Abstract
Purpose:
To investigate changes in anterior scleral thickness (AST) in patients with Posner–Schlossman Syndrome (PSS).
Methods:
Sixty-two patients with PSS were enrolled. AST was measured using swept-source optical coherence tomography at 0 mm (AST0), 1 mm (AST1), 2 mm (AST2), and 3 mm (AST3) from the scleral spur.
Results:
AST0, AST1, AST2, and AST3 were significantly reduced in PSS-affected eyes compared with that in fellow eyes. Furthermore, we divided patients into two subgroups based on the course of PSS: the short-term (PSS course < 1 year) subgroup and the long-term (PSS course ⩾ 1 year) subgroup. In the short-term subgroup, no significant differences in AST parameters (AST0, AST1, AST2, and AST3) were observed in PSS-affected and fellow eyes, while in the long-term subgroup, all the AST parameters (AST0, AST1, AST2, and AST3) were significantly reduced in PSS-affected eyes compared with that in fellow eyes.
Conclusions:
PSS-affected eyes demonstrated significantly reduced AST compared with that in fellow eyes. Moreover, a significant reduction of the AST was observed in long-term PSS-affected eyes, but not in short-term PSS-affected eyes. This indicates that changes in AST in PSS are time-dependent and progressive. Significant reduction of the AST is observed only after a certain period of time.
Keywords
Introduction
Posner–Schlossman Syndrome (PSS) is an acute, intermittent, recurrent glaucomatocyclitis characterized by anterior uveitis with markedly elevated intraocular pressure (IOP).1–6 Repeated attacks of PSS may result in glaucomatous optic nerve damage, and ultimately lead to blindness.7–10 Research has shown that approximately 26.4% of patients with PSS develop glaucoma, while the exact etiology and pathophysiology of PSS remain unclear. 9
Our previous studies have revealed iris atrophy, trabecular meshwork (TM) edema, and Schlemm’s canal (SC) enlargement in patients with PSS.11,12 CMV is considered the leading etiology of PSS,7,8 with studies reporting that the iris is susceptible to CMV 13 and may serve as a reservoir for latent CMV.14,15 The direct viral invasion or vasculitis caused by CMV infection may be the cause of atrophy and thinning of the iris,11,16 with inflammation also playing an important role in this.11,17 This kind of anterior uveitis may also have an effect on the anterior scleral thickness (AST). Moreover, the TM and SC are situated in the limbus, which is located right between the cornea and anterior sclera. 17 The scleral spur (SS), which is part of the anterior sclera, is an important factor for maintaining TM and SC morphology.18,19 Therefore, alterations in AST may influence the morphology and function of the TM and SC. To date, no study has quantitatively assessed AST in PSS. Notably, AST has been reported to be significantly reduced in eyes with primary open-angle glaucoma 18 ; however, the changes in AST associated with secondary glaucoma, such as PSS, are not characterized. Therefore, the primary objective of this study was to investigate the effects of PSS on the AST.
Methods
Subjects
Sixty-two patients with PSS were enrolled in this cross-sectional study, which was conducted over an 18-month period from January 2021 to June 2022. All the patients underwent ophthalmic examinations (slit-lamp observation, IOP measurement, fundus observation, gonioscopy, corneal endothelial microscopy, axial length (AL) measurement (IOL-Master 500; Carl Zeiss Meditec, Dublin, OH, USA), and cup-to-disc (C/D) ratio assessment with fundus photographs (AFC-210; Nidek Co., Ltd., Gamagori, Aichi, Japan). The diagnostic criteria for PSS were as follows: (1) unilateral and recurrent disease episode; (2) transient elevated IOP with blurred vision; (3) hoar and white suet-shaped keratic precipitates and/or mild inflammation in the anterior chamber; (4) open anterior chamber angle without iris synechiae. 12 Patients/eyes with a history of uveitis, scleritis, ocular laser, ocular surgery, primary glaucoma, secondary glaucoma caused by conditions other than PSS, high myopia, or systemic diseases were excluded.
To further analyze the influence of the disease course of PSS on AST, we divided the patients into two subgroups based on the course of PSS: short-term (PSS course < 1 year) (n = 19) and long-term (PSS course ⩾ 1 year) subgroups (n = 43). In addition, PSS-affected eyes were further stratified into two subgroups based on the presence (n = 17) or absence (n = 45) of secondary glaucoma to investigate whether significant differences in AST existed between the two groups.
Swept-source optical coherence tomography imaging acquisition and processing
Both the PSS-affected and fellow eye of each patient underwent swept-source optical coherence tomography (SS-OCT) (CASIA SS-1000; Tomey Corporation, Nagoya, Japan). The nasal and temporal limbs were measured separately by an experienced examiner (XY) after adjusting the fixture to the corresponding scan areas.
The AST was measured as the distance from the episcleral blood vessels (thin hyporeflective area in the anterior part of the sclera) to the posterior boundary of the sclera (the line separating the hyperreflective sclera from the hyporeflective ciliary muscle).18,19 The AST was measured at 0 mm (AST0), 1 mm (AST1), 2 mm (AST2), and 3 mm (AST3) from the SS (Figure 1). The pupil diameter (PD) was measured as the distance between the two opposite sides of the pupillary margin of the iris. 20 All the measurements were conducted using the ImageJ software (National Institutes of Health, Bethesda, MD, USA) by an experienced observer (ML) blinded to the study information.
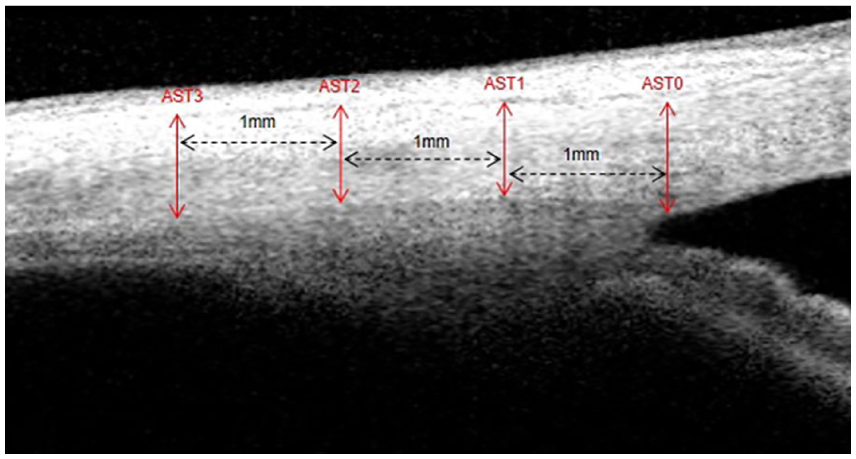
The measurements of AST. AST0, AST1, AST2, and AST3 are measured at 0 mm, 1 mm, 2 mm, and 3 mm from the scleral spur, respectively. AST, anterior scleral thickness.
Statistical analysis
Data are shown as mean ± standard deviation, where applicable. The normality of interocular differences was assessed using the Shapiro–Wilk test, and the Wilcoxon signed-rank test was used to compare measurements in the PSS-affected eyes with that of the fellow eyes, as the differences did not follow a normal distribution. Parameters with nasal and temporal values were compared with generalized estimating equations (GEEs) because GEEs take the correlation between the nasal and temporal measurements of one eye into account.11,12,18,21 Linear regression was used to determine the associations between AST and corneal endothelium (CE) density and the C/D ratio in eyes with PSS. Adjusted β coefficients for the associations between independent and dependent variables were assessed using GEEs. To test the reproducibility of AST measurements, the AST (including both nasal and temporal AST in PSS-affected and fellow eyes) was re-measured by the same observer. Intraobserver reproducibility was assessed using the intraclass correlation coefficient (ICC). All the analyses were performed using R-software version 3.4.3 (http://www.r-project.org) and EmpowerStats software (www.empowerstats.com; X&Y Solutions, Inc., Boston, MA, USA). All the tests were two-tailed, and p < 0.05 was defined as statistically significant.
Results
As shown in Table 1, no significant differences in AL or PD were observed between the PSS-affected and fellow eyes (both p > 0.05). The CE density was significantly lower, and the C/D ratio and IOP were significantly greater in the PSS-affected eyes compared with that in the fellow eyes (all p < 0.05).
Study subject characteristics.

C/D, cup-to-disc.
Wilcoxon signed-rank test.
Comparison of AST in PSS-affected and fellow eyes
AST0, AST1, AST2, and AST3 were significantly reduced in PSS-affected eyes compared with that in fellow eyes (all p < 0.05) (Table 2).
Comparisons of AST between PSS-affected and fellow eyes.

AST, anterior scleral thickness; AL, axial length.
The influence factors such as age, sex, and AL have been adjusted.
Generalized estimating equations.
Comparisons of AST between PSS-affected and fellow eyes in short-term (PSS course <1 year) and long-term (PSS course ⩾ 1 year) subgroups
In the short-term subgroup, no significant differences were observed between the PSS-affected and fellow eyes for all the AST parameters (AST0, AST1, AST2, and AST3) (all p > 0.05). In the long-term subgroup, all the AST parameters (AST0, AST1, AST2, and AST3) were significantly reduced in PSS-affected eyes compared with that in fellow eyes (all p < 0.05) (Table 3).
Comparisons of AST between PSS-affected and fellow eyes in short-term (course of PSS < 1 year) and long-term (course of PSS ⩾1 year) subgroups.
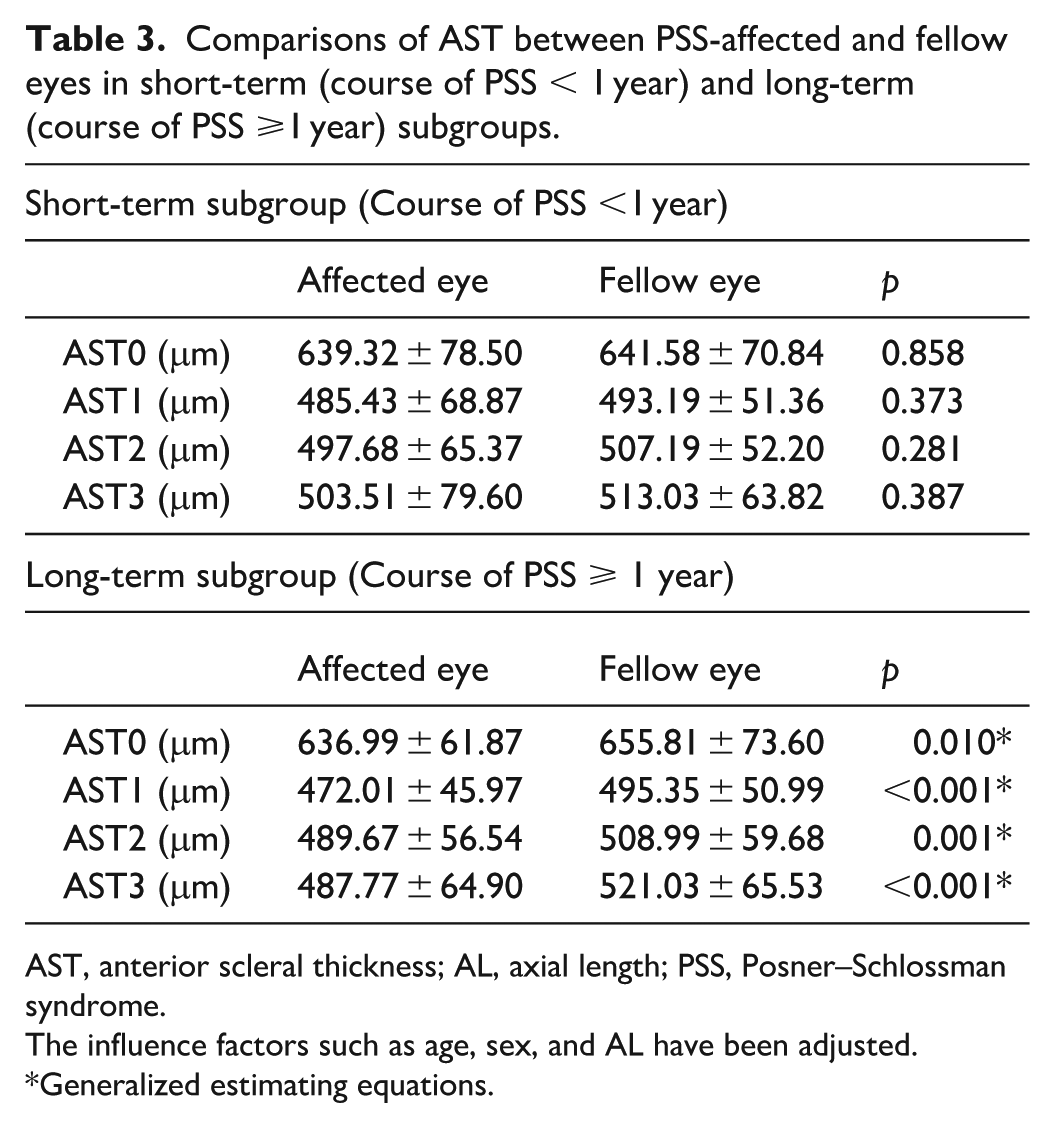
AST, anterior scleral thickness; AL, axial length; PSS, Posner–Schlossman syndrome.
The influence factors such as age, sex, and AL have been adjusted.
Generalized estimating equations.
Comparison of AST between PSS-affected eyes with and without secondary glaucoma
After adjusting for potential confounding factors, including age, sex, and AL, no statistically significant differences were observed between the two subgroups (with and without secondary glaucoma) for any of the measured AST parameters (Table 4).
Comparisons of AST between PSS-affected eyes with and without secondary glaucoma.
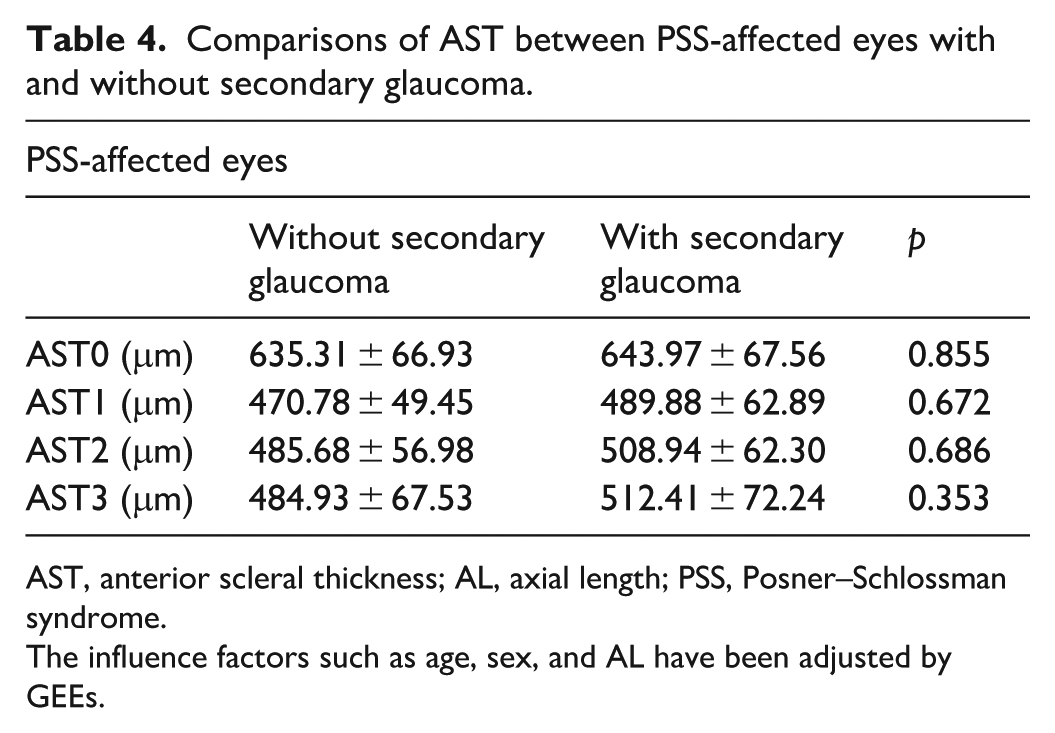
AST, anterior scleral thickness; AL, axial length; PSS, Posner–Schlossman syndrome.
The influence factors such as age, sex, and AL have been adjusted by GEEs.
Comparison of nasal and temporal AST between PSS-affected and fellow eyes
In the nasal subgroup, AST1 and AST3 were significantly different in PSS-affected and fellow eyes (both p < 0.05), whereas AST0 and AST2 showed no significant differences (both p > 0.05). In the temporal subgroup, AST0, AST1, AST2, and AST3 were significantly lower in PSS-affected eyes than that in fellow eyes (all p < 0.05) (Supplemental Table S1).
Comparison of nasal and temporal AST between PSS-affected and fellow eyes in short-term and long-term PSS subgroups
In the short-term subgroup, a significant difference between the PSS-affected and fellow eyes was observed only for nasal AST3 (p = 0.049). In contrast, in the long-term subgroup, significant differences were observed for all the AST measurements, except for nasal AST0 and AST2 (all p < 0.05) (Supplemental Table S2).
Univariate linear regression analysis of the association between AST and CE density in PSS-affected eyes
No significant associations between AST and CE density were observed in PSS-affected eyes (all p > 0.05) (Supplemental Table S3).
Univariate linear regression analysis of the association between AST and C/D ratio in PSS-affected eyes
No significant associations between AST and the C/D ratio were observed in PSS-affected eyes (all p > 0.05) (Supplemental Table S4).
Reproducibility of AST measurements
The results indicated that the reproducibility of the measurements was good. The ICC of the measurements ranged from 0.855 to 0.929 (Table 5).
Reproducibility of AST measurements.

AST, anterior scleral thickness; AL, axial length; ICC, intraclass correlation coefficient; PSS, Posner–Schlossman Syndrome.
Discussion
To date, no clinical observations of changes in AST in patients with PSS have been reported. In this study, we conducted quantitative AST measurements at different distances (0/1/2/3 mm) from the SS using SS-OCT and observed that all the AST parameters (AST0, AST1, AST2, and AST3) were significantly reduced in PSS-affected eyes compared with that in fellow eyes. Furthermore, when patients were divided into short-term and long-term PSS subgroups, the result pattern was significantly different. In patients with short-term PSS, none of the AST parameters (AST0, AST1, AST2, AST3) were significantly different to that of the fellow eyes; however, in patients with long-term PSS, all the AST parameters (AST0, AST1, AST2, AST3) were significantly different to that of the fellow eyes, with reduced AST in PSS-affected eyes. Previous studies have reported significant TM edema, CE loss, iris atrophy, and C/D enlargement in PSS-affected eyes.8–12 In addition to these findings, we also identified significantly reduced AST in PSS-affected eyes compared with that in fellow eyes in the current study. To the best of our knowledge, this is the first study to report changes in AST in patients with PSS.
As previously mentioned, PSS is associated with CMV infection,7,8 which leads to both iris atrophy 16 and CE loss.8,22,23 PSS is also associated with significantly elevated levels of inflammation proteins and cytokines in the anterior chamber.3,6,24 Therefore, anterior segmental inflammation may also contribute to iris atrophy and CE loss in PSS-affected eyes. 11 Aketa et al. suggested that iris injury is associated with increases in inflammation cytokines (e.g., TNF-α IL-1, IL-4, IL-6, IL-8, and MCP-1) in the aqueous humor, 17 and Alfawaz et al. indicated that anterior uveitis/inflammation has a negative effect on CE count. 25 Moreover, given that the anterior sclera is located right next to the CE and iris, CMV infection and anterior inflammation may also have negative effects on the anterior sclera, leading to its atrophy and reduction of the AST, similar to the changes in the iris and CE in PSS-affected eyes.
Although PSS has been reported as self-limited and benign, 1 glaucomatous optic nerve damage has also been reported in PSS-affected eyes.8–10 The potential reasons could be the acute elevation of IOP and subsequent decrease in ocular perfusion (determined by both local arterial blood pressure and IOP) during PSS attack.11,26 In addition to optic nerve damage, this could also contribute to iris atrophy in PSS-affected eyes. 11 Thus, the injurious effect of acute elevation of IOP and the subsequent decrease in ocular perfusion could be observed in both the anterior (iris atrophy) and posterior (glaucomatous optic nerve damage) segments of PSS-affected eyes, indicating that the whole eyeball/ocular tissue could be affected. Accordingly, we speculated that in addition to the iris and optic nerve, the mechanical pressure and ischemia caused by high IOP in PSS-affected eyes may also act on the anterior sclera, resulting in anterior sclera atrophy and AST reduction. Thus, acute elevation in IOP could be an additional reason for the reduction of the AST in PSS-affected eyes.
Another interesting phenomenon observed in the current study was significant reduction of the AST in long-term PSS-affected eyes, but not in short-term PSS-affected eyes. Accordingly, we speculated that the atrophy of the anterior sclera and reduction of the AST in PSS might be time-dependent and progressive. Therefore, in the short-term period after PSS onset, the sclera was able to withstand the negative influences of PSS (CMV infection, anterior inflammation, mechanical pressure, and ischemia caused by high IOP); thus, no significant changes in the anterior sclera and AST occurred. However, when the course of PSS reached 1 year or longer, pathological changes in the anterior sclera and AST gradually became significant. Considering the close relationship between AST reduction and the course of PSS, we also speculated that reduction of the AST in PSS-affected eyes might also be a clinical indicator for long-term PSS.
This study had certain limitations. First, this was a clinical observational study in which only morphological data derived from SS-OCT were collected and analyzed. CMV load testing was not performed. Therefore, the potential influence of the CMV status on AST was not evaluated in the present study. Second, we only observed the anterior sclera and not the posterior sclera in this study. Third, this was a cross-sectional observational study and not a longitudinal follow-up study. Therefore, dynamic changes in AST were not recorded. Fourth, during the PSS attack, short-term applications of IOP-lowering medications were chosen, and IOP-lowering medications may have affected AST. 27 However, all the administration of IOP-lowering treatment was short-term and temporary. Only a small subset of study patients (3 of 62, less than 5%) received prostaglandin analog treatment. Thus, the potential effect of IOP-lowering medications on AST in patients with PSS may be limited in this study. Fifth, the number of episodes per patient was not recorded in this study. Sixth, a sample size calculation was not performed prior to the initiation of this study. Seventh, this study was conducted in China. Considering that the prevalence of CMV infection varies across geographic regions and countries,28,29,30 our results may therefore only be specifically applicable to a Chinese/Asian population.
Conclusion
This is the first study to report that PSS-affected eyes have significantly reduced ASTs compared with that in fellow eyes. Moreover, significant reduction in AST was observed in long-term PSS-affected eyes, but not in short-term PSS-affected eyes. Thus, changes in AST in PSS are time-dependent and progressive and significant reduction of AST occurs only after a certain period of time after PSS onset.
Supplemental Material
sj-docx-1-smo-10.1177_20503121251379680 – Supplemental material for Quantification of anterior scleral thickness in Posner–Schlossman syndrome
Supplemental material, sj-docx-1-smo-10.1177_20503121251379680 for Quantification of anterior scleral thickness in Posner–Schlossman syndrome by Mu Li, Dan Zhao, Xiaoqin Yan and Zhiqi Chen in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121251379680 – Supplemental material for Quantification of anterior scleral thickness in Posner–Schlossman syndrome
Supplemental material, sj-docx-2-smo-10.1177_20503121251379680 for Quantification of anterior scleral thickness in Posner–Schlossman syndrome by Mu Li, Dan Zhao, Xiaoqin Yan and Zhiqi Chen in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121251379680 – Supplemental material for Quantification of anterior scleral thickness in Posner–Schlossman syndrome
Supplemental material, sj-docx-3-smo-10.1177_20503121251379680 for Quantification of anterior scleral thickness in Posner–Schlossman syndrome by Mu Li, Dan Zhao, Xiaoqin Yan and Zhiqi Chen in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121251379680 – Supplemental material for Quantification of anterior scleral thickness in Posner–Schlossman syndrome
Supplemental material, sj-docx-4-smo-10.1177_20503121251379680 for Quantification of anterior scleral thickness in Posner–Schlossman syndrome by Mu Li, Dan Zhao, Xiaoqin Yan and Zhiqi Chen in SAGE Open Medicine
Footnotes
Ethical considerations
This study was approved by the ethical committee of Tongji Hospital, Huazhong University of Science and Technology (TJ-IRB20201024).
Consent to participate
Written informed consent was obtained from the study subjects.
Consent for publication
Written informed consent was obtained from the study subjects.
Author Contributions
Conceptualization: ML, DZ, XY, and ZC; Methodology: ML, DZ, XY, and ZC; Software: ML; Validation: ML, DZ, XY, and ZC; Formal Analysis: ML, DZ, and XY; Investigation: ML, DZ, XY, and ZC; Resources: XY and ZC; Data Curation: ML and DZ; Writing – Original Draft Preparation: ML and DZ; Writing – Review & Editing: XY and ZC; Visualization: ML, DZ, XY, and ZC; Supervision: ZC; Project Administration: ZC; Funding Acquisition: ML, XY, and ZC.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Grant No. 82000893, 81800821) and Natural science foundation of Hainan Province (Grant No. 823MS175).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
