Abstract
Spectral domain optical coherence tomography has changed the way we view retinal diseases by providing us with high-resolution, cross-sectional images. It is very important for diagnosing and keeping a track of cystoid macular oedema, especially when it is caused by vascular conditions such as branch retinal vein occlusion. A 47-year-old man with poorly controlled type 2 diabetes mellitus presented with reduced visual acuity in his left eye. Amsler grid testing showed that the patient had metamorphopsia. The fundus examination indicated inferotemporal branch retinal vein occlusion, necessitating additional assessment. Spectral domain optical coherence tomography confirmed cystoid macular oedema with increased macula thickness and disruption of the external limiting membrane and ellipsoid zone, indicating disorganization of the outer retinal layers with photoreceptor involvement. There was also a partial posterior vitreous detachment. The changes in the outer retinal layers are important optical coherence tomography biomarkers that are linked to reduced visual acuity. The patient received an intravitreal injection of ranibizumab along with an adjunctive course of topical nepafenac (a nonsteroidal anti-inflammatory drug). During follow-up, spectral domain optical coherence tomography showed resolution of macular oedema, and the best-corrected visual acuity improved from 20/80 to 20/40, and the metamorphopsia was diminished. A small intraretinal cyst close to the fovea, hard exudates, and progressing posterior vitreous detachment were, however, seen. This case underscores the significance of systemic metabolic dysfunction, the prognostic utility of optical coherence tomography biomarkers, and the necessity of therapeutic strategies in the management of branch retinal vein occlusion.
Keywords
Introduction
Optical coherence tomography (OCT) is a non-invasive diagnostic imaging technique that utilizes low-coherence interferometry to acquire serial cross-sectional images of ocular structures. OCT uses near-infrared light with a wavelength of 830 nm to measure the optical reflectivity of various tissues. 1 In clinical practice, OCT offers several advantages: it enables imaging without tissue damage, provides high resolution (~10 μm), allows precise visualization of microstructural changes, does not require contrast agents, and achieves imaging depths >1 mm. It is commonly used to differentiate between the retinal layers and assess the structural pattern of the macula.2,3
Spectral domain (SD)-OCT, a more advanced variant, offers superior resolution and speed, enhancing visualization of retinal morphology. It enables detailed classification of diabetic macular oedema (MO) into patterns such as diffuse retinal thickening, MO, subretinal fluid accumulation, and abnormalities at the vitreomacular interface, aiding in better diagnosis and personalized treatment planning. 4
Cystoid MO (CMO) is a common retinal condition that can result from various ocular disorders, particularly central retinal vein occlusion (CRVO) and branch retinal vein occlusion (BRVO). 5 BRVO refers to occlusion of a retinal venule and is most frequently observed in the superotemporal quadrant (58%–66%), compared with the inferotemporal quadrant (29%–34%). 6 The incidence of BRVO increases with age and is less common in individuals aged 40–49 years (0.38%) than in those aged 70–79 years (1.66%). 7 CMO associated with BRVO is primarily attributed to breakdown of the inner blood-retinal barrier, secondary to venous obstruction and vascular endothelial growth factor (VEGF) mediated vascular permeability, resulting in leakage of plasma and proteins into the macular region. This results in the accumulation of extracellular fluid within the outer plexiform and inner nuclear layers, leading to the formation of multiple intraretinal cystoid spaces. 8 On OCT, these appear as hyporeflective areas that may coalesce into larger cystic spaces within the macula, making OCT a critical tool for diagnosing, monitoring treatment response, and assessing the prognosis of CMO.
Current advancements using deep learning-guided OCT analyses have identified emerging imaging OCT biomarkers such as disorganization of the retinal outer layers (DROL), defined by disruption of the external limiting membrane (ELM), ellipsoid zone (EZ), and retinal pigment epithelium (RPE), indicating outer retinal structural compromise and photoreceptor involvement. This study also demonstrates that retinal vein occlusion exhibits significantly higher rates of outer retinal layer obscuration, suggesting distinct structural alterations in retinal vein occlusion-related MO. 9
Patient presentation
A 47-year-old male patient reported for an eye examination, with a complaint of a sudden onset of blurred vision in the left eye. There was no history of ocular trauma or prior eye surgery. Systemic evaluation revealed a known history of poorly controlled type 2 diabetes mellitus (HbA1c: 11.9%) and pancreatitis, for which the patient was on regular medication. Baseline laboratory investigations, including complete blood count and metabolic profile, were within normal limits. Coagulation parameters, such as prothrombin time, activated partial thromboplastin time, and international normalized ratio, were within reference ranges.
Upon ocular examination, the right eye had a best-corrected visual acuity (BCVA) of 20/20, N6, while the left eye measured 20/80, N12. Considering the decreased vision in the left eye, qualitative central visual field screening was performed using Amsler’s grid, which showed normal results in the right eye. However, in the left eye, distortion of lines was noted, suggestive of metamorphopsia. Pupillary examination revealed no relative afferent pupillary defect. Intraocular pressure measured was 14 mmHg in the right and 12 mmHg in the left, both within the normal range. Slit-lamp examination of the anterior segment was within normal limits. Crystalline lens changes were noted, specifically grade 1 nuclear sclerotic cataract in both eyes.
Posterior segment examination showed mild pigmentary changes at the macula along with the presence of collateral vessels in the right eye, whereas in the left eye, findings were indicative of an inferotemporal BRVO, marked by hard exudates near the macula, collateral vessels, and dot-blot haemorrhages, raising suspicion of associated MO (Figure 1). These findings correlated with the reduced visual acuity, and an OCT was advised to further evaluate and confirm the presence and extent of MO.

Fundus images obtained using optical coherence tomography. (a, b) Five-line raster scans with the green lines indicating the scan positions, and the blue line indicates the selected scan line. (c, d) Macular cube 512 × 128 scans. (a) RE shows collateral vessels (blue circle). (b) LE reveals inferotemporal branch retinal vein occlusion (red box). (c) RE displays foveal pigmentary changes (green circle). (d) LE shows a magnified view of haemorrhages and the venous occlusion site (orange box).
OCT revealed subtle structural irregularities at the level of the RPE in the right eye, with no signs of MO (Figure 2). The left eye showed features consistent with CMO, characterized by abnormal accumulation of extracellular fluid forming multiple cystic spaces, increased macular thickness from the internal limiting membrane (ILM) to the RPE due to oedema. Disruption of the ELM and EZ represents important OCT imaging biomarkers that indicate DROL with potential photoreceptor involvement. A partial posterior vitreous detachment (PVD) was also noted (Figures 2 and 3).

Macular cube images of RE. (a) Subtle RPE alterations (brown arrows). (b) Irregularities in the RPE (brown arrows). (c) Normal macular thickness. LE: (d, e) reveal cystoid macular oedema (white arrow), external limiting membrane disruption (orange arrow), and partial posterior vitreous detachment (yellow arrow). (f) Increased macular thickness with pink colour coding denoting oedema (blue box).
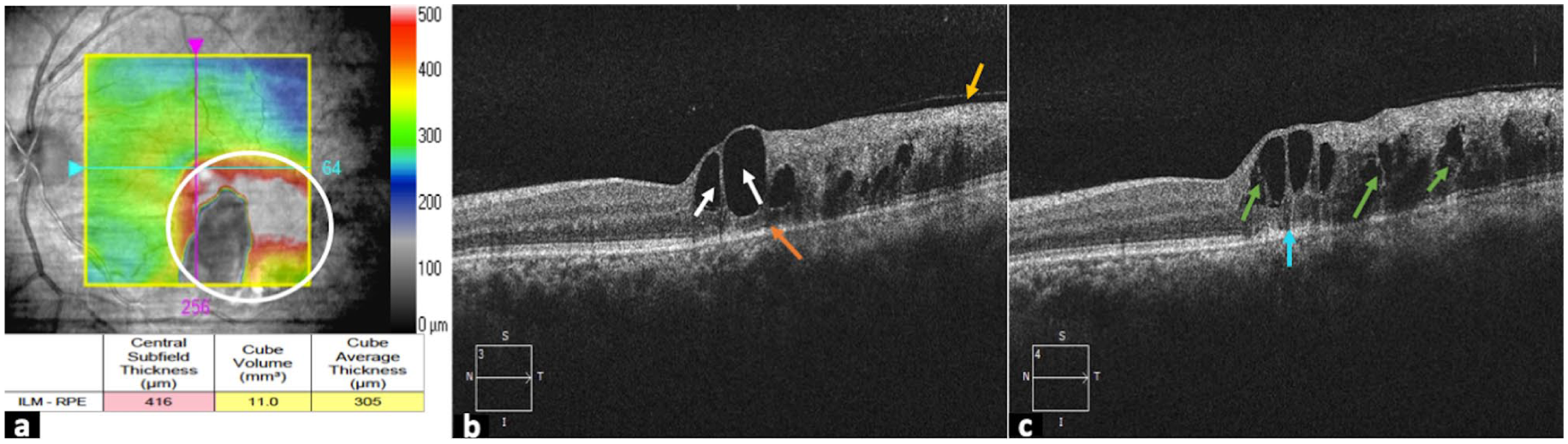
(a) Pre-treatment optical coherence tomography colour-coded internal limiting membrane to retinal pigment epithelium thickness maps overlaid on the fundus image of the left eye show an oedematous state, marked by red colour code (white circle), with central subfield thickness of 416 μm. (b) Reveals cystic hyporeflective spaces (white arrows), disruption of the external limiting membrane (orange arrow), and posterior vitreous detachment (yellow arrow). (c) Disruption of the ellipsoid zone (blue arrow) and retinoschisis (green arrow).
Management and outcome
The patient was immediately advised to undergo, and subsequently received, an intravitreal injection of ranibizumab, an anti-VEGF, in the left eye. He was further prescribed nepafenac eye drops, a topical nonsteroidal anti-inflammatory drug (NSAID), to be used three times daily for the left eye, and lubrex eye drops four times daily for both eyes for 4 weeks, along with advice for strict glycaemic control.
At the 4-week follow-up, the BCVA was 20/20, N6 in the right eye, and had improved to 20/40, N8 in the left eye. Amsler’s grid testing showed a marked reduction in metamorphopsia in the left eye compared to the initial visit. The patient also reported symptomatic improvement. To evaluate treatment outcomes and prognosis, follow-up OCT was performed. It revealed resolving CMO with the absence of cystic spaces at the macula, improved foveal contour, and decreased macular thickness from the ILM to the RPE compared to the prior oedematous state. Importantly, there was partial restoration of ELM and EZ, suggesting improvement in outer retinal integrity. Although a small residual intraretinal cyst near the fovea, persistent hard exudates, and progressive signs of PVD were still observed in the left eye (Figure 4), the overall structural and biomarker profile showed significant recovery. He was further advised to continue the same eye drops for 4 weeks and return for follow-up.
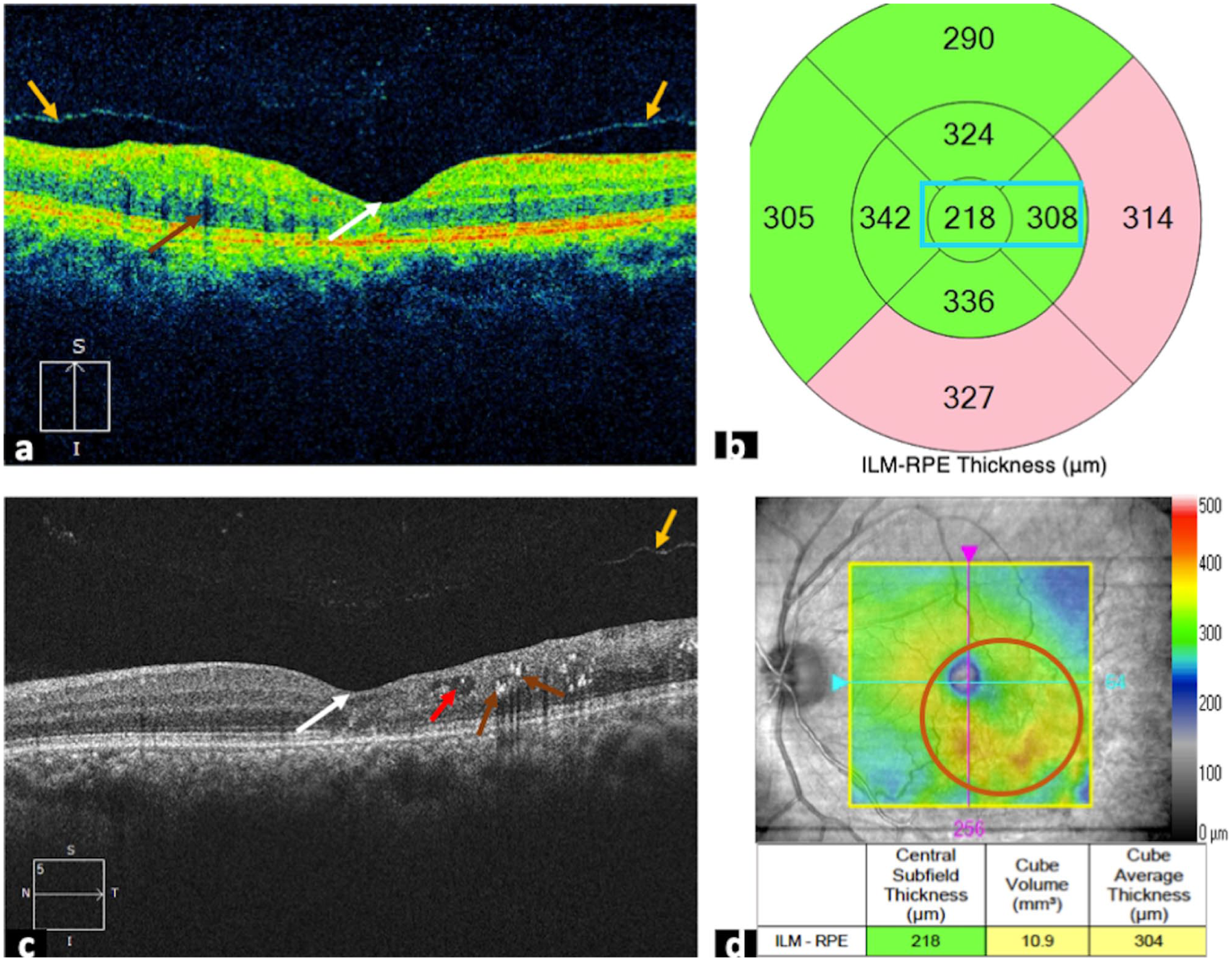
Post-treatment optical coherence tomography macular cube images of the left eye. (a) Reveals improved foveal contour (white arrow), hard exudates shadowing (brown arrow), and advancing posterior vitreous detachment (yellow arrows). (b) Green colour coding indicating normal macular thickness at the centre and adjacent quadrant (blue box). (c) No cystic spaces at the central macula region (white arrow), a small intraretinal cyst (red arrow), hard exudates (brown arrows), and progressing posterior vitreous detachment (yellow arrow). (d) Highlights the fundus image of the resolving site of branch retinal vein occlusion and oedema (orange circle).
Discussion
CMO is a sight-threatening condition that adversely affects visual outcomes and may develop secondary to various retinal pathologies, including diabetic retinopathy, retinal vein occlusion (CRVO and BRVO), age-related macular degeneration, and epiretinal membrane.4,10 BRVO, being the second most common retinal vascular disorder after diabetic retinopathy, is typically associated with systemic vascular risk factors such as hypertension, hyperlipidaemia, and diabetes mellitus.11,12 Although it predominantly affects older individuals, its occurrence in younger patients warrants evaluation for underlying hypercoagulable or thrombophilic states.
In this case, comprehensive laboratory investigations revealed normal coagulation parameters, and no evidence of systemic hypercoagulability was identified. Given the patient’s age (47 years), an age group with a lower prevalence of BRVO, the presence of poorly controlled diabetes mellitus (HbA1c: 11.9%) likely played a significant contributory role. Chronic hyperglycaemia induces endothelial dysfunction, increased vascular permeability, and microvascular damage, thereby predisposing to venous occlusive events.7,12 Inferotemporal BRVO, as observed in this case, may lead to macular involvement when the occluded venous branch drains the macular circulation, thereby increasing the risk of CMO and visual impairment. 13
OCT, particularly SD-OCT, is considered the gold standard for evaluating retinal layers and the macula. 14 Its high-resolution cross-sectional imaging enables accurate detection and detailed visualization of MO, as demonstrated in this case. The OCT report provides a comprehensive scan analysis, including a colour-coded ILM–RPE thickness map superimposed on the fundus image (Figure 3(a)), where warmer colours highlight areas of oedema. A thickness map (Figures 2(c), (f), and 3(b)) illustrates average retinal thickness values across the central 1 mm zone and two concentric rings (3 and 6 mm), each divided into four quadrants. The average ILM–RPE thickness is presented both numerically and as colour codes for each subfield. In this case, retinal thickness in the left eye ranged from 416 to 394 μm within the 1–6 mm zones, indicating thickening of the macular region. Additionally, both the inner and outer rings showed increased thickness values compared to the right eye. The central region appeared pink on the colour-coded map, consistent with significant MO.
The greyscale OCT scan of the left eye (Figure 3(b) and (c)) shows multiple well-defined hyporeflective cystic spaces within the central macula, consistent with CMO. These intraretinal cystic spaces extend inferotemporally across the scan, indicating fluid accumulation within the retinal layers due to impaired venous outflow from obstructive venous retinopathy. 5 The cystoid spaces are filled with serous fluid and are seen predominantly in the inner and outer retinal layers, especially in the outer plexiform layer, where fluid often accumulates in a petaloid configuration.8,10 In addition to these findings, several OCT-based imaging biomarkers were evident in this case. Disruption of the ELM and EZ was observed, indicating outer retinal structural compromise and potential photoreceptor involvement. Integrity of these outer retinal layers has been recognized as an important prognostic biomarker in MO secondary to retinal vascular disorders. Recent deep learning-guided OCT analyses have further highlighted DROL and increased outer layer obscuration as distinguishing biomarkers in retinal vein occlusion. 9 Moreover, the integrity of the photoreceptor layer has been shown to correlate more closely with the visual function, providing a more direct indicator of functional impairment. 15 In our patient, the presence of ELM and EZ disruption likely contributed to the initial reduction in visual acuity, while the subsequent resolution of oedema and improvement in vision may suggest partial resolution of outer retinal layers.
OCT has the highest accuracy for detecting CMO compared with other investigative procedures and is useful for monitoring retinal thickness both before and after treatment, offering qualitative and quantitative analysis.14,16 The degree of visual acuity loss in MO depends on several factors such as macular thickening, duration of oedema, macular capillary perfusion, and photoreceptor damage. 11 In this case, the patient’s BCVA was 20/80 prior to treatment, indicating moderate visual impairment. Combination management was indicated with intravitreal injection of ranibizumab along with a course of topical NSAID. Although BRVO‑related oedema is largely VEGF‑driven, inflammation also contributes. Prostaglandins generated via cyclooxygenase-2 (COX-2) amplify VEGF and increase vascular permeability. 17 Inhibiting COX-2 with NSAIDs can therefore reduce prostaglandin-mediated leakage and provide adjunctive benefit in resolving oedema.18,19
The selection of anti-VEGF (ranibizumab) was based on its established efficacy in the treatment of BRVO-related MO. Comparative studies have demonstrated that ranibizumab and bevacizumab achieve similar visual and anatomical outcomes, while aflibercept has also shown proven effectiveness.20,21 Although bevacizumab is widely used off-label due to its lower cost, ranibizumab was chosen in this case because of its immediate availability and well-established clinical evidence base. The combination treatment approach with topical NSAID and anti-VEGF therapy in this case may have contributed to the reduction of inflammatory-mediated vascular permeability and MO, as evidenced on follow-up, with BCVA improving to 20/40, and central subfield thickness reducing to 219 μm (Figure 4), reflecting favourable anatomical and functional outcomes. The patient was further scheduled for continued monitoring, as many BRVO patients require repeat injections, as oedema often recurs within a few months, so ongoing imaging is necessary.
The present case also highlights the importance of OCT in detecting and tracking the progression of PVD. Although PVD is commonly age-related, systemic conditions such as diabetes mellitus, as seen in this case, can contribute to earlier or abnormal onset of PVD. Additionally, PVD has been observed alongside retinal vein occlusions and may have a contributing role in the development or persistence of CMO through vitreomacular traction. 22 Clinical studies have shown that eyes beginning with stage 2 or 3 PVD tend to progress to higher stages over time. By contrast, some cases starting at stage 1 remain stable, while others eventually show progression. 13 In this patient, stage 1 PVD was initially observed, and subsequent follow-up OCT imaging showed signs of progression, highlighting the importance of regular imaging in monitoring disease dynamics (Supplemental Material).
Conclusion
This case highlights the occurrence of CMO secondary to BRVO in a relatively young patient with poorly controlled diabetes mellitus, underscoring the role of systemic metabolic dysfunction in retinal vascular events. OCT imaging revealed outer retinal biomarkers, including ELM and EZ disruption, which correlated with visual impairment. Treatment with anti-VEGF and adjunctive NSAID led to the resolution of MO and marked visual improvement. This case emphasizes the importance of systemic risk factor control, OCT biomarker analyses, and therapeutic strategies for optimizing outcomes in BRVO.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X261436550 – Supplemental material for Imaging cystoid macular oedema secondary to branch retinal vein occlusion with spectral domain optical coherence tomography: A case report
Supplemental material, sj-docx-1-sco-10.1177_2050313X261436550 for Imaging cystoid macular oedema secondary to branch retinal vein occlusion with spectral domain optical coherence tomography: A case report by Samuel Livingstone Kumaran, Gokulakrishnan Kannaki Renuga Sri, Beena Esther Thomas and Shiju Monickaraj Selvister in SAGE Open Medical Case Reports
Footnotes
ORCID iDs
Ethical considerations
This research has received approval from the Institutional Ethics Committee, SRIHER(DU), August 20, 2025, with approval number CSP/25/AUG/166/429.
Consent to participate
Informed consent was obtained from the patient for the use of their retinal OCT images. Only non-identifiable imaging data for the presented case and no personal or demographic information is disclosed.
Author contributions
Samuel Livingstone Kumaran: concepts; design; definition of intellectual content; literature search; clinical studies; data acquisition; data analysis; manuscript preparation; manuscript editing; manuscript review. G. K. Renuga Sri: concepts; design; definition of intellectual content; literature search; clinical studies; data acquisition; data analysis; manuscript preparation; manuscript editing; guarantor. Beena Esther Thomas: concepts; design; definition of intellectual content; clinical studies; data acquisition; data analysis; manuscript editing. Shiju Monickaraj Selvister: concepts; design; definition of intellectual content; clinical studies; data acquisition; manuscript review. All authors contributed to the preparation of this case report and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data that support the findings of this case report, including anonymized clinical OCT images, are available from the corresponding author upon request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
