Abstract
Objectives:
Legionella pneumophila is a waterborne bacterium known to cause Legionnaires’ disease, a severe form of pneumonia particularly affecting older and immune compromised individuals. This study evaluates the level of risk awareness, clinical practices, and barriers to optimal management of nosocomial Legionnaires’ disease in Ethiopia’s two largest hospitals.
Methods:
We employed a cross-sectional survey design, utilizing a self-administered questionnaire to collect data from 374 practicing physicians at two specialized hospitals in Addis Ababa.
Results:
A total of 324 questionnaires were returned, yielding a response rate of 86.6%. Eighty percent (n = 270) reported some level of awareness of Legionnaires’ disease as a clinical condition. However, 43% of the physicians were unaware of the association between hospital water systems and the risk of nosocomial Legionnaires’ disease. Fifty-seven percent of the respondents indicated the absence of technical guidelines to inform the diagnosis and management of nosocomial Legionnaires’ disease. Most physicians relied on clinical assessment (57%, n = 51) for diagnosing the disease, citing diagnostic barriers (68%, n = 140) and perceived low reliability of existing laboratory tests as key challenges. Notably, physicians with limited knowledge about Legionnaires’ disease were associated with a 79% reduction in accurate diagnosis of cases. Additionally, poor hospital water quality (43%) and inadequate infection prevention and control measures were identified as significant contributors to the persistence of waterborne hospital-acquired pneumonia.
Conclusions:
This study highlights physicians’ limited awareness of the risks posed by hospital water systems in the transmission of Legionnaires’ disease. Additionally, the diagnosis of Legionnaires’ disease is impeded by the lack of clinical guidelines and specific diagnostic testing capabilities. These findings underscore the urgent need to revise hospital-acquired pneumonia protocols, strengthen infection prevention and control guidelines, and enhance hospital water management practices.
Keywords
Introduction
In 1976, a pneumonia outbreak in Philadelphia led to the identification of a novel illness, subsequently named Legionellosis. 1 Extensive investigations revealed that Legionella pneumophila, a bacterial pathogen within the family Legionellaceae, was the etiological agent of Legionellosis. 2 Since then, Legionellosis cases and outbreaks have been uncovered in multiple countries and settings. 3 Further research has shown that Legionella bacteria, encompassing various species, commonly inhabit diverse water environments, including fresh water, warm water, water distribution systems, domestic, or institutional water systems. 4 Later research confirmed the primary mode of human transmission is inhalation of mist or swallowing water into the lungs. 5
Human infection with Legionella bacteria can manifest in various conditions, from a mild flu-like illness known as Pontiac fever to the more severe and potentially lethal pneumonia called Legionnaires’ disease. 6 The literature classifies Legionella-associated pneumonia based upon the location of the human-acquired pathogen. These are community-acquired, hospital-acquired, or travel-associated. 4 Hospital-acquired or nosocomial Legionnaires’ disease (LD), as the focus of this study, is a condition that largely impacts patients after staying 48 or more hours in a healthcare environment. 7
A systematic review estimated the global sero-prevalence of LD in the general population to be 10.5% in 2020. 8 Studies in the United States reported about 10,000 cases in 2019. 9 The EU noted 14,463 cases in 2023. 10 South Africa determined 93 cases 2018 to2020. 11 While fresh water samples in Ethiopia showed over 30% positivity, 12 there are no reports on the prevalence of LD in humans. The overall global trend reveals a rising incidence of LD, suggesting a growing impact of this infection that necessitates further investigation into its causes and effective management strategies. In the United States, the incidence increased exponentially between 2010 and 2012. 13 A Japanese trend analysis showcased an increase from 0.0004 cases per 100,000 population in 1999 to 1.37 cases in 2017. 14 The US Centers for Disease Control and Prevention estimates a 10% mortality rate from LD complications. In Europe, hospital-acquired pneumonia (HAP) is a leading cause of disability-adjusted life years (DALYs), with LD contributing up to 5% of case DALYs. 15 Lung failure, shock, and organ failure are common complications. 16
Nosocomial LD is primarily linked to patient exposure to hospital water systems contaminated with Legionella spp. Numerous studies have established the link between hospital water systems and LD cases.17–20 Susceptible hosts can become infected by inhaling droplets or aspirating water from sources such as showerheads, sink faucets, decorative water features, hot water tanks, heaters, and cooling towers, posing significant health risks to seriously ill hospital patients.21,22 Hospital water systems, especially in facilities facing water supply or quality challenges, can favor Legionella proliferation, posing a significant transmission risk to susceptible patients.23–25 The risk is heightened in health facilities lacking standardized water safety or infection prevention practices to prevent health care-associated infections, thereby increasing the likelihood of pathogen growth and transmission.26,27 Many low and middle-income countries (LMICs) lack LD surveillance systems, leading to scarce data on its magnitude. This data gap obscures the true global burden.28,29 Subsequently, healthcare workers in LMICs often lack the evidence needed for LD prevention and management. Clinicians, in particular, have limited evidence to understand case patterns or link cases to risk factors. This study examines physicians’ levels of awareness of the risk, clinical practices, and barriers related to LD prevention, diagnosis, and management in Ethiopia’s two largest hospitals.
This study hypothesized that physician awareness regarding LD risk factors is suboptimal, current clinical practices related to its diagnosis and management do not consistently align with international evidence or best practices, and significant barriers hinder effective LD prevention, diagnosis, and management.
To our knowledge, this is the first study in Ethiopia to explore physicians’ perspectives on hospital-acquired LD. The findings will provide foundational data for future research, capacity-building, and policy development to enhance LD prevention, diagnosis, and management. This study aims to raise awareness among healthcare providers and policymakers about LD as an under-recognized public health threat in Ethiopia.
Methods
Study design
This facility-based cross-sectional study was conducted from February 2024 to April 2024. A sample size of 374 was calculated using Epi InfoTM 7.1.1.14, based on a single population proportion with an anticipated prevalence of 63.7% 30 and a 95% confidence interval.
Study setting
The study population comprised physicians practicing in Tikur Anbessa Specialized Hospital (TASH) and St. Paul’s Hospital Millennium Medical College (SPHMMC), two of Ethiopia’s largest referral hospitals. Each facility serves over 2000 patients daily, primarily with referrals from across the country. Physicians not currently practicing or those working outside these facilities were excluded from the study.
TASH was established in 1961 as Ethiopia’s primary tertiary referral center, providing specialized and comprehensive medical care. It is affiliated with Addis Ababa University Medical Faculty and serves as the largest teaching hospital in the country. It has over 950 staff and a 700-bed capacity.
SPHMMC, established in 1968 and governed by a board under the Federal Ministry of Health, is a rapidly growing teaching hospital in terms of services and patient volume. It has over 2800 staff with a 700-bed capacity.
Variables and measurement
Dependent variables: The study evaluates three primary variables: (1) Awareness of the risk of hospital water, defined as the percentage of physicians who recognize the potential role of hospital water in transmission of nosocomial LD; and (2) Diagnosed LD, which measures the percentage of physicians who claim to have diagnosed a case of LD in their clinical practice.
Independent variables: The study collected data on several demographic and professional variables: (1) Age, measured in years and treated as a continuous variable; (2) Gender, categorized as male or female; (3) Years of Experience, reflecting the number of years the physician has been practicing in their field; (4) Department, which specifies the area of medicine or healthcare the professional works in (e.g., Internal Medicine, Infectious Disease, Emergency Medicine). (5) Qualification refers to the highest educational degree or professional certification held by the physcians.
Data collection tool and procedures
Data were collected using a self-administered quantitative questionnaire. This instrument comprised closed-ended and open-ended questions covering the demographics, the national LD and HAP context, water linkage, risk factors awareness, diagnostic and management experiences, diagnostic barriers, opportunities, and perceived needs. The data collection tool was adopted from similar research conducted on LD and HAP and customized to the Ethiopian context.31–34 To ensure reliability and clarity, the questionnaire was pretested on 10% (n = 40) of the study population in a similar hospital within Addis Ababa, and necessary adjustments were made before data collection.
Data processing and analysis
Completed questionnaires were checked for completeness and consistency, entered into Epi Info 7.1.1.14, transferred to Microsoft Excel, and analyzed using IBM Statistical Package for Social Sciences (SPSS) software Version 26. Descriptive and inferential analyses were performed afterdata import, cleaning, and organization. Chi-square tests assessed associations between categorical variables, and binary logistic regression was applied to identify factors contributing to outcome variables. A p-value of less than 0.05 was considered statistically significant.
Bias mitigation measures: Statistical weighting was used to adjust the analysis and better reflect the target population. The study focused on recent experiences rather than long-term recollections to minimize rcall bias. Age, Years of Experience, Department, and Qualification are controlled for confounding during the logistic regression analysis.
Ethical considerations: Addis Ababa University College of Health Sciences (IRB approval No. 077/21/SPH) granted institutional reveiw board approval. Study participants signed written informed consent prior to data collection.
Results
Sociodemographics
Of the 374 physicians who consented, 324 returned completed questionnaires, yielding an 86.6% response rate. Internal Medicine accounted for the largest proportion of respondents (49%), followed by Emergency Department (26.9%), and Anesthesiology (9.9%). Most of the study participants were resident physicians, comprising 68.8% of the sample. Specialists constituted 21% of the respondents, while general practitioners comprised 9.3%. A significant proportion had 2–5 years (66.4%) of practice, and 12% were female doctors (12%). See Table 1 for detailed demographic characteristics.
Physician responses regarding the risk of hospital water for LD disaggregated by demographic characteristics, at Tikur Anbessa and St. Paul’s Hospitals, Addis Ababa, Ethiopia, February 2024–April 2024.
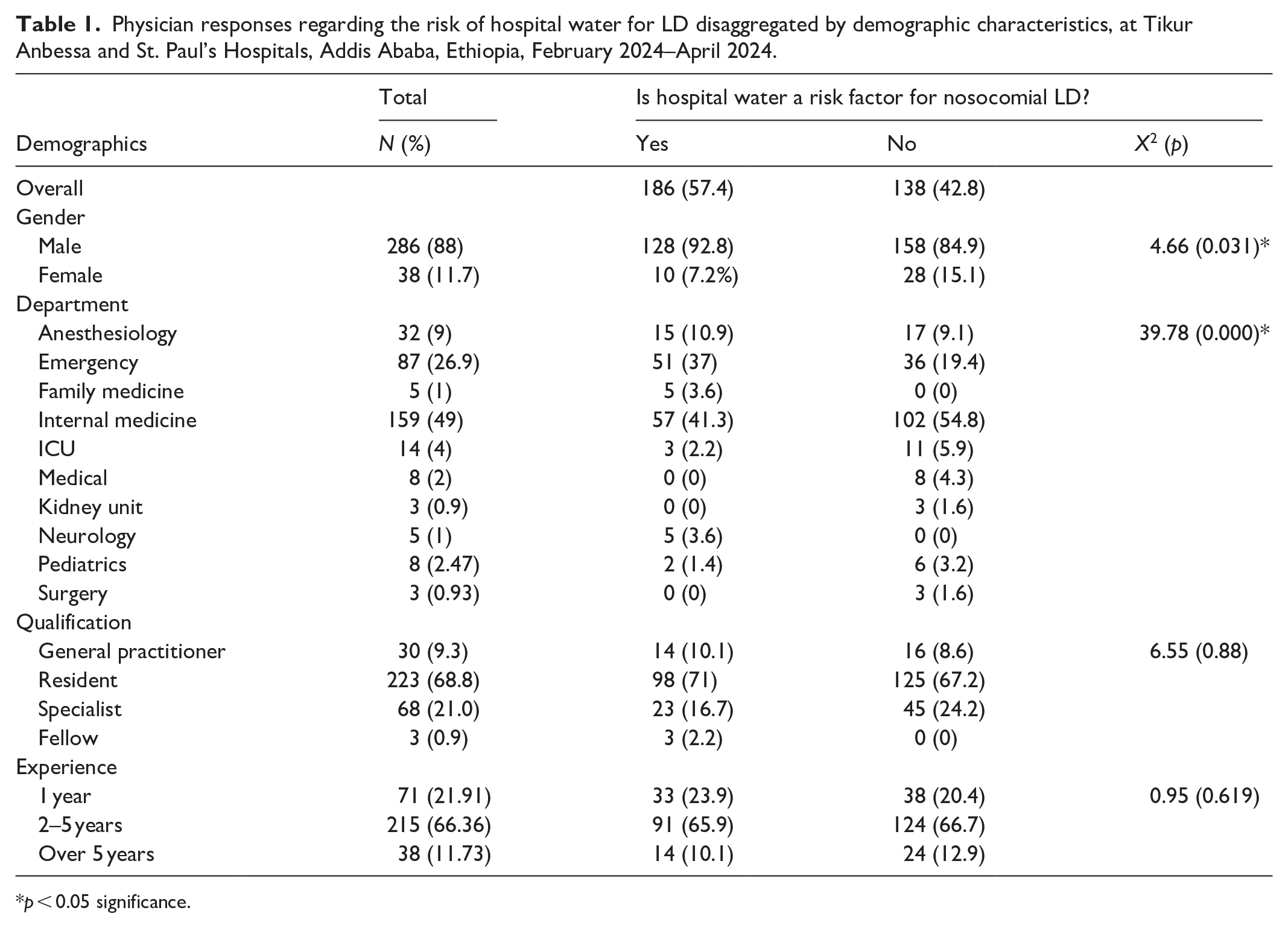
p < 0.05 significance.
Risk awareness
Eighty percent of the physicians (n = 270) asserted having some level of awareness of LD as a clinical condition, while 17% (n = 54) had never heard of it. Eighty-six percent (n = 278) of the physicians reported no prior engagement in LD-related discussions, while 14% (n = 46) indicated participation in such clinical conversations. Ninety-three percent (n = 301) believe there should be opportunities to discuss these issues.
Regarding the hospital departments at high risk of nosocomial pneumonia, 43% identified Intensive Care Units (ICUs), 13% Internal Medicine, and 13% Emergency Department. When asked about cases at risk, 20% (n = 54) cited critically ill patients, 13% immune-compromised patients, and 11% patients with malignancies.
Fifty-seven percent (n = 187) of the physicians were aware that hospital water can be a risk factor for nosocomial pneumonia. In contrast, 13% (n = 42) do not consider it a risk, and 30% (n = 96) are unaware of this association. Thirty-nine percent (n = 97) cited aerosolized water as the main mode of transmission, followed by showering (32%) and drinking water (26%). Regarding the most encountered water-borne microorganisms of nosocomial pneumonia, P. aeruginosa (58%), K. pneumoniae (15%), and Acinetobacter species (11%) were the top three pathogens reported by the physicians.
Our binary logistic regression analysis revealed that a physician’s hospital department (p = 0.003) and gender (p = 0.049) significantly predicted their recognition of hospital water as a risk factor for LD. On the other hand, qualification (p = 0.590) and experience (p = 0.403) were not significant predictors. Further details are available in Table 1.
When asked about the factors perpetuating waterborne nosocomial pneumonia, 43% (n = 24) cited shortage of water and infection prevention and care (IPC) supplies, followed by poor-quality patient samples (36%), and avoidance of high healthcare expenditures (9%).
Physicians were asked about their awareness of nosocomial LD risk factors. These questions covered several risk factors, including old age, ICU admission, treatment failure, history of alcohol use, recent travel history, preexisting respiratory illnesses, and chronic diseases. The responses are summarized in Figure 1.

Percentage of awareness on risk factors for LD by physicians at Tikur Anbessa and St. Paul’s Hospitals, Addis Ababa, Ethiopia, February 2024–April 2024.
Clinical practices
Regarding the availability of clinical guidelines for LD management, 57% of the physicians reported the lack of such resources. In addition, 92% (n = 296) indicated the lack of a Legionella testing algorithm, 70% (n = 227) confirmed the absence of an LD surveillance system.
Physicians were asked to describe access to pneumonia testing. One-third (33.5%, n = 162) indicated the presence of general sputum or tracheal aspirate culture test, 24.4% (n = 118) chest radiology, and about 2 in 10 cited hematology (19%, n = 91) tests.
All participating physicians (100%) concurred that their hospitals lacked access to LD-specific laboratory tests, specifically confirming the unavailability of sputum or bronchoalveolar lavage culture, urinary antigen testing, and molecular diagnostic methods.
Twenty-eight percent (n = 91) of physicians reported having made a presumptive diagnosis of nosocomial LD in at least one patient. This diagnosis was based on clinical symptoms in 57% of these cases, clinical and radiological findings in 21%, and clinical and laboratory results in 22%. Approximately 4.6% (n = 15) of the physicians reported having encountered a confirmed case of LD in the past.
Approximately 17% (n = 50) of the physicians experienced hospital-acquired pneumonia outbreaks of undetermined pathogens, with 3% (n = 11) suspecting LD involvement. Twenty-two percent reported an average of three nosocomial pneumonia cases at a given time. In managing nosocomial pneumonia cases, 76% (n = 261) prioritized patient outcomes, 12% reduced antibiotic resistance, 9% adverse effect suppression, and 2% focused on infection reduction. Azithromycin (22%) and vancomycin (20%) were the most commonly prescribed drugs, leading to a positive prognosis in 79% of the treated cases (n = 38).
Holding other variables constant, general practitioners had 87% lower odds of diagnosing LD than residents. See Table 2 for details on the impact of qualification on Legionella diagnosis.
Physicians’ responses regarding “If ever diagnosed LD” and “need for LD training” disaggregated by demographic characteristics, at Tikur Anbessa and St. Paul’s Hospitals, Addis Ababa, Ethiopia, February 2024–April 2024.

p < 0.05 significance.
Logistic regression analysis showed that physicians’ department, qualification, experience, and gender did not predict legionella diagnosis. However, qualification was a significant predictor of physicians’ training needs on LD (p = 0.004). Further details can be found in Table 3.
Physicians’ responses regarding “If ever diagnosed LD” and “need for LD training” disaggregated by demographic characteristics, at Tikur Anbessa and St. Paul’s Hospitals, Addis Ababa, Ethiopia, February 2024–April 2024.

p < 0.05 significance.
Barriers
Understanding barriers to nosocomial pneumonia diagnosis and management is crucial for effective interventions. The primary reason for missed nosocomial pneumonia cases was a reduced index of suspicion (53%, n = 129), absence of diagnostic tests (26%), and long test turnaround time (7%). A significant obstacle to ensuring timely and targeted HAP treatment is the extended waiting period for the necessary diagnostic testing. Further details are illustrated in Figure 2.

A List of barriers to targeted nosocomial pneumonia treatment reported by Physicians at Tikur Anbessa and St. Paul’s Hospitals, Addis Ababa, Ethiopia, February 2024–April 2024.
Physicians identified several key barriers hospitals should address to improve HAP diagnosis and management. These included a shortage of health care resources (22%), limitations in diagnostic capacity (23%), and low awareness among healthcare providers (28%).
Recognizing the urgent need to enhance waterborne nosocomial pneumonia management, 93% of the physicians recommended Legionella training (n = 301) for health workers. Specific needs included technical guidelines on nosocomial pneumonia with LD (48%, n = 86), contextualized case definitions (43%, n = 155), availability of specific diagnostic tests (30%), and surveillance system installation (9%).
Recommended strategies for improving waterborne HAP management were enhancing IPC practices and supplies (29%), increasing health workers’ awareness (27%), and developing diagnostic and treatment protocols (23%).
Discussion
At least 10 cases of LD per million population are estimated to occur annually in the high-income countries, while the burden in most LMICs remains largely unknown.13,35–37 The presence of surveillance systems allows for trend analysis, enabling prompt actions to limit transmission and lessen the impact on healthcare systems.28,38 However, healthcare workers remain the frontline in identifying and managing cases, irrespective of formal tracking. This study envisaged exploring the LD landscape in Ethiopia through interviews with physicians at two specialized hospitals.
Although there is a high level of awareness (80%) among physicians, and the mode of transmission is widely understood, the finding that over two out of five physicians could not establish a link between hospital water systems and nosocomial LD is a cause for concern. Our finding (57%) is higher than the 34% reported by Alaga et al. 39 in Michigan, where physicians’ knowledge of water as a risk factor for LD was comparatively lower. The disparity in these findings may be attributed to several factors. First, the higher prevalence of waterborne diseases in Africa may contribute to greater physician expertise in diagnosing and managing such conditions, including LD. Second, the limited availability of advanced diagnostic technologies in sub-Saharan Africa likely necessitates a stronger reliance on clinical skills and knowledge, potentially leading to increased awareness among healthcare providers.
We confirmed that the absence of technical guidelines on LD poses a major challenge in identifying cases. This finding aligns with the conclusion of Van Kenhove et al., 40 who stated that most sub-Saharan African countries lack essential technical resources, such as job aides, standard operating procedures, or case definitions for use by healthcare workers. This gap is likely to perpetuate poor identification and mismanagement of cases in LMIC.
Among the 28% of physicians who considered LD as a differential diagnosis, the majority did so based on clinical findings (57%, n = 51). This reliance was mainly driven by diagnostic barriers (68%, n = 140), which included the unavailability of appropriate laboratory tests, long test turnaround time, and poor-quality samples. Notably, a qualitative study in Switzerland demonstrated that even in settings with better access to laboratory facilities, some physicians held limited awareness of the available Legionella diagnostic tests. 31 This suggests that challenges in Legionellosis diagnosis are not solely confined to resource-limited environments. In the context of this study, it is crucial to highlight that neither the participating hospitals nor the national referral laboratory in Ethiopia offered any of the recommended laboratory tests for LD. The lack of diagnostic capacity stems from the fact that LD is not currently classified as a disease of public health importance in Ethiopia, 41 resulting in a gap in surveillance and diagnostic infrastructure.
Absence or poor quality of diagnostic facilities significantly influences treatment strategies for Legionellosis. Faced with these limitations, a substantial majority of physicians (96%, n = 311) reported resorting to empirical treatment approaches, deviating from targeted therapies. This reliance on empirical methods due to diagnostic shortcomings aligns with findings from a Malawian study, where 22% of physicians identified the need for improvements in laboratory service turnaround times. 42 Moreover, an Ethiopian survey revealed a broader concern regarding laboratory services, with nearly half of the physicians expressing dissatisfaction with the reliability and accuracy of laboratory test results from public hospitals. 43 These findings collectively underscore the critical need for strengthening diagnostic infrastructure and improving laboratory services to facilitate accurate diagnosis and targeted treatment of LD.
Our study revealed a statistically significant association between a physician’s likelihood of diagnosing Legionella cases and their department, residency status, and years of experience. Specifically, physicians within the Internal Medicine Department, those in residency training, and those with more extensive experience demonstrated a higher probability of diagnosing Legionella. This finding partially aligns with the conclusions of Ajmi et al., 44 who emphasized the strong association between physician experience and enhanced patient care. Furthermore, the evidence presented by Perez-Villadóniga et al. 45 supports our observation regarding resident physicians, highlighting their positive impact on hospital productivity, a finding consistent with our study’s results. However, it is noteworthy that perceived adequate clinical knowledge did not significantly correlate with the capacity to diagnose Legionella. This suggests that while knowledge is essential, it does not, in isolation, guarantee effective clinical practice. The translation of knowledge into accurate diagnoses and appropriate management necessitates the integration of practical skills and clinical competence.
Our study revealed that approximately one in five physicians had encountered hospital-acquired pneumonia outbreaks, and LD was “suspected” as one of the contributing causes in a subset of these instances. However, due to the absence of a notification system for LD, verification of these outbreaks through medical records was not possible. The absence of a surveillance system for a potentially preventable disease suggests that LD cases are likely being misdiagnosed in Ethiopia. Consequently, the true incidence and burden remain unclear due to a lack of a dedicated tracking system or population-based studies. This is further illustrated by the rapid identification and effective response to a recent LD outbreak in Poland in late 2023, which underscores the critical importance of implementing public health data collection methods. 46 The establishment or customization of existing systems should be a priority for Ethiopian health authorities, as they are essential for preventing unnecessary morbidity, mortality, and prolonged hospital stays. 47
Our study participants identified poor hospital water quality (43%) in conjunction with compromised IPC interventions as significant contributors to waterborne nosocomial pneumonia in Ethiopia. This situation is presumed to be exacerbated by the persistent water rationing practices in Addis Ababa, which have been in effect for several years. The regular municipal water cut-offs, lasting for multiple days each week, contribute to temporary or intermittent water stagnation within the plumbing systems of the two hospitals.48,49 This stagnation creates an environment conducive to the proliferation of waterborne pathogens, including L. pneumophila, within the hospital water systems. 50
Strengths and limitations
This study is the first to investigate LD—a potentially severe yet often overlooked infection—within the Ethiopian healthcare context, where data on this disease is limited. By focusing on physicians’ perspectives, the study provides insights into real-world clinical practices and challenges, helping to fill a significant knowledge gap. Additionally, its focus on nosocomial LD in LMICs offers novel and relevant findings that could contribute to improving hospital infection control in resource-limited settings. However, this study is subject to certain limitations. First, the reliance solely on interviews, without a diagnostic component to triangulate their claims, restricts the accurate determination of the LD burden. Second, the study’s confinement to two voluntary hospitals within the capital city potentially introduces a bias toward the generalizability of the study to all hospitals in Ethiopia. Lastly, the lack of in-depth qualitative exploration of physicians’ experiences and perceptions limits the nuanced insights that such an approach could provide, which quantitative methods might overlook.
Conclusion
This study aimed to illuminate the landscape of nosocomial LD in Ethiopia, focusing on the experiences of practicing physicians in two hospitals. Our findings revealed a good level of awareness regarding patient vulnerability and modes of transmission. However, awareness of the association of hospital water with nosocomial LD was limited, likely due to the absence of clinical guidelines and inadequate training, contributing to a low index of suspicion. Despite the nonspecific clinical presentation, some physicians suspected potential LD cases based on clinical conditions but could not verify the diagnosis due to the unavailability of Legionella diagnostics in the country. These observations suggest that LD cases are likely misclassified, or attributed to other pathogens in Ethiopia. The lack of resources for the purchase of diagnostic and treatment tools further hinders healthcare workers from accurately identifying and managing Legionella cases, potentially leading to preventable infections among hospitalized patients. Our literature review corroborated these challenges by demonstrating the absence of a diagnostic and treatment algorithm for LD, the unavailability of specific Legionella tests, and the lack of a water management program to guide healthcare workers in addressing hospital-acquired Legionella infections in Ethiopia.
To effectively combat LD in Ethiopia, immediate and multifaceted action is imperative across several fronts, including the development and dissemination of clinical guidelines, the enhancement of access to quality diagnostic and medication, and the implementation of targeted physician training. Concurrently, the establishment of a surveillance system and hospital water system management standard will be instrumental in preventing the incidence and outbreaks of waterborne nosocomial infections in the future.
We strongly recommend a revision of the existing hospital IPC guidelines to proactively mitigate the risk of waterborne infections within healthcare settings. The proposed revision should specifically incorporate evidence-based strategies for the prevention of waterborne infections. This proactive approach is essential because once established, waterborne pathogens like Legionella can be difficult and costly to eradicate from hospital waters, and can pose significant health risks, including severe pneumonia and even mortality.
Future research should broaden the study’s scope to encompass a wide range of hospitals, particularly in rural areas, and include community-acquired LD. Employing a mixed-methods approach, integrating surveys with laboratory tests would enhance triangulation of findings and mitigate potential bias.
Footnotes
Acknowledgements
The authors would like to extend sincere appreciation to physicians working at TASH and St. Paul’s Hospital Millennium Medical College for agreeing to be part of this study and share their perspectives. We would like to thank the administration teams of both hospitals for approval to carry out this study within their premises. The authors gratefully acknowledge Jonathan Drewry for his valuable contribution in reviewing and editing the language to achieve professional academic standards.
ORCID iDs
Ethical considerations
The College of Health Sciences Institutional Review Board (IRB) at Addis Ababa University approved our interview protocol (reference number: 077/21/SPH) on February 9, 2022. All respondents provided written informed consent before the commencement of interviews.
Consent for publication
Written informed consent was obtained from interviewed physicians for the collection, analysis, and subsequent publication of their responses.
Author contributions
Etsub Brhanesilassie Hailemichael: conceptualization, methodology, validation, investigation, data curation, validation, analysis, write up, review, editing and visualization; Adey Feleke Desta: conceptualization, methodology, review and supervision; Girma Taye: conceptualization, methodology, review and validation; Sirak Robel Gari: conceptualization, validation and supervision; Sibhatu Biadgilign: methodology, validation, analysis, editing, review; Wondwossen Amogne: conceptualization, methodology, validation, review and supervision.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The dataset used for this study can be made available upon request. The corresponding author will provide access to the study datasets upon reasonable request.
