafy and Theresia Feline Husen; resources, Dudy Arman Hanafy, Wahyu Prima Erdianto, Theresia Feline Husen, Ilona Nathania,Ruth Angelica, Widya Trianita Suwatri, Pasati Lintangella, and Priscillia Prasetyo; Data curation: Dudy Arman Hanafy, Theresia Feline Husen; writing—original draft preparation, Dudy Arman Hanafy, Theresia Feline Husen, Ilona Nathania, and Ruth Angelica; writing—review and editing, Dudy Arman Hanafy, Theresia Feline Husen, Ilona Nathania,Ruth Angelica, and Wahyu Prima Erdianto; visualization, Theresia Feline Husen, Ilona Nathania, and Ruth Angelica; supervision, Dudy Arman Hanafy and Widya Trianita Suwatri; project administration, Theresia Feline Husen; funding acquisition, Dudy Arman Hanafy. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Not applicable.
Informed consent
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
